All Exams > NEET > 30-Day Revision Course for NEET > All Questions
All questions of Coordination Compounds for NEET Exam
The correct IUPAC name of the complex Fe(C5H5)2 is _- a)Cyclopentadienyl iron (II)
- b)Bis (cyclopentadienyl) iron (II)
- c)Dicyclopentadiency ferrate (II)
- d)Ferrocene
Correct answer is option 'B'. Can you explain this answer?
The correct IUPAC name of the complex Fe(C5H5)2 is _
a)
Cyclopentadienyl iron (II)
b)
Bis (cyclopentadienyl) iron (II)
c)
Dicyclopentadiency ferrate (II)
d)
Ferrocene
| | Alok Mehta answered |
The iron complex may be treated as cationic part, and C5H5- is a bidentate ligand therefore name can be assigned as follows “dicyclopentadienyl Iron (II) cation”.
The hybrisation of Co in [Co(H2O)6]3+ is :- a)d2sp3
- b)dsp2
- c)dsp3
- d)spd3
Correct answer is option 'A'. Can you explain this answer?
The hybrisation of Co in [Co(H2O)6]3+ is :
a)
d2sp3
b)
dsp2
c)
dsp3
d)
spd3
| | Rajeev Saxena answered |
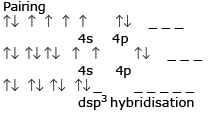
In this complex compound the total charge is +3 as H2O is a neutral compound so the oxidation state of cobalt is +3 and the electronic configuration of Co is 3d7 4s2. So, Co(+3)=4d6 and H2O is a weak ligand so there is no pairing of electron. So,4s 4p3 and 4d2 orbital make hybrid orbital to have a hybridization of d2sp3.
In the complex PtCl4.3NH3 the number of ionisable chlorines is- a)1
- b)3
- c)2
- d)0
Correct answer is option 'B'. Can you explain this answer?
In the complex PtCl4.3NH3 the number of ionisable chlorines is
a)
1
b)
3
c)
2
d)
0
 | Srishti Kaur answered |
Pt has coordination number of 4 so 3 chlorine will come outside the coordination sphere.
In the formation of complex entity, the central atom/ion acts as- a)Bronsted acid
- b)Lewis base
- c)Lewis acid
- d)Bronsted base
Correct answer is option 'C'. Can you explain this answer?
In the formation of complex entity, the central atom/ion acts as
a)
Bronsted acid
b)
Lewis base
c)
Lewis acid
d)
Bronsted base
| | Arka Das answered |
Explanation:
In the formation of a complex entity, a central atom/ion acts as a Lewis acid. This can be explained as follows:
Lewis Acid and Lewis Base:
According to Lewis acid-base theory, a Lewis acid is a species that accepts a pair of electrons to form a coordinate covalent bond, while a Lewis base is a species that donates a pair of electrons to form a coordinate covalent bond.
Formation of Complex Entity:
A complex entity is formed by the coordination of a central atom/ion with one or more ligands. Ligands are molecules or ions that donate a pair of electrons to the central atom/ion to form a coordinate covalent bond.
Role of Central Atom/Ion:
In the formation of a complex entity, the central atom/ion acts as a Lewis acid because it accepts a pair of electrons from the ligands to form a coordinate covalent bond. The central atom/ion has an incomplete outer shell, which makes it electron deficient and thus able to accept electrons from other species.
Examples:
Some examples of complex entities and their central atom/ion are as follows:
- In [Fe(CN)6]4-, Fe2+ acts as the central ion, which accepts electrons from the CN- ligands.
- In [Cu(NH3)4]2+, Cu2+ acts as the central ion, which accepts electrons from the NH3 ligands.
- In [Ag(NH3)2]+, Ag+ acts as the central ion, which accepts electrons from the NH3 ligands.
Conclusion:
Thus, we can conclude that in the formation of a complex entity, the central atom/ion acts as a Lewis acid because it accepts a pair of electrons from the ligands to form a coordinate covalent bond.
In the formation of a complex entity, a central atom/ion acts as a Lewis acid. This can be explained as follows:
Lewis Acid and Lewis Base:
According to Lewis acid-base theory, a Lewis acid is a species that accepts a pair of electrons to form a coordinate covalent bond, while a Lewis base is a species that donates a pair of electrons to form a coordinate covalent bond.
Formation of Complex Entity:
A complex entity is formed by the coordination of a central atom/ion with one or more ligands. Ligands are molecules or ions that donate a pair of electrons to the central atom/ion to form a coordinate covalent bond.
Role of Central Atom/Ion:
In the formation of a complex entity, the central atom/ion acts as a Lewis acid because it accepts a pair of electrons from the ligands to form a coordinate covalent bond. The central atom/ion has an incomplete outer shell, which makes it electron deficient and thus able to accept electrons from other species.
Examples:
Some examples of complex entities and their central atom/ion are as follows:
- In [Fe(CN)6]4-, Fe2+ acts as the central ion, which accepts electrons from the CN- ligands.
- In [Cu(NH3)4]2+, Cu2+ acts as the central ion, which accepts electrons from the NH3 ligands.
- In [Ag(NH3)2]+, Ag+ acts as the central ion, which accepts electrons from the NH3 ligands.
Conclusion:
Thus, we can conclude that in the formation of a complex entity, the central atom/ion acts as a Lewis acid because it accepts a pair of electrons from the ligands to form a coordinate covalent bond.
From the stability constant (hypothetical values), given below, predict which is the strongest ligand:- a)Cu2+ + 4NH3
 [Cu(NH3)4]2+, K = 4.5 × 1011
[Cu(NH3)4]2+, K = 4.5 × 1011 - b)Cu2+ + 4CN-
 [Cu(CN)4]2- , K = 2.0 × 1027
[Cu(CN)4]2- , K = 2.0 × 1027 - c)Cu2+ + 2en
 [Cu(en)2]2+, K = 3.0 × 1015
[Cu(en)2]2+, K = 3.0 × 1015 - d)Cu2+ + 4H2O
 [Cu(H2O)4]2+, K = 9.5 × 108
[Cu(H2O)4]2+, K = 9.5 × 108
Correct answer is option 'B'. Can you explain this answer?
From the stability constant (hypothetical values), given below, predict which is the strongest ligand:
a)
Cu2+ + 4NH3  [Cu(NH3)4]2+, K = 4.5 × 1011
[Cu(NH3)4]2+, K = 4.5 × 1011
b)
Cu2+ + 4CN-  [Cu(CN)4]2- , K = 2.0 × 1027
[Cu(CN)4]2- , K = 2.0 × 1027
c)
Cu2+ + 2en  [Cu(en)2]2+, K = 3.0 × 1015
[Cu(en)2]2+, K = 3.0 × 1015
d)
Cu2+ + 4H2O  [Cu(H2O)4]2+, K = 9.5 × 108
[Cu(H2O)4]2+, K = 9.5 × 108
 | Raghav Yadav answered |
Higher the value of K higher will be strength of ligand & more will be thermodynamic stability of complex produced.
KCl.MgCl2.6H2O is a- a)Double salt
- b)Mixed salt
- c)Basic salt
- d)Complex salt
Correct answer is option 'A'. Can you explain this answer?
KCl.MgCl2.6H2O is a
a)
Double salt
b)
Mixed salt
c)
Basic salt
d)
Complex salt
 | Srestha Choudhury answered |
KCl.MgCl2.6H2O is double salt
For the complex ion dichlorido bis (ethylene diamine) cobalt (III), select the correct statement.- a)It has three isomers, two of them are optically active and one is optically inactive.
- b)It has three isomers, all of them are optically active.
- c)It has three isomers, all of them are optically inactive.
- d)It has one optically active isomer and two geometrical isomers.
Correct answer is option 'A'. Can you explain this answer?
For the complex ion dichlorido bis (ethylene diamine) cobalt (III), select the correct statement.
a)
It has three isomers, two of them are optically active and one is optically inactive.
b)
It has three isomers, all of them are optically active.
c)
It has three isomers, all of them are optically inactive.
d)
It has one optically active isomer and two geometrical isomers.
 | Milan Datta answered |
Explanation:
The complex ion dichlorido bis(ethylene diamine) cobalt (III) has the formula [Co(en)2Cl2]+. Let's analyze the structure and isomers of this complex ion to determine the correct statement.
Structure:
The central cobalt (III) ion is coordinated by two ethylenediamine (en) ligands and two chloride (Cl) ligands. The ethylenediamine ligand is a bidentate ligand, meaning that it can form two coordination bonds with the central metal ion. The chloride ligand is a monodentate ligand, meaning that it can form only one coordination bond with the central metal ion.
Isomers:
Isomers are compounds that have the same chemical formula but different structural arrangements. In this case, there are three possible isomers for the complex ion [Co(en)2Cl2]+.
1. Geometrical isomerism:
Geometrical isomerism occurs when there is restricted rotation around a bond, resulting in different spatial arrangements. In the case of [Co(en)2Cl2]+, there are two possible geometrical isomers:
- cis-[Co(en)2Cl2]+: In this isomer, the two ethylenediamine ligands are adjacent to each other, and the two chloride ligands are also adjacent to each other.
- trans-[Co(en)2Cl2]+: In this isomer, the two ethylenediamine ligands are opposite to each other, and the two chloride ligands are also opposite to each other.
2. Optical isomerism:
Optical isomerism occurs when a compound is chiral, meaning it does not possess a plane of symmetry. Chiral compounds exist in two forms known as enantiomers, which are mirror images of each other. In the case of [Co(en)2Cl2]+, there is only one chiral center, which is the cobalt ion.
- Each geometrical isomer can exist as either an R or S enantiomer, resulting in two optically active isomers.
- The trans-[Co(en)2Cl2]+ isomer is optically inactive because its mirror image can be superimposed on it.
Conclusion:
Based on the analysis, the correct statement is option A. The complex ion dichlorido bis(ethylene diamine) cobalt (III) has three isomers, two of them are optically active (cis-[Co(en)2Cl2]+ R and S enantiomers) and one is optically inactive (trans-[Co(en)2Cl2]+).
The complex ion dichlorido bis(ethylene diamine) cobalt (III) has the formula [Co(en)2Cl2]+. Let's analyze the structure and isomers of this complex ion to determine the correct statement.
Structure:
The central cobalt (III) ion is coordinated by two ethylenediamine (en) ligands and two chloride (Cl) ligands. The ethylenediamine ligand is a bidentate ligand, meaning that it can form two coordination bonds with the central metal ion. The chloride ligand is a monodentate ligand, meaning that it can form only one coordination bond with the central metal ion.
Isomers:
Isomers are compounds that have the same chemical formula but different structural arrangements. In this case, there are three possible isomers for the complex ion [Co(en)2Cl2]+.
1. Geometrical isomerism:
Geometrical isomerism occurs when there is restricted rotation around a bond, resulting in different spatial arrangements. In the case of [Co(en)2Cl2]+, there are two possible geometrical isomers:
- cis-[Co(en)2Cl2]+: In this isomer, the two ethylenediamine ligands are adjacent to each other, and the two chloride ligands are also adjacent to each other.
- trans-[Co(en)2Cl2]+: In this isomer, the two ethylenediamine ligands are opposite to each other, and the two chloride ligands are also opposite to each other.
2. Optical isomerism:
Optical isomerism occurs when a compound is chiral, meaning it does not possess a plane of symmetry. Chiral compounds exist in two forms known as enantiomers, which are mirror images of each other. In the case of [Co(en)2Cl2]+, there is only one chiral center, which is the cobalt ion.
- Each geometrical isomer can exist as either an R or S enantiomer, resulting in two optically active isomers.
- The trans-[Co(en)2Cl2]+ isomer is optically inactive because its mirror image can be superimposed on it.
Conclusion:
Based on the analysis, the correct statement is option A. The complex ion dichlorido bis(ethylene diamine) cobalt (III) has three isomers, two of them are optically active (cis-[Co(en)2Cl2]+ R and S enantiomers) and one is optically inactive (trans-[Co(en)2Cl2]+).
Which of the following is π complex :- a)Trimethyl aluminium
- b)Ferrocene
- c)Diethyl zinc
- d)Nickel tetra carbonyl
Correct answer is option 'B'. Can you explain this answer?
Which of the following is π complex :
a)
Trimethyl aluminium
b)
Ferrocene
c)
Diethyl zinc
d)
Nickel tetra carbonyl
 | Nidhi Nambiar answered |
Al(C2H5)3 σ - complex
Fe(C5H5)2 π - complex
Zn(C2H5)2 σ - complex
[Ni(CO)4] σ - complex
Fe(C5H5)2 π - complex
Zn(C2H5)2 σ - complex
[Ni(CO)4] σ - complex
In the complex Fe(CO)x, the value of x is- a)3
- b)2
- c)4
- d)5
Correct answer is option 'D'. Can you explain this answer?
In the complex Fe(CO)x, the value of x is
a)
3
b)
2
c)
4
d)
5
 | Maitri Sharma answered |
Complex carbonyls follow Sidwick's EAN rule i.e compound with EAN 36 will be relatively more stable than other metal carbonyls. Iron pentacarbonyl has EAN number of 36 = Z−X+Y = (26−0+2x)
[Z = atomic number, X = oxidation state of metal, Y= total electrons donated by ligand]
∴x = 5. So, the formula will be Fe(CO)5.
[Z = atomic number, X = oxidation state of metal, Y= total electrons donated by ligand]
∴x = 5. So, the formula will be Fe(CO)5.
In the complex Fe(CO)x, the value of x is :- a)3
- b)4
- c)5
- d)6
Correct answer is option 'C'. Can you explain this answer?
In the complex Fe(CO)x, the value of x is :
a)
3
b)
4
c)
5
d)
6
 | Aarya Dasgupta answered |
[Fe(CO)x]0
EAN = 26 + 2x = 36
x = 5
EAN = 26 + 2x = 36
x = 5
The structure of iron pentacarbonyl is :- a)Square pyramidal
- b)Trigonal bipyramidal
- c)Squrare planar
- d)None of these
Correct answer is option 'B'. Can you explain this answer?
The structure of iron pentacarbonyl is :
a)
Square pyramidal
b)
Trigonal bipyramidal
c)
Squrare planar
d)
None of these
| | Geetika Shah answered |
[Fe(CO)5] TBP
CO is strong field ligand
Fe - 3d64s2
CO is strong field ligand
Fe - 3d64s2
The IUPAC name [CoCl(NO2)(en)2]Cl is- a)Chloridonitrobis (ethylenediamine) cobalt(II) chloride
- b)Chloridobis (ethylenediamine) nitrito-N-cobalt(III) chloride
- c)Bis(ethylenediamine) chloronitrocobalt(III) chloride
- d)Chloridonitrobis (ethylenediamine)cobalt(III) chloride
Correct answer is option 'B'. Can you explain this answer?
The IUPAC name [CoCl(NO2)(en)2]Cl is
a)
Chloridonitrobis (ethylenediamine) cobalt(II) chloride
b)
Chloridobis (ethylenediamine) nitrito-N-cobalt(III) chloride
c)
Bis(ethylenediamine) chloronitrocobalt(III) chloride
d)
Chloridonitrobis (ethylenediamine)cobalt(III) chloride
 | Baishali Chakraborty answered |
Name of ligands name first in alphabetical order followed by anme of central ion.
Which among the following has square pyramidal geometry?- a)Tetracarbonylnickel(0)
- b)Hexaamminecobalt(II) nitrate
- c)Pentacarbonyliron(0)
- d)Bis(acetylacetonato)oxovanadium(IV)
Correct answer is option 'D'. Can you explain this answer?
Which among the following has square pyramidal geometry?
a)
Tetracarbonylnickel(0)
b)
Hexaamminecobalt(II) nitrate
c)
Pentacarbonyliron(0)
d)
Bis(acetylacetonato)oxovanadium(IV)
 | Malavika Shah answered |
Bis(acetylacetonato)oxovanadium(IV) has square pyramidal geometry.
The oxidation state of Mo in its oxo-complex species [Mo2O4(C2H4 )2(H2O)2]2- is :- a)+2
- b)+3
- c)+4
- d)+5
Correct answer is option 'B'. Can you explain this answer?
The oxidation state of Mo in its oxo-complex species [Mo2O4(C2H4 )2(H2O)2]2- is :
a)
+2
b)
+3
c)
+4
d)
+5
 | Charvi Ahuja answered |
Let the O. N. of Mo in the complex is x
2x + (–2) × 2 + 0 × 2 + 0 × 2 = – 2
2x + (–2) × 2 + 0 × 2 + 0 × 2 = – 2
The correct IUPAC name of Mn3(C0)12 is- a)Dodecacarbonylmanganate(0)
- b)Manganicdodecacarbonyl(0)
- c)Dodecacarbonylmanganese(0)
- d)Dodecacarbonylmaganic(II)
Correct answer is option 'C'. Can you explain this answer?
The correct IUPAC name of Mn3(C0)12 is
a)
Dodecacarbonylmanganate(0)
b)
Manganicdodecacarbonyl(0)
c)
Dodecacarbonylmanganese(0)
d)
Dodecacarbonylmaganic(II)
 | Kunal Pillai answered |
Ligands are named 1st followed by the name of central metal ion.
The EAN of platinum in potassium hexachloroplatinate (IV) is :- a)46
- b)86
- c)36
- d)84
Correct answer is option 'B'. Can you explain this answer?
The EAN of platinum in potassium hexachloroplatinate (IV) is :
a)
46
b)
86
c)
36
d)
84
 | Dishani Kulkarni answered |
K2+4[PtCl6]
At No. Pt = 78
EAN = 78 – 4 + 6 × 2 = 86
At No. Pt = 78
EAN = 78 – 4 + 6 × 2 = 86
In which of the following complexes the nickel metal is in highest oxidation state.- a)Ni(CO)4
- b)[Cr(NH3)6]2[NiF 6]3
- c)[Ni(NH3)6](BF4) 2
- d)K4[Ni(CN)6]
Correct answer is option 'B'. Can you explain this answer?
In which of the following complexes the nickel metal is in highest oxidation state.
a)
Ni(CO)4
b)
[Cr(NH3)6]2[NiF 6]3
c)
[Ni(NH3)6](BF4) 2
d)
K4[Ni(CN)6]
 | Milan Datta answered |
Introduction:
In coordination chemistry, the oxidation state of a metal ion refers to the charge that the metal ion would have if all the ligands were removed along with the electron pairs that were shared with the ligands. The oxidation state of a metal ion can range from positive to negative depending on the number of electrons it has gained or lost.
Explanation:
To determine the oxidation state of nickel in each complex, we need to consider the oxidation states of the other elements and the overall charge of the complex.
a) Ni(CO)4:
In this complex, each carbon monoxide (CO) ligand is considered neutral because carbon has an oxidation state of +2 and oxygen has an oxidation state of -2. Therefore, the overall charge of the complex is 0. Since there are no other ligands present, the oxidation state of nickel must be 0.
b) [Cr(NH3)6]2[NiF6]3:
In this complex, the oxidation state of chromium is +3 since each ammonia (NH3) ligand is neutral and the overall charge of the complex is 2+. The oxidation state of fluorine is -1. Therefore, to balance the charges, the oxidation state of nickel must be +3.
c) [Ni(NH3)6](BF4)2:
In this complex, the oxidation state of boron in the tetrafluoroborate (BF4) ion is +3, and the oxidation state of fluorine is -1. Since the overall charge of the complex is 0, the oxidation state of nickel must be +2 to balance the charges.
d) K4[Ni(CN)6]:
In this complex, the oxidation state of potassium is +1. The cyanide (CN) ligand is considered neutral, with carbon having an oxidation state of +2 and nitrogen having an oxidation state of -3. Therefore, to balance the charges, the oxidation state of nickel must be +2.
Conclusion:
Among the given complexes, the complex [Cr(NH3)6]2[NiF6]3 has the highest oxidation state of nickel, which is +3.
In coordination chemistry, the oxidation state of a metal ion refers to the charge that the metal ion would have if all the ligands were removed along with the electron pairs that were shared with the ligands. The oxidation state of a metal ion can range from positive to negative depending on the number of electrons it has gained or lost.
Explanation:
To determine the oxidation state of nickel in each complex, we need to consider the oxidation states of the other elements and the overall charge of the complex.
a) Ni(CO)4:
In this complex, each carbon monoxide (CO) ligand is considered neutral because carbon has an oxidation state of +2 and oxygen has an oxidation state of -2. Therefore, the overall charge of the complex is 0. Since there are no other ligands present, the oxidation state of nickel must be 0.
b) [Cr(NH3)6]2[NiF6]3:
In this complex, the oxidation state of chromium is +3 since each ammonia (NH3) ligand is neutral and the overall charge of the complex is 2+. The oxidation state of fluorine is -1. Therefore, to balance the charges, the oxidation state of nickel must be +3.
c) [Ni(NH3)6](BF4)2:
In this complex, the oxidation state of boron in the tetrafluoroborate (BF4) ion is +3, and the oxidation state of fluorine is -1. Since the overall charge of the complex is 0, the oxidation state of nickel must be +2 to balance the charges.
d) K4[Ni(CN)6]:
In this complex, the oxidation state of potassium is +1. The cyanide (CN) ligand is considered neutral, with carbon having an oxidation state of +2 and nitrogen having an oxidation state of -3. Therefore, to balance the charges, the oxidation state of nickel must be +2.
Conclusion:
Among the given complexes, the complex [Cr(NH3)6]2[NiF6]3 has the highest oxidation state of nickel, which is +3.
The hybridization of nickel in tetracarbonyl nickel is- a)sp3
- b)sp3d2
- c)dsp2
- d)sp3d
Correct answer is option 'A'. Can you explain this answer?
The hybridization of nickel in tetracarbonyl nickel is
a)
sp3
b)
sp3d2
c)
dsp2
d)
sp3d
 | Ashwini Chakraborty answered |
The hybridization of an atom refers to the mixing of atomic orbitals to form new hybrid orbitals, which have different shapes and energies than the original atomic orbitals. In the case of tetracarbonyl nickel, the nickel atom is bonded to four carbon monoxide (CO) ligands.
The central nickel atom in tetracarbonyl nickel is surrounded by four ligands, which means it has a coordination number of 4. In order to determine the hybridization of the nickel atom, we can use the concept of the Valence Shell Electron Pair Repulsion (VSEPR) theory.
According to VSEPR theory, the electron pairs around the central atom will arrange themselves in a way that maximizes the distance between them, resulting in a geometry that minimizes electron-electron repulsion. In the case of tetracarbonyl nickel, the four CO ligands are arranged in a symmetrical tetrahedral geometry around the central nickel atom.
To determine the hybridization of the nickel atom, we can count the number of electron pairs around it. In tetracarbonyl nickel, there are four sigma bonds between the nickel atom and the four CO ligands, as well as four lone pairs on the nickel atom. This gives a total of eight electron pairs around the nickel atom.
The hybridization of an atom is determined by the number of sigma bonds and lone pairs around it. In this case, since there are four sigma bonds and four lone pairs, the nickel atom in tetracarbonyl nickel is sp3 hybridized.
The sp3 hybrid orbitals are formed by the mixing of one s orbital and three p orbitals on the nickel atom. These hybrid orbitals are oriented in a tetrahedral arrangement, pointing towards the four CO ligands.
In summary, the hybridization of the nickel atom in tetracarbonyl nickel is sp3, as there are four sigma bonds and four lone pairs around the central atom.
The central nickel atom in tetracarbonyl nickel is surrounded by four ligands, which means it has a coordination number of 4. In order to determine the hybridization of the nickel atom, we can use the concept of the Valence Shell Electron Pair Repulsion (VSEPR) theory.
According to VSEPR theory, the electron pairs around the central atom will arrange themselves in a way that maximizes the distance between them, resulting in a geometry that minimizes electron-electron repulsion. In the case of tetracarbonyl nickel, the four CO ligands are arranged in a symmetrical tetrahedral geometry around the central nickel atom.
To determine the hybridization of the nickel atom, we can count the number of electron pairs around it. In tetracarbonyl nickel, there are four sigma bonds between the nickel atom and the four CO ligands, as well as four lone pairs on the nickel atom. This gives a total of eight electron pairs around the nickel atom.
The hybridization of an atom is determined by the number of sigma bonds and lone pairs around it. In this case, since there are four sigma bonds and four lone pairs, the nickel atom in tetracarbonyl nickel is sp3 hybridized.
The sp3 hybrid orbitals are formed by the mixing of one s orbital and three p orbitals on the nickel atom. These hybrid orbitals are oriented in a tetrahedral arrangement, pointing towards the four CO ligands.
In summary, the hybridization of the nickel atom in tetracarbonyl nickel is sp3, as there are four sigma bonds and four lone pairs around the central atom.
Which of the following complex exhibits geometrical isomerism:- a)[Zn(gly)2]
- b)[Cu(en)(NH3)2]+
- c)[PtBrCl(NH3)(py)]
- d)[Ni(CN)2(CO)2]2-
Correct answer is option 'C'. Can you explain this answer?
Which of the following complex exhibits geometrical isomerism:
a)
[Zn(gly)2]
b)
[Cu(en)(NH3)2]+
c)
[PtBrCl(NH3)(py)]
d)
[Ni(CN)2(CO)2]2-
 | Nidhi Nambiar answered |
[PtBrCl NH3 Py] Pt2+ is dsp2 hybridised & hence geometry is square planner & sq. planner complex with four different ligands shows geometrical isomerism.
The IUPAC name of AlCl3.4(EtOH) is- a)Aluminium(III) chloride-4-ethanol
- b)Aluminium (IV) chloride-4-hydroxyethane
- c)Aluminium (II) chloride-4-ethanol
- d)Aluminium chloride-4-ethanol
Correct answer is option 'A'. Can you explain this answer?
The IUPAC name of AlCl3.4(EtOH) is
a)
Aluminium(III) chloride-4-ethanol
b)
Aluminium (IV) chloride-4-hydroxyethane
c)
Aluminium (II) chloride-4-ethanol
d)
Aluminium chloride-4-ethanol
 | Ashish Nambiar answered |
This is double salt so name is written in sequential manner.
Type of isomerism exhibited by [Cr(NCS)(NH3)5] [ZnCl4] :- a)Coordination isomerism
- b)Linkage isomerism
- c)Ionization isomerism
- d)Both coordination and linkage isomerism
Correct answer is option 'D'. Can you explain this answer?
Type of isomerism exhibited by [Cr(NCS)(NH3)5] [ZnCl4] :
a)
Coordination isomerism
b)
Linkage isomerism
c)
Ionization isomerism
d)
Both coordination and linkage isomerism
 | Shail Chakraborty answered |
Since both cation & anion constitute coordination sphere so it exhibit coordination isomerism and contains ambident ligand so, it shows linkage isomerism
According to Werner’s theory , the primary valences of the central atom- a)Are equal to its coordination number
- b)Decide the geometry of the complex
- c)Are satisfied by negative ions
- d)Are satisfied by negative ions or neutral molecules
Correct answer is option 'C'. Can you explain this answer?
According to Werner’s theory , the primary valences of the central atom
a)
Are equal to its coordination number
b)
Decide the geometry of the complex
c)
Are satisfied by negative ions
d)
Are satisfied by negative ions or neutral molecules
 | Nidhi Nambiar answered |
Primary valency is equal to no. of ligands attachted which are negatively charges.
Which of the following ligand gives chelate complexes?- a)NH3
- b)SCN--
- c)Pyridine
- d)C2O42-
Correct answer is option 'D'. Can you explain this answer?
Which of the following ligand gives chelate complexes?
a)
NH3
b)
SCN--
c)
Pyridine
d)
C2O42-
 | Samridhi Bajaj answered |
Oxalate is a bidentate ligand.
Which complex is likely to show optical activity- a)Trans-[CoCl2(NH3)4 ]+
- b)[Cr(H2O)6]3+
- c)Cis-[Co(NH3)2(en)2 ]3+
- d)Trans-[Co(NH3)2(en)2 ]3+
Correct answer is option 'C'. Can you explain this answer?
Which complex is likely to show optical activity
a)
Trans-[CoCl2(NH3)4 ]+
b)
[Cr(H2O)6]3+
c)
Cis-[Co(NH3)2(en)2 ]3+
d)
Trans-[Co(NH3)2(en)2 ]3+
 | Dishani Kulkarni answered |
Cis [Co(NH3)2(en)2]3+
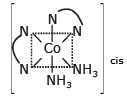
No POS optically active rest compound has POS.
No POS optically active rest compound has POS.
Trioxalato aluminate (III) and tetrafluorido-borate (III) ions are respectively :- a)[Al(C2O4)3], [BF4]3-
- b)[Al(C2O4)3]3+ , [BF4]3+
- c)[Al(C2O4)3]3- , [BF4]-
- d)[Al(C2O4)3]2- , [BF4]2-
Correct answer is option 'C'. Can you explain this answer?
Trioxalato aluminate (III) and tetrafluorido-borate (III) ions are respectively :
a)
[Al(C2O4)3], [BF4]3-
b)
[Al(C2O4)3]3+ , [BF4]3+
c)
[Al(C2O4)3]3- , [BF4]-
d)
[Al(C2O4)3]2- , [BF4]2-
| | Maya Reddy answered |
Trioxalato aluminate (III) and tetrafluorido-borate (III) ions
- Trioxalato aluminate (III) ion: [Al(C2O4)3]^3-
- Tetrafluorido-borate (III) ion: [BF4]^-
Correct combination
The correct combination of these ions is option 'C', which is [Al(C2O4)3]^3- and [BF4]^-. This means that the trioxalato aluminate (III) ion has a 3- charge and the tetrafluorido-borate (III) ion has a 1- charge.
Explanation
- The trioxalato aluminate (III) ion consists of one aluminum ion (Al^3+) complexed with three oxalate ions (C2O4^2-) to balance the charge.
- The tetrafluorido-borate (III) ion consists of one boron ion (B^3+) complexed with four fluoride ions (F-) to balance the charge.
- Therefore, the correct notation for these ions is [Al(C2O4)3]^3- and [BF4]^-.
A complex compound in which the oxidation number of a metal is zero is- a)K4 [Fe (CN)6]
- b)K3 [Fe (CN)6]
- c)[Ni (CO)4]
- d)[Pl (NH3 )4]Cl2
Correct answer is option 'C'. Can you explain this answer?
A complex compound in which the oxidation number of a metal is zero is
a)
K4 [Fe (CN)6]
b)
K3 [Fe (CN)6]
c)
[Ni (CO)4]
d)
[Pl (NH3 )4]Cl2
| | Sankar Singh answered |
A complex compound in which the oxidation number of metal is zero is [Ni(CO)4]. In this complex, the oxidation number of both metal and ligand is zero.
The oxidation number of metal in the complexes K4[Fe(CN)6], K3[Fe(CN)6] and [Pt(NH3)4]Cl2 are +2, +3 and +2 respectively.
The oxidation number of metal in the complexes K4[Fe(CN)6], K3[Fe(CN)6] and [Pt(NH3)4]Cl2 are +2, +3 and +2 respectively.
Which is not true about a ligand?- a)A monodentate ligand cannot be chelating ligand
- b)A multidentate ligand cannot cause chelation
- c)It can be monodentate of multidentate
- d)It can act as a Lewis Base
Correct answer is option 'B'. Can you explain this answer?
Which is not true about a ligand?
a)
A monodentate ligand cannot be chelating ligand
b)
A multidentate ligand cannot cause chelation
c)
It can be monodentate of multidentate
d)
It can act as a Lewis Base
 | Anu Basu answered |
Multidentate ligand always act as chelating Ligand
Which of the following can be termed as mixed complex?- a)[Co(NH3)4NO2Cl]Cl
- b)K4[Fe(CN)6]
- c)K2FeSO4
- d)[Cu(NH4)]SO4
Correct answer is option 'A'. Can you explain this answer?
Which of the following can be termed as mixed complex?
a)
[Co(NH3)4NO2Cl]Cl
b)
K4[Fe(CN)6]
c)
K2FeSO4
d)
[Cu(NH4)]SO4
 | Niti Saha answered |
In A one and more different types of ligands are present.
Which is used in cancer therapy?- a)Cyanocobalamine
- b)Cis-Platin
- c)Zeise’s salt
- d)EDTA
Correct answer is option 'B'. Can you explain this answer?
Which is used in cancer therapy?
a)
Cyanocobalamine
b)
Cis-Platin
c)
Zeise’s salt
d)
EDTA
 | Vandana Menon answered |
Cis Platin (cis – [Pt(NH3)2(Cl)2] is a anti cancer drug.
Type of isomerism exhibited by [Ir(OCN)2(H2O)3]- a)Hydrate isomerism
- b)Linkage isomerism
- c)Polymerization isomerism
- d)Both (B) and (C)
Correct answer is option 'D'. Can you explain this answer?
Type of isomerism exhibited by [Ir(OCN)2(H2O)3]
a)
Hydrate isomerism
b)
Linkage isomerism
c)
Polymerization isomerism
d)
Both (B) and (C)
| | Sankar Singh answered |
Explanation:
The given complex is [Ir(OCN)2(H2O)3]. It has two isomers, i.e., linkage and polymerization isomers.
Linkage Isomerism:
In linkage isomerism, the ligand can coordinate through a different atom of the same ligand to form a different compound. Here, the OCN^- ion can coordinate via nitrogen (N) or carbon (C).
- If OCN^- binds through N atom, it forms a linkage isomer.
- If OCN^- binds through C atom, it forms a normal isomer.
Hence, [Ir(OCN-N)2(H2O)3] and [Ir(OCN-C)2(H2O)3] are linkage isomers.
Polymerization Isomerism:
In polymerization isomerism, the ligand can have different bridging modes between two metal centers. Here, the OCN^- ion can act as a bridging ligand to form a polymer chain.
- If OCN^- bridges between two Ir centers, it forms a polymerization isomer.
- If OCN^- binds to only one Ir center, it forms a normal isomer.
Hence, [Ir(OCN)2(H2O)3] and [Ir(OCN)2(μ-OCN)(H2O)2] are polymerization isomers.
Conclusion:
Thus, the given complex exhibits both linkage and polymerization isomerism.
The given complex is [Ir(OCN)2(H2O)3]. It has two isomers, i.e., linkage and polymerization isomers.
Linkage Isomerism:
In linkage isomerism, the ligand can coordinate through a different atom of the same ligand to form a different compound. Here, the OCN^- ion can coordinate via nitrogen (N) or carbon (C).
- If OCN^- binds through N atom, it forms a linkage isomer.
- If OCN^- binds through C atom, it forms a normal isomer.
Hence, [Ir(OCN-N)2(H2O)3] and [Ir(OCN-C)2(H2O)3] are linkage isomers.
Polymerization Isomerism:
In polymerization isomerism, the ligand can have different bridging modes between two metal centers. Here, the OCN^- ion can act as a bridging ligand to form a polymer chain.
- If OCN^- bridges between two Ir centers, it forms a polymerization isomer.
- If OCN^- binds to only one Ir center, it forms a normal isomer.
Hence, [Ir(OCN)2(H2O)3] and [Ir(OCN)2(μ-OCN)(H2O)2] are polymerization isomers.
Conclusion:
Thus, the given complex exhibits both linkage and polymerization isomerism.
In which complex is the transition metal in zero oxidation state:- a)[Co(NH3)6Cl2]
- b)[Fe(H2O)6]SO4
- c)H[Co(CO)4]
- d)K4[Ni(CN)4]
Correct answer is option 'D'. Can you explain this answer?
In which complex is the transition metal in zero oxidation state:
a)
[Co(NH3)6Cl2]
b)
[Fe(H2O)6]SO4
c)
H[Co(CO)4]
d)
K4[Ni(CN)4]
 | Nilanjan Malik answered |
The answer to the question is option 'D' - K4[Ni(CN)4].
Explanation:
In order to determine the oxidation state of the transition metal in each complex, we need to analyze the ligands and their charge contributions.
a) [Co(NH3)6Cl2]:
- In this complex, we have six ammine (NH3) ligands and two chloride (Cl) ligands.
- Ammine is a neutral ligand, so it does not contribute any charge.
- Chloride is a negatively charged ligand, contributing a charge of -1 each. Since there are two chloride ligands, the total charge contribution is -2.
- Since the overall charge of the complex is 0, the oxidation state of the central transition metal (Co) must be +2 to balance the charge.
- Therefore, the transition metal in this complex is in the +2 oxidation state.
b) [Fe(H2O)6]SO4:
- In this complex, we have six water (H2O) ligands and a sulfate (SO4) ligand.
- Water is a neutral ligand, so it does not contribute any charge.
- Sulfate is a negatively charged ligand, contributing a charge of -2.
- Since the overall charge of the complex is 0, the oxidation state of the central transition metal (Fe) must be +2 to balance the charge.
- Therefore, the transition metal in this complex is in the +2 oxidation state.
c) H[Co(CO)4]:
- In this complex, we have four carbonyl (CO) ligands.
- Carbonyl is a neutral ligand, so it does not contribute any charge.
- Since there is no other ligand or counterion present in the complex, the overall charge of the complex is +1.
- Therefore, the oxidation state of the central transition metal (Co) must be -1 to balance the charge.
- Therefore, the transition metal in this complex is in the -1 oxidation state.
d) K4[Ni(CN)4]:
- In this complex, we have four cyanide (CN) ligands.
- Cyanide is a negatively charged ligand, contributing a charge of -1 each. Since there are four cyanide ligands, the total charge contribution is -4.
- Since the overall charge of the complex is -4 (as indicated by the K4 counterions), the oxidation state of the central transition metal (Ni) must be 0 to balance the charge.
- Therefore, the transition metal in this complex is in the 0 oxidation state.
Conclusion:
The transition metal in the complex K4[Ni(CN)4] is in the zero oxidation state.
Explanation:
In order to determine the oxidation state of the transition metal in each complex, we need to analyze the ligands and their charge contributions.
a) [Co(NH3)6Cl2]:
- In this complex, we have six ammine (NH3) ligands and two chloride (Cl) ligands.
- Ammine is a neutral ligand, so it does not contribute any charge.
- Chloride is a negatively charged ligand, contributing a charge of -1 each. Since there are two chloride ligands, the total charge contribution is -2.
- Since the overall charge of the complex is 0, the oxidation state of the central transition metal (Co) must be +2 to balance the charge.
- Therefore, the transition metal in this complex is in the +2 oxidation state.
b) [Fe(H2O)6]SO4:
- In this complex, we have six water (H2O) ligands and a sulfate (SO4) ligand.
- Water is a neutral ligand, so it does not contribute any charge.
- Sulfate is a negatively charged ligand, contributing a charge of -2.
- Since the overall charge of the complex is 0, the oxidation state of the central transition metal (Fe) must be +2 to balance the charge.
- Therefore, the transition metal in this complex is in the +2 oxidation state.
c) H[Co(CO)4]:
- In this complex, we have four carbonyl (CO) ligands.
- Carbonyl is a neutral ligand, so it does not contribute any charge.
- Since there is no other ligand or counterion present in the complex, the overall charge of the complex is +1.
- Therefore, the oxidation state of the central transition metal (Co) must be -1 to balance the charge.
- Therefore, the transition metal in this complex is in the -1 oxidation state.
d) K4[Ni(CN)4]:
- In this complex, we have four cyanide (CN) ligands.
- Cyanide is a negatively charged ligand, contributing a charge of -1 each. Since there are four cyanide ligands, the total charge contribution is -4.
- Since the overall charge of the complex is -4 (as indicated by the K4 counterions), the oxidation state of the central transition metal (Ni) must be 0 to balance the charge.
- Therefore, the transition metal in this complex is in the 0 oxidation state.
Conclusion:
The transition metal in the complex K4[Ni(CN)4] is in the zero oxidation state.
The oxidation state of Ag in tollen’s reagent is- a)+1.5
- b)0
- c)+2
- d)+1
Correct answer is option 'D'. Can you explain this answer?
The oxidation state of Ag in tollen’s reagent is
a)
+1.5
b)
0
c)
+2
d)
+1
 | Ayush Joshi answered |
The silver-producing reaction in this demonstration is one that is commonly used in basic organic laboratory classes to identify aldehydes. The reaction called "The Silver Mirror Test" or Tollens' Test is accomplished by mixing aqueous silver nitrate with aqueous ammonia to produce a solution known as Tollens' reagent. Although this solution contains only a very weak oxidizing agent, it is strong enough to oxidize the aldehyde functional group. As this oxidation occurs, silver is reduced from the +1 oxidation state to metallic silver. This metallic silver is deposited on the walls of the test tube producing a reflective "mirror".
Consider the following statements, "According the Werner's theory. :(1) Ligands are connected to the metal ions by covalent bonds.(2) Secondary valencies have directional properties.(3) Secondary valencies are non-ionisable.(4) Secondary valencies are satisfied by either neutral or negative legands.Of these statements.- a)2, 3 and 4 are correct
- b)2 and 3 are correct
- c)1 and 3 are correct
- d)1, 2 and 4 are correct
Correct answer is option 'A'. Can you explain this answer?
Consider the following statements, "According the Werner's theory. :
(1) Ligands are connected to the metal ions by covalent bonds.
(2) Secondary valencies have directional properties.
(3) Secondary valencies are non-ionisable.
(4) Secondary valencies are satisfied by either neutral or negative legands.
Of these statements.
a)
2, 3 and 4 are correct
b)
2 and 3 are correct
c)
1 and 3 are correct
d)
1, 2 and 4 are correct
 | Amrita Sarkar answered |
Consider werner’s theroy
A complex of platinum, ammonia and chloride produces four ions per molecule in the solution. The structure consistent with the observation is :- a)[Pt(NH3)4]Cl4
- b)[Pt(NH3)2Cl4]
- c)[Pt(NH3)5Cl]Cl3
- d)[Pt(NH3)4Cl2]Cl 2
Correct answer is option 'C'. Can you explain this answer?
A complex of platinum, ammonia and chloride produces four ions per molecule in the solution. The structure consistent with the observation is :
a)
[Pt(NH3)4]Cl4
b)
[Pt(NH3)2Cl4]
c)
[Pt(NH3)5Cl]Cl3
d)
[Pt(NH3)4Cl2]Cl 2
 | Anu Basu answered |
[Pt(NH3)4]Cl4 → 5 ions
[Pt(NH3)2Cl4] → 0 ions
[Pt(NH3)4Cl2]Cl2 → 3 ions
Which of the following complex will give white precipitate with barium chloride solution?- a)[Co(NH3)4Cl2]NO2
- b)[Cr(NH3)5Cl]SO4
- c)[Co(NH3)6]Br3
- d)[Cr(NH3)5SO4]Cl
Correct answer is option 'B'. Can you explain this answer?
Which of the following complex will give white precipitate with barium chloride solution?
a)
[Co(NH3)4Cl2]NO2
b)
[Cr(NH3)5Cl]SO4
c)
[Co(NH3)6]Br3
d)
[Cr(NH3)5SO4]Cl
 | Nidhi Yadav answered |
Since the reagent is BaCl2(aq), displacement reaction will take place. The resulting solution, if insoluble in water, will form a white precipitate. Both BaNO2 and BaCl2 are soluble in water and hence will not form a precipitate. BaSO4, however, is insoluble in water and hence forms a white precipitate.
Co(CO)4 follows EAN rule by :- a)Oxidizing character
- b)Reduction
- c)Dimerization
- d)All of these
Correct answer is option 'D'. Can you explain this answer?
Co(CO)4 follows EAN rule by :
a)
Oxidizing character
b)
Reduction
c)
Dimerization
d)
All of these
| | Rajeev Saxena answered |
Effective Atomic Number Rule-
[ EAN – Rule]
The total number of electrons linked with central metal atom or ion in a complex plus the electrons donated by the ligand is known as effective Atomic Number.
Sidgwick suggested that the central atom or ion will accept electron pairs from ligands till the EAN of the metal equalize itself with the atomic number of the nearest noble gas.
EAN= Z – x + 2nL
Z= Atomic number of the metal in the complex
x = Oxidation state of the metal in complex
n= Number of the ligands
L = Number of co-ordinate bonds formed by ligand
For monodentate ligand,
L=1
For Bidentate ligand
L=2
For Tridentate ligand
L=3
For Tetra dentate ligand
L=4
L=5 for Pentadentate & L = 6 for Hexadentate
The coordination number of Cr in [Cr (NH3)3 (H2O)3]- a)3
- b)6
- c)4
- d)2
Correct answer is option 'B'. Can you explain this answer?
The coordination number of Cr in [Cr (NH3)3 (H2O)3]
a)
3
b)
6
c)
4
d)
2
 | Sanchita Reddy answered |
Understanding Coordination Number
The coordination number of a metal ion in a complex refers to the number of ligand donor atoms that are bonded to it. In the case of the complex [Cr(NH3)3(H2O)3], we need to analyze the ligands attached to chromium (Cr).
Identifying Ligands
- The complex contains two types of ligands:
- Ammonia (NH3), which is a neutral ligand.
- Water (H2O), which is also a neutral ligand.
Counting the Ligands
- There are three NH3 ligands.
- There are three H2O ligands.
Calculating the Coordination Number
- To find the coordination number, we simply add the number of donor atoms from each ligand type:
- From NH3: 3 ligands x 1 donor atom each = 3
- From H2O: 3 ligands x 1 donor atom each = 3
- Total coordination number = 3 (from NH3) + 3 (from H2O) = 6
Conclusion
Thus, the coordination number of Cr in the complex [Cr(NH3)3(H2O)3] is 6. This means that option 'B' is correct. The coordination number reflects how many atoms are directly bonded to the metal ion, which in this case is chromium.
The coordination number of a metal ion in a complex refers to the number of ligand donor atoms that are bonded to it. In the case of the complex [Cr(NH3)3(H2O)3], we need to analyze the ligands attached to chromium (Cr).
Identifying Ligands
- The complex contains two types of ligands:
- Ammonia (NH3), which is a neutral ligand.
- Water (H2O), which is also a neutral ligand.
Counting the Ligands
- There are three NH3 ligands.
- There are three H2O ligands.
Calculating the Coordination Number
- To find the coordination number, we simply add the number of donor atoms from each ligand type:
- From NH3: 3 ligands x 1 donor atom each = 3
- From H2O: 3 ligands x 1 donor atom each = 3
- Total coordination number = 3 (from NH3) + 3 (from H2O) = 6
Conclusion
Thus, the coordination number of Cr in the complex [Cr(NH3)3(H2O)3] is 6. This means that option 'B' is correct. The coordination number reflects how many atoms are directly bonded to the metal ion, which in this case is chromium.
Chapter doubts & questions for Coordination Compounds - 30-Day Revision Course for NEET 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Coordination Compounds - 30-Day Revision Course for NEET in English & Hindi are available as part of NEET exam. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
;
Signup to see your scores go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup









