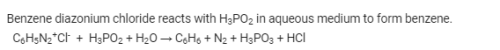All Exams > NEET > 30-Day Revision Course for NEET > All Questions
All questions of Amines for NEET Exam
When hypophosphorous acid is treated with diazonium salts, it is reduced to:- a)Arenes
- b)Methane
- c)Ethyl alcohol
- d)Amines
Correct answer is option 'A'. Can you explain this answer?
When hypophosphorous acid is treated with diazonium salts, it is reduced to:
a)
Arenes
b)
Methane
c)
Ethyl alcohol
d)
Amines
 | Mohit Rajpoot answered |
The correct answer is option A
Hypophosphorous acid when treated with diazonium salts is reduced to arenes.
ArN2Cl + H3PO2 + H2O ⟶ ArH + N2 + H3PO3 +HCl
ArN2Cl + H3PO2 + H2O ⟶ ArH + N2 + H3PO3 +HCl
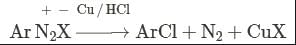
- a)Sandmeyers reaction
- b)Gattermanns reaction
- c)Dehydrogenation reaction
- d)Esterification reaction
Correct answer is option 'B'. Can you explain this answer?
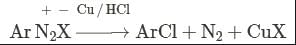
a)
Sandmeyers reaction
b)
Gattermanns reaction
c)
Dehydrogenation reaction
d)
Esterification reaction
 | Mohit Rajpoot answered |
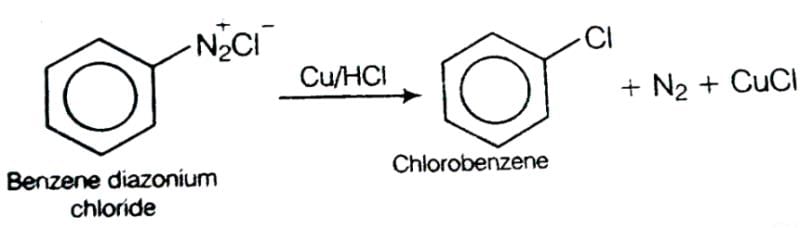 This reaction is called Gattermann reaction. In this reaction, Cl, Br and CN can be introduced into the benzene ring by simply treating diazonium salts with HCl, HBr, KCN. Respectively in presnce of copper powder instead of using Cu(I) salts.
This reaction is called Gattermann reaction. In this reaction, Cl, Br and CN can be introduced into the benzene ring by simply treating diazonium salts with HCl, HBr, KCN. Respectively in presnce of copper powder instead of using Cu(I) salts.Which of the following amine gives diazonium salt on reaction with HNO2?- a)(CH3)2NH
- b)CH3NH2
- c)C6H5NH2
- d)(CH3)3N
Correct answer is option 'C'. Can you explain this answer?
Which of the following amine gives diazonium salt on reaction with HNO2?
a)
(CH3)2NH
b)
CH3NH2
c)
C6H5NH2
d)
(CH3)3N
 | Mohit Rajpoot answered |
C6H5NH2 reacts with HNO2 to forms diazonium salts, the reaction are as follows,
C6H5NH2 + HNO2 ------> C6H5OH + H2O + N2
C6H5NH2 + HNO2 ------> C6H5OH + H2O + N2
Which of the following amine will form stable diazonium salt at 273-283 K ?- a)C6H5NH2
- b)C6H5N(CH3)2
- c)C2H5NH2
- d)C6H5CH2NH2
Correct answer is option 'A'. Can you explain this answer?
Which of the following amine will form stable diazonium salt at 273-283 K ?
a)
C6H5NH2
b)
C6H5N(CH3)2
c)
C2H5NH2
d)
C6H5CH2NH2
 | Sushil Kumar answered |
The correct answer is Option A.
Aromatic Primary amine will form the most stable diazonium salt because it releases water when it reacts with nitronium ions. If aliphatic primary amine reacts with nitrosonium ion it Also releases water but in this case water reacts with alkyl diazonium salt and it forms alcohol while benzene diazonium salt does not react with water at this temperature.
Aromatic Primary amine will form the most stable diazonium salt because it releases water when it reacts with nitronium ions. If aliphatic primary amine reacts with nitrosonium ion it Also releases water but in this case water reacts with alkyl diazonium salt and it forms alcohol while benzene diazonium salt does not react with water at this temperature.
The following reaction takes place in the presence of 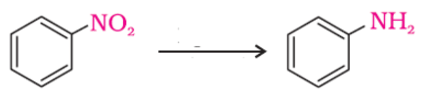
- a)NaOH/Pd
- b)H2/Pd
- c)HCl/Pd
- d)None of these
Correct answer is option 'B'. Can you explain this answer?
The following reaction takes place in the presence of
a)
NaOH/Pd
b)
H2/Pd
c)
HCl/Pd
d)
None of these
| | Anaya Patel answered |
−NO2 group is reduced to –NH2 using H2/Pd.
The stability of benzene diazonium salts is because of- a)Inductive effect
- b)Mesomeric effect
- c)Hyperconjugation
- d)Resonance
Correct answer is option 'D'. Can you explain this answer?
The stability of benzene diazonium salts is because of
a)
Inductive effect
b)
Mesomeric effect
c)
Hyperconjugation
d)
Resonance
| | Geetika Shah answered |
None of the errors that are pointed are correct.
Their stability is because of resonance. D is the correct answer.
The molecular formula of ethyl acetate is- a)C4H8O
- b)C4H8O2
- c)C5H10O2
- d)C5H8O2
Correct answer is option 'B'. Can you explain this answer?
The molecular formula of ethyl acetate is
a)
C4H8O
b)
C4H8O2
c)
C5H10O2
d)
C5H8O2
 | Tejas Singh answered |
Its molecular formula is C4H8O2
. When diazonium salt solution is treated with KI, it forms:- a)Bromobenzene
- b)Iodobenzene
- c)Phenol
- d)Acid
Correct answer is option 'B'. Can you explain this answer?
. When diazonium salt solution is treated with KI, it forms:
a)
Bromobenzene
b)
Iodobenzene
c)
Phenol
d)
Acid
 | Kshitij Pandey answered |
This reaction is unique this is only reaction in which idobenzene formed by diaazonium salt , to make idobenzene first we make diaazonium salt by aniline then we treated diaazonium salt with ki
C6H5N2Cl(diaazonium salt)+KI=C6H5I
C6H5N2Cl(diaazonium salt)+KI=C6H5I
Replacement of diazo group by other groups is helpful in preparing those substituted aromatic compounds which cannot be prepared by- a)Direct elimination
- b)Addition reaction
- c)Direct substitution
- d)Replacement reaction
Correct answer is option 'C'. Can you explain this answer?
Replacement of diazo group by other groups is helpful in preparing those substituted aromatic compounds which cannot be prepared by
a)
Direct elimination
b)
Addition reaction
c)
Direct substitution
d)
Replacement reaction
| | Vikas Kumar answered |
Replacement of diazo group by other groups is helpful in preparing those substituted aromatic compounds which cannot be prepared by direct substitution. ... The cyano group usually cannot be introduced by nucleophilic substitution of haloarenes, but such compounds can be easily prepared from diazonium salts.
The main product formed by treating an alkyl or benzyl halide with excess ammonia- a)Mixed
- b)Tertiary
- c)Secondary
- d)Primary
Correct answer is option 'D'. Can you explain this answer?
The main product formed by treating an alkyl or benzyl halide with excess ammonia
a)
Mixed
b)
Tertiary
c)
Secondary
d)
Primary
| | Amita Das answered |
The N in ammonia functions as the nucleophile and attacks the electrophilic C of the alkyl halide displacing the bromide and creating the new C-N bond.
Step 2: An acid/base reaction. The base (excess ammonia) deprotonates the positive N (ammonium) center creating the alkylation product, the primary amine.
When diazonium salt solution is treated with water at a temperature of 283 K it forms?- a)Ester
- b)Phenol
- c)Amines
- d)Alcohol
Correct answer is option 'B'. Can you explain this answer?
When diazonium salt solution is treated with water at a temperature of 283 K it forms?
a)
Ester
b)
Phenol
c)
Amines
d)
Alcohol
 | Vatturi Anjani answered |
Yes as when the temperature of the diazonium salt is allowed to rise upto 283K then the salt is reduced to phenol releasing N2 and hydrochloric acid as the by-products.
Benzene diazonium chloride on reaction with phenol in weakly basic medium gives:- a)p-Hydroxyazobenzene
- b)Benzene
- c)Diphenyl ether
- d)Chlorobenzene
Correct answer is option 'A'. Can you explain this answer?
Benzene diazonium chloride on reaction with phenol in weakly basic medium gives:
a)
p-Hydroxyazobenzene
b)
Benzene
c)
Diphenyl ether
d)
Chlorobenzene
| | Arka Das answered |
**Benzene diazonium chloride**
Benzene diazonium chloride (C6H5N2Cl) is an organic compound that is commonly used in diazotization reactions. It is formed by the reaction of aniline (C6H5NH2) with nitrous acid (HNO2) in the presence of hydrochloric acid (HCl).
**Reaction with Phenol**
When benzene diazonium chloride reacts with phenol (C6H5OH) in a weakly basic medium, it undergoes a substitution reaction known as the Sandmeyer reaction. This reaction involves the replacement of the diazonium group (-N2Cl) with the phenolic group (-OH) to form a new compound.
**Formation of p-Hydroxyazobenzene**
The reaction between benzene diazonium chloride and phenol results in the formation of p-hydroxyazobenzene (C12H10N2O), which is the correct answer (option A).
The reaction proceeds as follows:
1. The weakly basic medium provides the necessary conditions for the reaction to occur. It helps in the deprotonation of phenol to form the phenoxide ion (C6H5O-).
2. The diazonium group of benzene diazonium chloride is highly reactive and undergoes nucleophilic substitution. The nitrogen atom of the diazonium group attacks the phenoxide ion, leading to the formation of a new carbon-nitrogen bond.
3. The chlorine atom attached to the nitrogen atom is replaced by the phenoxide group, resulting in the formation of p-hydroxyazobenzene.
The reaction can be represented by the following equation:
C6H5N2Cl + C6H5O- → C12H10N2O + Cl-
**Explanation of Other Options**
Option B: Benzenedoes not participate in the reaction. It remains unchanged.
Option C: Diphenyl ether is not formed in this reaction. It involves the formation of a carbon-oxygen bond between two phenyl groups, which is not observed in the given reaction.
Option D: Chlorobenzene is not formed in this reaction. The chlorine atom from the diazonium chloride is replaced by the phenoxide group, not by another chlorine atom.
Thus, the correct answer is option A: p-Hydroxyazobenzene.
Benzene diazonium chloride (C6H5N2Cl) is an organic compound that is commonly used in diazotization reactions. It is formed by the reaction of aniline (C6H5NH2) with nitrous acid (HNO2) in the presence of hydrochloric acid (HCl).
**Reaction with Phenol**
When benzene diazonium chloride reacts with phenol (C6H5OH) in a weakly basic medium, it undergoes a substitution reaction known as the Sandmeyer reaction. This reaction involves the replacement of the diazonium group (-N2Cl) with the phenolic group (-OH) to form a new compound.
**Formation of p-Hydroxyazobenzene**
The reaction between benzene diazonium chloride and phenol results in the formation of p-hydroxyazobenzene (C12H10N2O), which is the correct answer (option A).
The reaction proceeds as follows:
1. The weakly basic medium provides the necessary conditions for the reaction to occur. It helps in the deprotonation of phenol to form the phenoxide ion (C6H5O-).
2. The diazonium group of benzene diazonium chloride is highly reactive and undergoes nucleophilic substitution. The nitrogen atom of the diazonium group attacks the phenoxide ion, leading to the formation of a new carbon-nitrogen bond.
3. The chlorine atom attached to the nitrogen atom is replaced by the phenoxide group, resulting in the formation of p-hydroxyazobenzene.
The reaction can be represented by the following equation:
C6H5N2Cl + C6H5O- → C12H10N2O + Cl-
**Explanation of Other Options**
Option B: Benzenedoes not participate in the reaction. It remains unchanged.
Option C: Diphenyl ether is not formed in this reaction. It involves the formation of a carbon-oxygen bond between two phenyl groups, which is not observed in the given reaction.
Option D: Chlorobenzene is not formed in this reaction. The chlorine atom from the diazonium chloride is replaced by the phenoxide group, not by another chlorine atom.
Thus, the correct answer is option A: p-Hydroxyazobenzene.
By treating diazonium salts with cuprous cyanide or KCN and copper powder it forms:- a)Citric acid
- b)Benzoic acid
- c)Aryl nitrile
- d)Oxalic acid
Correct answer is option 'C'. Can you explain this answer?
By treating diazonium salts with cuprous cyanide or KCN and copper powder it forms:
a)
Citric acid
b)
Benzoic acid
c)
Aryl nitrile
d)
Oxalic acid
| | Rishika Patel answered |
Formation of Aryl Nitrile from Diazonium Salts
Diazonium salts are compounds containing a positively charged nitrogen atom that is linked to an aromatic ring. These salts are often used in organic synthesis as a source of the aryl group. When treated with cuprous cyanide or KCN and copper powder, diazonium salts undergo a reaction known as Sandmeyer reaction, which forms aryl nitriles.
Reaction Mechanism
The reaction mechanism involves several steps:
1. Formation of Copper(I) Salt
First, cuprous cyanide or KCN is added to the diazonium salt solution to form a copper(I) salt. The copper(I) salt plays a crucial role in the reaction by catalyzing the formation of the aryl nitrile.
2. Formation of Aryl Copper(I) Intermediate
Next, copper powder is added to the reaction mixture, which reduces the copper(I) salt to copper metal. The copper metal then reacts with the diazonium salt to form an aryl copper(I) intermediate.
3. Formation of Aryl Cyanide
The aryl copper(I) intermediate then reacts with the cyanide ion from the cuprous cyanide or KCN to form the aryl cyanide. The reaction releases copper metal, which can then react with more diazonium salt to form more aryl copper(I) intermediate.
Overall Reaction
The overall reaction can be represented as follows:
ArN2+X- + CuCN/KCN → [CuX] + N2 + ArCu + HX
ArCu + CN- → ArCN + Cu
Where Ar represents the aryl group, X represents the anion of the diazonium salt, and HX represents the acid formed in the reaction.
Conclusion
In conclusion, the treatment of diazonium salts with cuprous cyanide or KCN and copper powder forms aryl nitriles through the Sandmeyer reaction. This reaction is an important method for the synthesis of aryl nitriles, which have a variety of applications in organic synthesis and industry.
Diazonium salts are compounds containing a positively charged nitrogen atom that is linked to an aromatic ring. These salts are often used in organic synthesis as a source of the aryl group. When treated with cuprous cyanide or KCN and copper powder, diazonium salts undergo a reaction known as Sandmeyer reaction, which forms aryl nitriles.
Reaction Mechanism
The reaction mechanism involves several steps:
1. Formation of Copper(I) Salt
First, cuprous cyanide or KCN is added to the diazonium salt solution to form a copper(I) salt. The copper(I) salt plays a crucial role in the reaction by catalyzing the formation of the aryl nitrile.
2. Formation of Aryl Copper(I) Intermediate
Next, copper powder is added to the reaction mixture, which reduces the copper(I) salt to copper metal. The copper metal then reacts with the diazonium salt to form an aryl copper(I) intermediate.
3. Formation of Aryl Cyanide
The aryl copper(I) intermediate then reacts with the cyanide ion from the cuprous cyanide or KCN to form the aryl cyanide. The reaction releases copper metal, which can then react with more diazonium salt to form more aryl copper(I) intermediate.
Overall Reaction
The overall reaction can be represented as follows:
ArN2+X- + CuCN/KCN → [CuX] + N2 + ArCu + HX
ArCu + CN- → ArCN + Cu
Where Ar represents the aryl group, X represents the anion of the diazonium salt, and HX represents the acid formed in the reaction.
Conclusion
In conclusion, the treatment of diazonium salts with cuprous cyanide or KCN and copper powder forms aryl nitriles through the Sandmeyer reaction. This reaction is an important method for the synthesis of aryl nitriles, which have a variety of applications in organic synthesis and industry.
The Hofmann elimination proceeds via a(n) __________ pathway.- a)SN2
- b)E2
- c)E1
- d)SN1
Correct answer is option 'B'. Can you explain this answer?
The Hofmann elimination proceeds via a(n) __________ pathway.
a)
SN2
b)
E2
c)
E1
d)
SN1
| | Rajat Kapoor answered |
The Hofmann Elimination is an elimination reaction that forms C-C double (pi) bonds that specifically occurs when the leaving group is NR3 [note] It proceeds through an E2 mechanism. Although the key concepts are no different than one learns in the chapter on elimination from way back in Org 1, it is often included in Org 2 as part of the grab-bag chapter on amines. Because, well… nitrogen.
p-amino azo benzene is obtained by treating diazoniumchloride with:- a)Phenol
- b)Aniline
- c)Alcohol
- d)Benzoic acid
Correct answer is option 'B'. Can you explain this answer?
p-amino azo benzene is obtained by treating diazoniumchloride with:
a)
Phenol
b)
Aniline
c)
Alcohol
d)
Benzoic acid
| | Lavanya Menon answered |
p-amino azo benzene is obtained by treating diazonium chloride with aniline. The reactions are specifically acid catalyzed and involve pre‐equilibrium formation of amine and diazonium salt followed by rate‐limiting attack of the diazonium ion at a C‐atom (C‐coupling) to give the corresponding amino azo compounds.
Hinsberg’s reagent reacts with primary and secondary amines to form sulphonamides. This reagent is also known as- a)N Methylbenzamide
- b)p – toluenesulphonyl chloride
- c)None of these
- d)Benzenesulphonyl chloride
Correct answer is option 'D'. Can you explain this answer?
Hinsberg’s reagent reacts with primary and secondary amines to form sulphonamides. This reagent is also known as
a)
N Methylbenzamide
b)
p – toluenesulphonyl chloride
c)
None of these
d)
Benzenesulphonyl chloride
 | Mohit Patel answered |
C6H5SO2Cl this is benzenesulphonyl chloride or hinsberg reagent.
Diazonium salts are used in the preparation of- a)Hormones
- b)Vitamins
- c)Dyes
- d)Proteins
Correct answer is option 'C'. Can you explain this answer?
Diazonium salts are used in the preparation of
a)
Hormones
b)
Vitamins
c)
Dyes
d)
Proteins
| | Vijay Bansal answered |
A diazonium salt is an organic compound that contains a nitrogen-nitrogen triple bond and some other generic side group that could be either alkyl (an alkane derivative) or aryl (benzene ring). The 'salt' portion of the name comes from the fact that the diazo (meaning 'di-nitrogen') portion of the compound is present as its ionic salt, with a chloride ion being a typical counter-ion for the positively charged nitrogen atom.
Which of the following amines is most soluble in water?- a)(CH3CH2CH2)2NH
- b)ethylamine
- c)pyrrolidine
- d)(CH3CH3)3N
Correct answer is option 'B'. Can you explain this answer?
Which of the following amines is most soluble in water?
a)
(CH3CH2CH2)2NH
b)
ethylamine
c)
pyrrolidine
d)
(CH3CH3)3N
 | Sankar Gupta answered |
Because to maximum hydrogen bonding.
An azo compound is formed when benzene diazonium chloride reacts with:- a)2,4,6-Trimethylphenol
- b)Both (A) and (B)
- c)Phenol
- d)Aniline
Correct answer is option 'B'. Can you explain this answer?
An azo compound is formed when benzene diazonium chloride reacts with:
a)
2,4,6-Trimethylphenol
b)
Both (A) and (B)
c)
Phenol
d)
Aniline
| | Manoj Chauhan answered |
Formation of Azo Compound
An azo compound is formed when benzene diazonium chloride reacts with 2,4,6-trimethylphenol and phenol. The reaction involves the substitution of diazonium group (-N2+) with the hydroxyl group (-OH) of the phenol. The reaction is called diazotization.
Diazotization Reaction
Diazotization is a chemical reaction that involves the conversion of an aromatic primary amine to a diazonium salt by the action of nitrous acid. It is a two-step process that involves the formation of a diazonium intermediate, which then reacts with a nucleophile to form the final product.
Step 1: Diazotization of Aromatic Primary Amine
In the first step of diazotization, an aromatic primary amine is treated with nitrous acid (HNO2) to form a diazonium salt. The reaction can be represented as:
R-NH2 + HNO2 → R-N2+Cl- + H2O
where R represents the aromatic group.
Step 2: Reaction with Nucleophile
In the second step, the diazonium salt reacts with a nucleophile (such as phenol or 2,4,6-trimethylphenol) to form the final product, which is an azo compound. The reaction can be represented as:
R-N2+Cl- + Ar-OH → R-N=N-Ar + HCl + H2O
where Ar represents the aromatic ring.
Formation of Azo Compound from Benzene Diazonium Chloride
When benzene diazonium chloride reacts with 2,4,6-trimethylphenol or phenol, an azo compound is formed. The reaction can be represented as:
C6H5-N2+Cl- + Ar-OH → C6H5-N=N-Ar + HCl + H2O
where Ar represents the aromatic ring of 2,4,6-trimethylphenol or phenol.
Therefore, option B (Both A and B) is the correct answer as both 2,4,6-trimethylphenol and phenol can react with benzene diazonium chloride to form azo compounds.
An azo compound is formed when benzene diazonium chloride reacts with 2,4,6-trimethylphenol and phenol. The reaction involves the substitution of diazonium group (-N2+) with the hydroxyl group (-OH) of the phenol. The reaction is called diazotization.
Diazotization Reaction
Diazotization is a chemical reaction that involves the conversion of an aromatic primary amine to a diazonium salt by the action of nitrous acid. It is a two-step process that involves the formation of a diazonium intermediate, which then reacts with a nucleophile to form the final product.
Step 1: Diazotization of Aromatic Primary Amine
In the first step of diazotization, an aromatic primary amine is treated with nitrous acid (HNO2) to form a diazonium salt. The reaction can be represented as:
R-NH2 + HNO2 → R-N2+Cl- + H2O
where R represents the aromatic group.
Step 2: Reaction with Nucleophile
In the second step, the diazonium salt reacts with a nucleophile (such as phenol or 2,4,6-trimethylphenol) to form the final product, which is an azo compound. The reaction can be represented as:
R-N2+Cl- + Ar-OH → R-N=N-Ar + HCl + H2O
where Ar represents the aromatic ring.
Formation of Azo Compound from Benzene Diazonium Chloride
When benzene diazonium chloride reacts with 2,4,6-trimethylphenol or phenol, an azo compound is formed. The reaction can be represented as:
C6H5-N2+Cl- + Ar-OH → C6H5-N=N-Ar + HCl + H2O
where Ar represents the aromatic ring of 2,4,6-trimethylphenol or phenol.
Therefore, option B (Both A and B) is the correct answer as both 2,4,6-trimethylphenol and phenol can react with benzene diazonium chloride to form azo compounds.
For producing amines, the reaction of nitro compounds with iron scrap is preferred because- a)HCl is very cheap
- b)HCl is formed on hydrolysis of FeCl2
- c)Not too much HCl is formed in the reaction
- d)Nitro compounds are easily available
Correct answer is option 'B'. Can you explain this answer?
For producing amines, the reaction of nitro compounds with iron scrap is preferred because
a)
HCl is very cheap
b)
HCl is formed on hydrolysis of FeCl2
c)
Not too much HCl is formed in the reaction
d)
Nitro compounds are easily available
 | Kavya Das answered |
This reaction is preferred because on reduction of nitro compouns to amine , Fe will get oxidize to Fe(II) a which on hydrolysis will produce HCl which is required in the reaction
Aniline does not undergo Friedel – Crafts reaction- a)Anilium ion deactivates any further reaction.
- b)aluminium chloride, the catalyst reacts with NH2 group
- c)nitrogen of aniline acquires positive charge
- d)all of these
Correct answer is option 'D'. Can you explain this answer?
Aniline does not undergo Friedel – Crafts reaction
a)
Anilium ion deactivates any further reaction.
b)
aluminium chloride, the catalyst reacts with NH2 group
c)
nitrogen of aniline acquires positive charge
d)
all of these
 | Gauri Sharma answered |
NH2 has lp of electron which reacts with AlCl3 or other catalyst that we add to form anilium ion.
Azo-dye test is given by:- a)All amines
- b)Only primary amines
- c)Only primary aliphatic amines
- d)Only primary aromatic amines
Correct answer is option 'D'. Can you explain this answer?
Azo-dye test is given by:
a)
All amines
b)
Only primary amines
c)
Only primary aliphatic amines
d)
Only primary aromatic amines
| | Naina Bansal answered |
Azo-Dye Test. This test is given by aromatic primary amines. Aromatic primary amines react with nitrous acid to form diazonium salts. These diazonium salts undergo coupling reaction with β-naphthol to form orange coloured azo dye.
Aniline does not undergo Friedel – Crafts reaction- a)Anilium ion deactivates any further reaction.
- b)aluminium chloride, the catalyst reacts with NH2 group
- c)nitrogen of aniline acquires positive charge
- d)all of these
Correct answer is option 'D'. Can you explain this answer?
Aniline does not undergo Friedel – Crafts reaction
a)
Anilium ion deactivates any further reaction.
b)
aluminium chloride, the catalyst reacts with NH2 group
c)
nitrogen of aniline acquires positive charge
d)
all of these
 | Aryan Dasgupta answered |
NH2 has lp of electron which reacts with AlCl3 or other catalyst that we add to form anilium ion.
The conversion of primary aromatic amines into diazonium salts is known as:- a)Diazotisation
- b)Alkylation
- c)Acylation
- d)Displacement
Correct answer is option 'A'. Can you explain this answer?
The conversion of primary aromatic amines into diazonium salts is known as:
a)
Diazotisation
b)
Alkylation
c)
Acylation
d)
Displacement
| | Gayathri Kumaran answered |
This rxtn helps in the production of diazonium salts. hence it is called diazotisation
Direct nitration of aniline yields significant amount of meta derivative. To obtain more p – nitro derivative, one or more of the below can be done- a)reacting with acetic anhydride
- b)acetylation reaction
- c)controlling the nitration reaction
- d)all of these
Correct answer is option 'D'. Can you explain this answer?
Direct nitration of aniline yields significant amount of meta derivative. To obtain more p – nitro derivative, one or more of the below can be done
a)
reacting with acetic anhydride
b)
acetylation reaction
c)
controlling the nitration reaction
d)
all of these
 | Janhavi Kaur answered |
Meta isomer form because of converstion of aniline to anilium ion which is META director. This can be prevented by acetylation of anilie by reaction with acetyl chloride or acetylanhydride.
What happens when benzene diazonium chloride is treated with potassium cyanide in presence of Cu powder?- a)Benzophenone
- b)Methyl isocyanide
- c)Acetonitrile
- d)Benzonitrile
Correct answer is option 'D'. Can you explain this answer?
What happens when benzene diazonium chloride is treated with potassium cyanide in presence of Cu powder?
a)
Benzophenone
b)
Methyl isocyanide
c)
Acetonitrile
d)
Benzonitrile
 | Pallavi Desai answered |
Reaction Overview
When benzene diazonium chloride (C6H5N2Cl) is treated with potassium cyanide (KCN) in the presence of copper powder (Cu), a significant reaction occurs that leads to the formation of benzonitrile (C6H5CN). This reaction is a classic example of nucleophilic substitution.
Mechanism of Reaction
- Formation of Benzene Diazonium Ion: Benzene diazonium chloride is generated from an aromatic amine and nitrous acid. This ion is highly reactive and can undergo various transformations.
- Nucleophilic Attack: In the presence of KCN, the cyanide ion (CN-) acts as a nucleophile. It attacks the positively charged nitrogen in the diazonium ion, resulting in the displacement of the nitrogen gas (N2), which is a stable and inert product.
- Formation of Benzonitrile: The final product of this reaction is benzonitrile, where the cyanide group replaces the diazonium moiety on the benzene ring. The overall reaction can be summarized as:
Benzene Diazonium Ion + CN- → Benzonitrile + N2
Significance of Copper Powder
- Copper's Role: Copper powder acts as a catalyst in this reaction, facilitating the transformation of the diazonium ion and ensuring the reaction proceeds smoothly.
Conclusion
The treatment of benzene diazonium chloride with potassium cyanide in the presence of copper powder ultimately produces benzonitrile, making option 'D' the correct answer. This reaction showcases the utility of diazonium salts in organic synthesis, particularly in forming nitriles from aromatic compounds.
When benzene diazonium chloride (C6H5N2Cl) is treated with potassium cyanide (KCN) in the presence of copper powder (Cu), a significant reaction occurs that leads to the formation of benzonitrile (C6H5CN). This reaction is a classic example of nucleophilic substitution.
Mechanism of Reaction
- Formation of Benzene Diazonium Ion: Benzene diazonium chloride is generated from an aromatic amine and nitrous acid. This ion is highly reactive and can undergo various transformations.
- Nucleophilic Attack: In the presence of KCN, the cyanide ion (CN-) acts as a nucleophile. It attacks the positively charged nitrogen in the diazonium ion, resulting in the displacement of the nitrogen gas (N2), which is a stable and inert product.
- Formation of Benzonitrile: The final product of this reaction is benzonitrile, where the cyanide group replaces the diazonium moiety on the benzene ring. The overall reaction can be summarized as:
Benzene Diazonium Ion + CN- → Benzonitrile + N2
Significance of Copper Powder
- Copper's Role: Copper powder acts as a catalyst in this reaction, facilitating the transformation of the diazonium ion and ensuring the reaction proceeds smoothly.
Conclusion
The treatment of benzene diazonium chloride with potassium cyanide in the presence of copper powder ultimately produces benzonitrile, making option 'D' the correct answer. This reaction showcases the utility of diazonium salts in organic synthesis, particularly in forming nitriles from aromatic compounds.
Arrange the following in order of increasing basicity: aniline, p – nitroaniline, p – toluidine,and p – methoxyaniline- a)p – methoxyaniline p – nitroaniline < aniline < p – toluidine
- b)p – nitroaniline < aniline < p – toluidine < p – methoxyaniline
- c)aniline < p – methoxyaniline p – nitroaniline < p – toluidine
- d)p – nitroaniline < aniline< p – methoxyaniline < p – toluidine
Correct answer is option 'B'. Can you explain this answer?
Arrange the following in order of increasing basicity: aniline, p – nitroaniline, p – toluidine,and p – methoxyaniline
a)
p – methoxyaniline p – nitroaniline < aniline < p – toluidine
b)
p – nitroaniline < aniline < p – toluidine < p – methoxyaniline
c)
aniline < p – methoxyaniline p – nitroaniline < p – toluidine
d)
p – nitroaniline < aniline< p – methoxyaniline < p – toluidine
 | Nilesh Goyal answered |
-OMe group at p position will increase the basicity more tha normal –CH3 group at p position. While presence of –NO2 at position will decrease the basicity.
Chapter doubts & questions for Amines - 30-Day Revision Course for NEET 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Amines - 30-Day Revision Course for NEET in English & Hindi are available as part of NEET exam. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
;
Signup on EduRev and stay on top of your study goals
10M+ students crushing their study goals daily