All Exams > NEET > 30-Day Revision Course for NEET > All Questions
All questions of Haloalkanes and Haloarenes for NEET Exam
C5H11Br is least soluble in:- a)Ether
- b)Alcohol
- c)Water
- d)None of these
Correct answer is 'C'. Can you explain this answer?
C5H11Br is least soluble in:
a)
Ether
b)
Alcohol
c)
Water
d)
None of these
| | Rajeev Saxena answered |
C5H11Br is only very slightly soluble in water. It is more soluble in organic solvents.
Which of the following is not true about optical isomers?- a)They rotate the plane polarized light.
- b)They are superimposable on their mirror image.
- c)They posses at least one chiral carbon atom.
- d)They are nonsuperimposable on their mirror image.
Correct answer is option 'B'. Can you explain this answer?
Which of the following is not true about optical isomers?
a)
They rotate the plane polarized light.
b)
They are superimposable on their mirror image.
c)
They posses at least one chiral carbon atom.
d)
They are nonsuperimposable on their mirror image.
| | Nisha Pillai answered |
Explanation:
Optical isomers are stereoisomers that exist in two mirror-image forms that are non-superimposable on each other. They are also known as enantiomers.
a) They rotate the plane polarized light: Optical isomers have the property of rotating the plane of polarized light in opposite directions. One isomer rotates the plane of polarized light in the clockwise direction and the other in the counterclockwise direction.
b) They are superimposable on their mirror image: This statement is false. Optical isomers are non-superimposable mirror images of each other. If we try to superimpose them, they will not match perfectly.
c) They possess at least one chiral carbon atom: Optical isomers possess chiral carbon atoms. Chiral carbon atoms are those carbon atoms that are attached to four different groups or atoms.
d) They are nonsuperimposable on their mirror image: As mentioned earlier, optical isomers are non-superimposable mirror images of each other. They have the same physical and chemical properties but differ in their biological activity, as they interact differently with other chiral molecules in living organisms.
Conclusion: Optical isomers are important in fields like medicinal chemistry, where knowing the activity of each isomer can help in designing drugs that are more effective and have fewer side effects. Therefore, it is crucial to understand the properties of optical isomers.
Optical isomers are stereoisomers that exist in two mirror-image forms that are non-superimposable on each other. They are also known as enantiomers.
a) They rotate the plane polarized light: Optical isomers have the property of rotating the plane of polarized light in opposite directions. One isomer rotates the plane of polarized light in the clockwise direction and the other in the counterclockwise direction.
b) They are superimposable on their mirror image: This statement is false. Optical isomers are non-superimposable mirror images of each other. If we try to superimpose them, they will not match perfectly.
c) They possess at least one chiral carbon atom: Optical isomers possess chiral carbon atoms. Chiral carbon atoms are those carbon atoms that are attached to four different groups or atoms.
d) They are nonsuperimposable on their mirror image: As mentioned earlier, optical isomers are non-superimposable mirror images of each other. They have the same physical and chemical properties but differ in their biological activity, as they interact differently with other chiral molecules in living organisms.
Conclusion: Optical isomers are important in fields like medicinal chemistry, where knowing the activity of each isomer can help in designing drugs that are more effective and have fewer side effects. Therefore, it is crucial to understand the properties of optical isomers.
Which of the following is a tertiary halogenoalkanes?- a)2-Bromopentane
- b)2-Bromo 3-methylpentane
- c)Bromopentane
- d)2-Bromo 2-methylpentane
Correct answer is option 'D'. Can you explain this answer?
Which of the following is a tertiary halogenoalkanes?
a)
2-Bromopentane
b)
2-Bromo 3-methylpentane
c)
Bromopentane
d)
2-Bromo 2-methylpentane
| | Anaya Patel answered |
√Br CH3 -( CH )- CH2 - CH2 - CH3 √CH3 The carbon with which the Br is bonded is bonded with another 3 carbon atoms. So haloalkane is 3.
Which one of the following is likely to give a precipitate with AgNO3 solution?- a)CHCl3
- b)(CH3)3CCl
- c)CH2=CH-Cl
- d)CCl4
Correct answer is option 'B'. Can you explain this answer?
Which one of the following is likely to give a precipitate with AgNO3 solution?
a)
CHCl3
b)
(CH3)3CCl
c)
CH2=CH-Cl
d)
CCl4
| | Gargi Ahuja answered |
Precipitation Reaction
A precipitation reaction is a chemical reaction that produces a precipitate, which is an insoluble solid that forms from the reaction of two soluble compounds.
AgNO3 Solution
AgNO3 is a soluble salt that dissociates to give Ag+ and NO3- ions in an aqueous solution.
Likely Precipitate
A compound is likely to give a precipitate with AgNO3 solution if it contains Cl- ions, as AgCl is insoluble in water and will form a white precipitate.
Analysis of Options
a) CHCl3
CHCl3 does not contain Cl- ions and will not give a precipitate with AgNO3 solution.
b) (CH3)3CCl
(CH3)3CCl contains Cl- ions and will give a precipitate with AgNO3 solution.
c) CH2=CH-Cl
CH2=CH-Cl does not contain Cl- ions and will not give a precipitate with AgNO3 solution.
d) CCl4
CCl4 does not contain Cl- ions and will not give a precipitate with AgNO3 solution.
Conclusion
Out of the given options, only (CH3)3CCl contains Cl- ions and will give a precipitate with AgNO3 solution, making option 'B' the correct answer.
A precipitation reaction is a chemical reaction that produces a precipitate, which is an insoluble solid that forms from the reaction of two soluble compounds.
AgNO3 Solution
AgNO3 is a soluble salt that dissociates to give Ag+ and NO3- ions in an aqueous solution.
Likely Precipitate
A compound is likely to give a precipitate with AgNO3 solution if it contains Cl- ions, as AgCl is insoluble in water and will form a white precipitate.
Analysis of Options
a) CHCl3
CHCl3 does not contain Cl- ions and will not give a precipitate with AgNO3 solution.
b) (CH3)3CCl
(CH3)3CCl contains Cl- ions and will give a precipitate with AgNO3 solution.
c) CH2=CH-Cl
CH2=CH-Cl does not contain Cl- ions and will not give a precipitate with AgNO3 solution.
d) CCl4
CCl4 does not contain Cl- ions and will not give a precipitate with AgNO3 solution.
Conclusion
Out of the given options, only (CH3)3CCl contains Cl- ions and will give a precipitate with AgNO3 solution, making option 'B' the correct answer.
Which of the following is not an aryl halide?- a)m-ClCH2C6H4CH2C(CH3)3
- b)(CH3)3CCH2CH3
- c)o-Br-C6H4CH(CH3)CH2CH3
- d)p-ClC6H4CH2CH(CH3)2
Correct answer is 'B'. Can you explain this answer?
Which of the following is not an aryl halide?
a)
m-ClCH2C6H4CH2C(CH3)3
b)
(CH3)3CCH2CH3
c)
o-Br-C6H4CH(CH3)CH2CH3
d)
p-ClC6H4CH2CH(CH3)2
| | Ankita Datta answered |
Because it is Open chain compound /Aliphatic Compound and not aryl halide as does not contain halogen atom. it's alkane simply.
Only One Option Correct TypeDirection (Q. Nos. 1-8) This section contains 8 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.Q. Which of the SN2 reaction is fastest?- a)
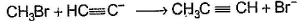
- b)
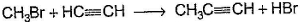
- c)
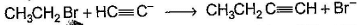
- d)
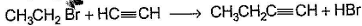
Correct answer is option 'A'. Can you explain this answer?
Only One Option Correct Type
Direction (Q. Nos. 1-8) This section contains 8 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q.
Which of the SN2 reaction is fastest?
a)
b)
c)
d)
| | Monika Devi answered |
SN2 reaction means this will be applicable for primary or 1 degree carbon atom and attack by the reagent is forwarded so on doing mechanism we getted that CH3 carry + charge & Br get - charge so acetyl group that have negative charge goes on methyl group & Br react with electronic species
Pick up the correct statement about alkyl halides.- a)They are associated with each other by H-bonds.
- b)They dissolve easily in organic solvents.
- c)They dissolve in water quickly.
- d)They do not contain any polar bond in their molecules.
Correct answer is option 'B'. Can you explain this answer?
Pick up the correct statement about alkyl halides.
a)
They are associated with each other by H-bonds.
b)
They dissolve easily in organic solvents.
c)
They dissolve in water quickly.
d)
They do not contain any polar bond in their molecules.
| | Preeti Iyer answered |
In general organic compounds are soluble in organic solvents. Hence alkyl halides are soluble in organic solvents.
One Integer Value Correct TypeDirection (Q. Nos. 18-20) This section contains 3 questions. When worked out will result in an integer from 0 to 9 (both inclusive).Consider the following reaction,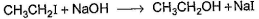 Q. The above reaction was started taking equal concentrations of ethyl iodide and NaOH. After 1.0 h, concentration of iodoethane is dropped to (1/3)rd of initial value. By what factor, the rate of reaction would have been decreased by the same time?
Q. The above reaction was started taking equal concentrations of ethyl iodide and NaOH. After 1.0 h, concentration of iodoethane is dropped to (1/3)rd of initial value. By what factor, the rate of reaction would have been decreased by the same time?
Correct answer is '9'. Can you explain this answer?
One Integer Value Correct Type
Direction (Q. Nos. 18-20) This section contains 3 questions. When worked out will result in an integer from 0 to 9 (both inclusive).
Consider the following reaction,
Q.
The above reaction was started taking equal concentrations of ethyl iodide and NaOH. After 1.0 h, concentration of iodoethane is dropped to (1/3)rd of initial value. By what factor, the rate of reaction would have been decreased by the same time?
| | Priyanka Sharma answered |
Rate = A [C2H5I] [OH-] Rate is linear function of both alkyl halide and nucleophile concentration both will decrease by same factor. Hence, if after 1.0 h concentration of iodoethane decreases to 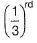 concentration of hydroxide will also decrease by the same factor and rate by
concentration of hydroxide will also decrease by the same factor and rate by 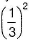 , i.e. by 9 times.
, i.e. by 9 times.
Elimination of bromine from 2-bromobutane results in the formation of- a)Predominantly 2-butene
- b)Equimolar mixture of 1 and 2-butene
- c) Predominantly 1-butene
- d)Predominantly 2-butyne.
Correct answer is option 'A'. Can you explain this answer?
Elimination of bromine from 2-bromobutane results in the formation of
a)
Predominantly 2-butene
b)
Equimolar mixture of 1 and 2-butene
c)
Predominantly 1-butene
d)
Predominantly 2-butyne.
| | Palak Basak answered |
Elimination of Bromine from 2-Bromobutane
The elimination reaction of bromine from 2-bromobutane is a type of E2 (bimolecular elimination) reaction. During this reaction, the bromine atom is removed from the carbon chain, resulting in the formation of a double bond.
E2 Reaction Mechanism
The E2 reaction occurs in a single step, involving the simultaneous removal of a proton and a leaving group (in this case, bromine) from adjacent carbon atoms. The leaving group takes a pair of electrons from the carbon-hydrogen bond, forming a double bond between the two carbon atoms.
The reaction mechanism can be summarized as follows:
1. The base (usually a strong base such as hydroxide ion, OH-) approaches the hydrogen atom on the carbon adjacent to the bromine atom.
2. The base abstracts the proton, creating a carbanion intermediate and leaving a bromide ion.
3. The carbanion intermediate then rearranges to form the most stable alkene through a transition state.
4. Finally, the bromide ion combines with a proton from the solvent to form HBr.
Stability of Alkenes
The stability of alkenes is determined by the degree of substitution at the double bond. The more substituted the double bond, the more stable the alkene. This stability is due to the increased electron density around the double bond, which is provided by the alkyl groups attached to the carbon atoms.
Explanation of the Correct Answer
In the case of 2-bromobutane, elimination of bromine results in the formation of 2-butene. This is because the double bond in 2-butene is more substituted than the double bond in 1-butene. Therefore, the stability of the alkene product, 2-butene, is higher compared to that of 1-butene.
The stability of alkenes follows the order: tertiary > secondary > primary. In 2-butene, the double bond is formed between two secondary carbons, making it more stable than 1-butene, which has a double bond between a primary and a secondary carbon.
Hence, the correct answer is option 'A' - Predominantly 2-butene.
It is important to note that while the formation of 2-butene is favored, a small amount of 1-butene may also be formed as a minor product due to the presence of a primary carbon adjacent to the double bond. However, the major product will be 2-butene.
The elimination reaction of bromine from 2-bromobutane is a type of E2 (bimolecular elimination) reaction. During this reaction, the bromine atom is removed from the carbon chain, resulting in the formation of a double bond.
E2 Reaction Mechanism
The E2 reaction occurs in a single step, involving the simultaneous removal of a proton and a leaving group (in this case, bromine) from adjacent carbon atoms. The leaving group takes a pair of electrons from the carbon-hydrogen bond, forming a double bond between the two carbon atoms.
The reaction mechanism can be summarized as follows:
1. The base (usually a strong base such as hydroxide ion, OH-) approaches the hydrogen atom on the carbon adjacent to the bromine atom.
2. The base abstracts the proton, creating a carbanion intermediate and leaving a bromide ion.
3. The carbanion intermediate then rearranges to form the most stable alkene through a transition state.
4. Finally, the bromide ion combines with a proton from the solvent to form HBr.
Stability of Alkenes
The stability of alkenes is determined by the degree of substitution at the double bond. The more substituted the double bond, the more stable the alkene. This stability is due to the increased electron density around the double bond, which is provided by the alkyl groups attached to the carbon atoms.
Explanation of the Correct Answer
In the case of 2-bromobutane, elimination of bromine results in the formation of 2-butene. This is because the double bond in 2-butene is more substituted than the double bond in 1-butene. Therefore, the stability of the alkene product, 2-butene, is higher compared to that of 1-butene.
The stability of alkenes follows the order: tertiary > secondary > primary. In 2-butene, the double bond is formed between two secondary carbons, making it more stable than 1-butene, which has a double bond between a primary and a secondary carbon.
Hence, the correct answer is option 'A' - Predominantly 2-butene.
It is important to note that while the formation of 2-butene is favored, a small amount of 1-butene may also be formed as a minor product due to the presence of a primary carbon adjacent to the double bond. However, the major product will be 2-butene.
One or More than One Options Correct TypeDirection (Q. Nos. 9-14) This section contains 6 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.Q. In a SN2 reaction rate of reaction depends on- a)concentration of substrate
- b)concentration of nucleophile
- c)nature of leaving group
- d)number of lone pairs on donor atom
Correct answer is option 'A,B,C'. Can you explain this answer?
One or More than One Options Correct Type
Direction (Q. Nos. 9-14) This section contains 6 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q.
In a SN2 reaction rate of reaction depends on
a)
concentration of substrate
b)
concentration of nucleophile
c)
nature of leaving group
d)
number of lone pairs on donor atom
 | Arnab Chavan answered |
In a SN2 reaction, Rate = k[substrate] [nucleophile]
Hence, rate depends both on concentration of substrate as well as nucleophile. Rate of reaction also depends on nature of leaving group. A better leaving group gives lower activation energy hence, greater rate of reaction. Number of lone pair has no role to play in nucieophilicity hence reactivity.
Hence, rate depends both on concentration of substrate as well as nucleophile. Rate of reaction also depends on nature of leaving group. A better leaving group gives lower activation energy hence, greater rate of reaction. Number of lone pair has no role to play in nucieophilicity hence reactivity.
Which statement is true about SN2 mechanism?- a)The rate of reaction increases on increasing strength of the nucleophile
- b)The reaction is faster in polar aprotic solvents
- c)The rate of reaction increases as the leaving group ability increases
- d)Ali'of the above
Correct answer is option 'D'. Can you explain this answer?
Which statement is true about SN2 mechanism?
a)
The rate of reaction increases on increasing strength of the nucleophile
b)
The reaction is faster in polar aprotic solvents
c)
The rate of reaction increases as the leaving group ability increases
d)
Ali'of the above
 | Arnab Chavan answered |
Stronger the nucleophile, faster the SN2 reaction. Polar aprotic solvent solvate cations, makes anionic nucleophile more available for reaction, hence faster reaction. A better leaving group lowers the activation energy increasing rate of SN2 reaction.
A solution of (–)-1-chloro-1-phenylethane in toluene racemises slowly in the presence of a small amount of SbCl5, due to the formation of - a)Carbocation
- b)Free radical
- c)Carbanion
- d)Carbene
Correct answer is option 'A'. Can you explain this answer?
A solution of (–)-1-chloro-1-phenylethane in toluene racemises slowly in the presence of a small amount of SbCl5, due to the formation of
a)
Carbocation
b)
Free radical
c)
Carbanion
d)
Carbene
| | Anand Chaudhary answered |
Sorry, could you please provide the equation or problem you need a solution for?
A correct statement about transition state of SN2 reaction is - a)the transition state proceeds and follow an unstable reaction intermediate
- b)the transition state will always have net negative charge
- c)existence of this state implies an exothermic reaction
- d)the single transition state represents the point of maximum free energy of the reaction
Correct answer is option 'D'. Can you explain this answer?
A correct statement about transition state of SN2 reaction is
a)
the transition state proceeds and follow an unstable reaction intermediate
b)
the transition state will always have net negative charge
c)
existence of this state implies an exothermic reaction
d)
the single transition state represents the point of maximum free energy of the reaction
 | Anuj Iyer answered |
SN2 reaction is a one step (concerted) reaction that involes a single transition state.
Consider the reaction given below.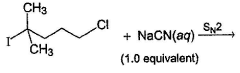 Q. The correct statement(s) applicable to the above reaction is/are
Q. The correct statement(s) applicable to the above reaction is/are- a)Cl- is substituted predominantly
- b) Cl- is a better leaving group
- c)substitution of I- in the above reaction required greater activation energy than for Cl-
- d)addition of some Nal catalyse the substitution reaction
Correct answer is option 'A,C,D'. Can you explain this answer?
Consider the reaction given below.
Q.
The correct statement(s) applicable to the above reaction is/are
a)
Cl- is substituted predominantly
b)
Cl- is a better leaving group
c)
substitution of I- in the above reaction required greater activation energy than for Cl-
d)
addition of some Nal catalyse the substitution reaction
 | Prashanth Banerjee answered |
Steric hindrance plays the most important role in SN2 reaction. Hence, although Cl is poorer leaving group than I, Cl is substituted predominantly in the above reaction due to less steric hindrance at α-carbon. Addition of Nal replace Cl by I and substitution becomes easier.
What is the correct increasing order of reactivity of the followings in SN2 reaction ?I. CH2 = CH — Br
II. CH2 = CH— I
III. CH3CH2CH2 — I
IV. CH3OCH2CH2 — I - a)I < II < III < IV
- b)III < II < I < IV
- c)II < III < IV < I
- d)II < I < III < IV
Correct answer is option 'C'. Can you explain this answer?
What is the correct increasing order of reactivity of the followings in SN2 reaction ?
I. CH2 = CH — Br
II. CH2 = CH— I
III. CH3CH2CH2 — I
IV. CH3OCH2CH2 — I
II. CH2 = CH— I
III. CH3CH2CH2 — I
IV. CH3OCH2CH2 — I
a)
I < II < III < IV
b)
III < II < I < IV
c)
II < III < IV < I
d)
II < I < III < IV
 | Tejas Singh answered |
Allyl bromide (I) is most reactive among the given halides as pi bonds from allylic position stabilises the transition state. Vinyl iodide (II) is least reactive due to partial double bond character. Electron withdrawing inductive effect of CH3O- increases reactivity of (IV) over (III)
Consider the following compound,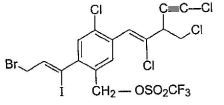 Q. If the above compound is treated with excess of NaCN(aq), how many CN- group would be incorporated by SN2 reaction?
Q. If the above compound is treated with excess of NaCN(aq), how many CN- group would be incorporated by SN2 reaction?
Correct answer is '3'. Can you explain this answer?
Consider the following compound,
Q.
If the above compound is treated with excess of NaCN(aq), how many CN- group would be incorporated by SN2 reaction?
| | Ritika Sengupta answered |
Only the circled groups are substituted in SN2,
A SN2 reaction involves back side attack of nucleophile at the α-carbon of substrate because- a)both nucleophile and leaving group are electron rich, nucleophilic attack occur from most distant position
- b)there is greater electron density on the back side of substrate
- c)there is not enough physical space in the front side from where leaving group leaves the substrate
- d)substrate possesses a hole in the backside
Correct answer is option 'A,C'. Can you explain this answer?
A SN2 reaction involves back side attack of nucleophile at the α-carbon of substrate because
a)
both nucleophile and leaving group are electron rich, nucleophilic attack occur from most distant position
b)
there is greater electron density on the back side of substrate
c)
there is not enough physical space in the front side from where leaving group leaves the substrate
d)
substrate possesses a hole in the backside
 | Anuj Iyer answered |
Both nucleophile and leaving group are electron rich species, nucleophile attacks from backside (most remote position) to have minimum repulsion with leaving group. Also, due to the presence of leaving group on front side there is less physical space for attack of nucleophile.
In SN2 reactions, the correct order of reactivity for the following compounds: CH3Cl, CH3CH2Cl, (CH3)2CHCl and (CH3)3CCl is- a)(CH3)2CHCl > CH3CH2Cl > CH3Cl > (CH3)3CCl
- b)CH3Cl > CH3CH2Cl > (CH3)2CHCl > (CH3)3CCl
- c)CH3Cl > (CH3)2CHCl > CH3CH2Cl > (CH3)3CCl
- d)CH3CH2Cl > CH3Cl > (CH3)2CHCl > (CH3)3CCl
Correct answer is option 'B'. Can you explain this answer?
In SN2 reactions, the correct order of reactivity for the following compounds: CH3Cl, CH3CH2Cl, (CH3)2CHCl and (CH3)3CCl is
a)
(CH3)2CHCl > CH3CH2Cl > CH3Cl > (CH3)3CCl
b)
CH3Cl > CH3CH2Cl > (CH3)2CHCl > (CH3)3CCl
c)
CH3Cl > (CH3)2CHCl > CH3CH2Cl > (CH3)3CCl
d)
CH3CH2Cl > CH3Cl > (CH3)2CHCl > (CH3)3CCl
| | Mahesh Chaudhary answered |
B) CH3CH2Cl
c) (CH3)2CHCl
d) (CH3)3CCl
c) (CH3)2CHCl
d) (CH3)3CCl
1-chlorobutane is more reactive than 2-chloro-2-methyl propane in a SN2 reaction because- a)α-carbon is less crowded in 1-chlorobutane
- b)α-carbon is less electropositive in 1-chlorobutane
- c)electron donating inductive effect of three methyl group is greater in 2-chloro 2-methyl propane
- d)α-carbon is more electropositive in 1-chlorobutan
Correct answer is option 'A,C,D'. Can you explain this answer?
1-chlorobutane is more reactive than 2-chloro-2-methyl propane in a SN2 reaction because
a)
α-carbon is less crowded in 1-chlorobutane
b)
α-carbon is less electropositive in 1-chlorobutane
c)
electron donating inductive effect of three methyl group is greater in 2-chloro 2-methyl propane
d)
α-carbon is more electropositive in 1-chlorobutan
 | Arnab Chavan answered |
As indicated in the given reaction, α-carbon is less hindered in 1- chloropropane. Also + I effect of three methyl groups in 2- chloro-2-methyl propane decreases electropositive character of α-carbon, decreases reactivity further in SN2 reaction.
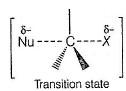
The correct statement concerning a SN2 reaction is- a)the reaction mechanism involve atleast one reactive intermediate
- b)transition state is pentavalent
- c)product is formed after passing through several transition states
- d)nucleophile attacks from front side on which leaving group is present
Correct answer is option 'B'. Can you explain this answer?
The correct statement concerning a SN2 reaction is
a)
the reaction mechanism involve atleast one reactive intermediate
b)
transition state is pentavalent
c)
product is formed after passing through several transition states
d)
nucleophile attacks from front side on which leaving group is present
 | Tejas Singh answered |
The transition state in SN2 reaction is pentavalent as indicated here.
Comprehension TypeDirection (Q. Nos. 15-17) This section contains a paragraph, describing theory, experiments, data, etc. Three Questions related to the paragraph have been given. Each question has only one correct answer among the four given options (a), (b), (c) and (d).PassageThe general mechanism of a SN2 reaction is as follows.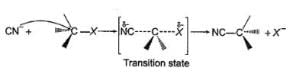 Factors that limit the rate of reaction are steric hindrance at α-cabron of substrate and strength of C—X bond. Any factor which stabilises transition state increases the rate of reaction.Q. Consider the following SN2 reaction,
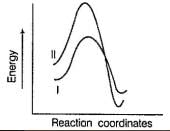
Factors that limit the rate of reaction are steric hindrance at α-cabron of substrate and strength of C—X bond. Any factor which stabilises transition state increases the rate of reaction.Q. Consider the following SN2 reaction,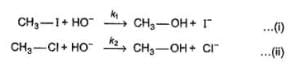 Which of the following energy diagram is correctly labelled?
Which of the following energy diagram is correctly labelled?- a)
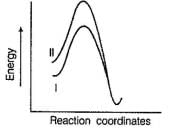
- b)
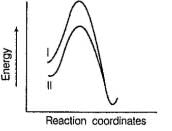
- c)
- d)
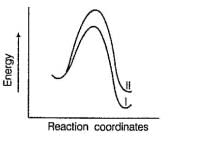
Correct answer is option 'B'. Can you explain this answer?
Comprehension Type
Direction (Q. Nos. 15-17) This section contains a paragraph, describing theory, experiments, data, etc. Three Questions related to the paragraph have been given. Each question has only one correct answer among the four given options (a), (b), (c) and (d).
Passage
The general mechanism of a SN2 reaction is as follows.

Factors that limit the rate of reaction are steric hindrance at α-cabron of substrate and strength of C—X bond. Any factor which stabilises transition state increases the rate of reaction.
Q.
Consider the following SN2 reaction,

Which of the following energy diagram is correctly labelled?
a)

b)

c)

d)

 | Tejas Singh answered |
Products are same in both reactions, hence same potential energies of products are shown. Also I- is better leaving group, has lower activation energy in SN2 reaction as indicated by curve-l in diagram.
Under identical experimental condition, how many of the following substrate react at faster rate with aqueous NaOH than 2-bromobutane as substrate?
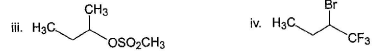



Correct answer is '6'. Can you explain this answer?
Under identical experimental condition, how many of the following substrate react at faster rate with aqueous NaOH than 2-bromobutane as substrate?
| | Chirag Joshi answered |
Compounds (II), (III), (IV), (V), (VIII) and (X) react at faster rate than 2-bromobutane in SN2 reaction.
The correct statement regarding a SN2 reaction is/are- a)reaction involving stronger bond formation to α-carbon occur at faster rate
- b)reaction shows kinetic isotopic effect
- c)substitution of
 lower the activation energy of reaction
lower the activation energy of reaction - d)presence of electron withdrawing group in substrate increases reactivity
Correct answer is option 'B,C,D'. Can you explain this answer?
The correct statement regarding a SN2 reaction is/are
a)
reaction involving stronger bond formation to α-carbon occur at faster rate
b)
reaction shows kinetic isotopic effect
c)
substitution of  lower the activation energy of reaction
lower the activation energy of reaction
d)
presence of electron withdrawing group in substrate increases reactivity
 | Anuj Iyer answered |
Stronger bond formation at α-carbon affects the stability of product (thermodynamics) not the rate (kinetics) of reaction. Change of isotope at α-position or in halogens affect the rate, hence show kinetic isotopic effect. Presence of electron withdrawing group in substrate increases electrophilicity of α-carbon hence, increases its reactivity towards nucleophiles.
Consider the following SN2 reaction,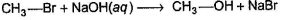 Q. Which of the following could increase the reactivity (rate) of reaction ?
Q. Which of the following could increase the reactivity (rate) of reaction ?- a)Increasing NaOH concentration
- b)Adding some Nal
- c)Replacing CHsBr by 14CH3— Br
- d) Using NaO18 H in place of NaOH
Correct answer is option 'A,B,D'. Can you explain this answer?
Consider the following SN2 reaction,
Q.
Which of the following could increase the reactivity (rate) of reaction ?
a)
Increasing NaOH concentration
b)
Adding some Nal
c)
Replacing CHsBr by 14CH3— Br
d)
Using NaO18 H in place of NaOH
 | Anuj Iyer answered |
Increasing concentration of nucleophile (NaOH) increases the reactivity. Adding some Nal also catalyse reaction because of following equilibrium.
Now reaction occur with CH3I which has better leaving group. Heavier isotope forms stronger covalent bond. Hence, replacing CH3Br by 14CH3Br stabilises substrate, decreases rate whereas replacing NaOH by NaO18H stabilises transition state increases rate of reaction.
Now reaction occur with CH3I which has better leaving group. Heavier isotope forms stronger covalent bond. Hence, replacing CH3Br by 14CH3Br stabilises substrate, decreases rate whereas replacing NaOH by NaO18H stabilises transition state increases rate of reaction.
Consider the two lines shown in the diagram given below. Q. Which of the following apply appropriately to a SN2 reaction?
Q. Which of the following apply appropriately to a SN2 reaction?- a)Graph I could represent Cl- and Graph II represents I- leaving group
- b)Graph I could represent HO- and Graph II represents CH3COO- nucleophile
- c)Graph I could represent H2O and Graph II H2S as nucleophile
- d)Graph I could represent (CH3)2NH and Graph II to (CH3)2N- nucleophiles
Correct answer is option 'B'. Can you explain this answer?
Consider the two lines shown in the diagram given below.
Q.
Which of the following apply appropriately to a SN2 reaction?
a)
Graph I could represent Cl- and Graph II represents I- leaving group
b)
Graph I could represent HO- and Graph II represents CH3COO- nucleophile
c)
Graph I could represent H2O and Graph II H2S as nucleophile
d)
Graph I could represent (CH3)2NH and Graph II to (CH3)2N- nucleophiles
 | Om Kumar answered |
HO- is stronger nucleophile than CH3COO- . Hence, lower activation energy of (I) than (II).
PassageThe general mechanism of a SN2 reaction is as follows.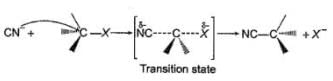 Factors that limit the rate of reaction are steric hindrance at α-cabron of substrate and strength of C—X bond. Any factor which stabilises transition state increases the rate of reaction.Q. Which of the following is not a correct representation of SN2 reaction ?
Factors that limit the rate of reaction are steric hindrance at α-cabron of substrate and strength of C—X bond. Any factor which stabilises transition state increases the rate of reaction.Q. Which of the following is not a correct representation of SN2 reaction ?- a)
- b)
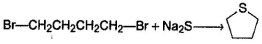
- c)CH3CH2OH + NaNH2 → CH3CH2NH2
- d)
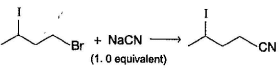
Correct answer is option 'C'. Can you explain this answer?
Passage
The general mechanism of a SN2 reaction is as follows.

Factors that limit the rate of reaction are steric hindrance at α-cabron of substrate and strength of C—X bond. Any factor which stabilises transition state increases the rate of reaction.
Q.
Which of the following is not a correct representation of SN2 reaction ?
a)
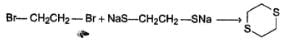
b)
c)
CH3CH2OH + NaNH2 → CH3CH2NH2
d)
 | Om Kumar answered |
Following neutralisation reaction is preferred over SN2 reaction :
What is the correct increasing order of reactivity of the following in the SN2 reaction?
- a)I < II < III < IV
- b)III < II < I < IV
- c)II < III < IV < I
- d)Ill < I < II < I V
Correct answer is option 'D'. Can you explain this answer?
What is the correct increasing order of reactivity of the following in the SN2 reaction?
a)
I < II < III < IV
b)
III < II < I < IV
c)
II < III < IV < I
d)
Ill < I < II < I V
 | Prashanth Banerjee answered |
(IV) is most reactive as it is benzylic as well as electron withdrawing effect of — NO2 further increases the reactivity. (Ill) is least reactive due to resonance effect resulting in partial double bond character between carbon and chlorine. (I) is less reactive in SN2 reaction than II, due to greater steric hindrance in (I).
PassageThe general mechanism of a SN2 reaction is as follows.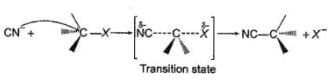 Factors that limit the rate of reaction are steric hindrance at α-cabron of substrate and strength of C—X bond. Any factor which stabilises transition state increases the rate of reaction.Q. In the previous question, under identical reaction condition, i.e. tem perature, concentration of substrate and nucleophile, the correct relationship between rate constant is
Factors that limit the rate of reaction are steric hindrance at α-cabron of substrate and strength of C—X bond. Any factor which stabilises transition state increases the rate of reaction.Q. In the previous question, under identical reaction condition, i.e. tem perature, concentration of substrate and nucleophile, the correct relationship between rate constant is- a)kI < kII
- b)kI + kII
- c)kI > kII
- d)kI = kII
Correct answer is option 'C'. Can you explain this answer?
Passage
The general mechanism of a SN2 reaction is as follows.

Factors that limit the rate of reaction are steric hindrance at α-cabron of substrate and strength of C—X bond. Any factor which stabilises transition state increases the rate of reaction.
Q.
In the previous question, under identical reaction condition, i.e. tem perature, concentration of substrate and nucleophile, the correct relationship between rate constant is
a)
kI < kII
b)
kI + kII
c)
kI > kII
d)
kI = kII
 | Prashanth Banerjee answered |
Since I- is better leaving group, kI > kII
Consider the two lines shown in the diagram given below.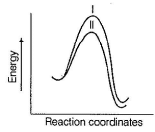 Q. In a SN2 reaction, these two lines compare the effect of the
Q. In a SN2 reaction, these two lines compare the effect of the- a)substrate
- b)nucleophile
- c)leaving group
- d)substrate or nucleophile
Correct answer is option 'D'. Can you explain this answer?
Consider the two lines shown in the diagram given below.
Q.
In a SN2 reaction, these two lines compare the effect of the
a)
substrate
b)
nucleophile
c)
leaving group
d)
substrate or nucleophile
 | Prashanth Banerjee answered |
Substrates are same as they are at same potential energies. Nucleophiles are different as magnifested by different activation energies and different products with different potential energies,
Chapter doubts & questions for Haloalkanes and Haloarenes - 30-Day Revision Course for NEET 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Haloalkanes and Haloarenes - 30-Day Revision Course for NEET in English & Hindi are available as part of NEET exam. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
;
Signup to see your scores go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup