All Exams > NEET > 30-Day Revision Course for NEET > All Questions
All questions of Purification and Characterisation of Organic Compounds for NEET Exam
The best method for the separation of naphthalene and benzoic acid from their mixture is- a)chromatography
- b)crystallisation
- c)distillation
- d)sublimation
Correct answer is option 'D'. Can you explain this answer?
The best method for the separation of naphthalene and benzoic acid from their mixture is
a)
chromatography
b)
crystallisation
c)
distillation
d)
sublimation
| | Nishtha Shah answered |
Understanding the Separation Methods
When separating naphthalene and benzoic acid from their mixture, it's essential to consider the physical properties of both compounds. Naphthalene is a non-polar aromatic hydrocarbon, while benzoic acid is a polar compound with acidic properties.
Why Sublimation is the Best Method
Sublimation is a process where a solid turns directly into vapor without passing through the liquid phase. This property is particularly useful for naphthalene, which sublimes at room temperature:
- Naphthalene: Sublimes easily at around 80°C, allowing it to transition to gas without heating to its melting point.
- Benzoic Acid: Has a higher melting point (about 122°C) and does not sublimate under normal conditions.
Separation Process
1. Heating the Mixture: When the mixture is heated, naphthalene will sublime first.
2. Collecting Naphthalene Vapor: The vapor can be collected and cooled, allowing it to solidify separately from benzoic acid.
3. Remaining Benzoic Acid: After naphthalene is removed, benzoic acid can be purified further through recrystallization if necessary.
Advantages of Sublimation
- Efficiency: This method provides a quick and effective way to separate the compounds, as it capitalizes on their differing physical properties.
- Minimal Reagents: Sublimation requires no additional solvents or reagents, making it a cleaner method.
- High Purity: The sublimation process yields high-purity naphthalene, avoiding contamination from solvents.
In conclusion, sublimation is the most suitable method for separating naphthalene and benzoic acid due to the significant differences in their sublimation properties, ensuring efficient and effective separation.
When separating naphthalene and benzoic acid from their mixture, it's essential to consider the physical properties of both compounds. Naphthalene is a non-polar aromatic hydrocarbon, while benzoic acid is a polar compound with acidic properties.
Why Sublimation is the Best Method
Sublimation is a process where a solid turns directly into vapor without passing through the liquid phase. This property is particularly useful for naphthalene, which sublimes at room temperature:
- Naphthalene: Sublimes easily at around 80°C, allowing it to transition to gas without heating to its melting point.
- Benzoic Acid: Has a higher melting point (about 122°C) and does not sublimate under normal conditions.
Separation Process
1. Heating the Mixture: When the mixture is heated, naphthalene will sublime first.
2. Collecting Naphthalene Vapor: The vapor can be collected and cooled, allowing it to solidify separately from benzoic acid.
3. Remaining Benzoic Acid: After naphthalene is removed, benzoic acid can be purified further through recrystallization if necessary.
Advantages of Sublimation
- Efficiency: This method provides a quick and effective way to separate the compounds, as it capitalizes on their differing physical properties.
- Minimal Reagents: Sublimation requires no additional solvents or reagents, making it a cleaner method.
- High Purity: The sublimation process yields high-purity naphthalene, avoiding contamination from solvents.
In conclusion, sublimation is the most suitable method for separating naphthalene and benzoic acid due to the significant differences in their sublimation properties, ensuring efficient and effective separation.
A is a lighter phenol and B is an aromatic carboxylic acid. Separation of a mixture of A and B can be carried out easily by using a solution of- a)sodium hydroxide
- b)sodium sulphate
- c)calcium chloride
- d)sodium bicarbonate
Correct answer is option 'D'. Can you explain this answer?
A is a lighter phenol and B is an aromatic carboxylic acid. Separation of a mixture of A and B can be carried out easily by using a solution of
a)
sodium hydroxide
b)
sodium sulphate
c)
calcium chloride
d)
sodium bicarbonate
 | Aryan Dasgupta answered |
Separation of Phenols and Carboxylic Acids
Separating a mixture of a lighter phenol (A) and an aromatic carboxylic acid (B) can effectively be achieved using a sodium bicarbonate solution. Here's a detailed explanation of why this method works.
Properties of the Compounds
- Phenol (A):
- A weak acid but does not react significantly with sodium bicarbonate.
- Remains soluble in organic solvents and retains its original form in aqueous solutions.
- Aromatic Carboxylic Acid (B):
- A stronger acid compared to phenol.
- Reacts with sodium bicarbonate to form a water-soluble sodium salt and carbon dioxide gas.
Separation Process
1. Dissolution in Sodium Bicarbonate:
- When the mixture is treated with sodium bicarbonate solution, the aromatic carboxylic acid (B) reacts to form a sodium salt of the acid.
- This reaction generates carbon dioxide, which can be observed as effervescence.
2. Formation of Layers:
- The sodium salt of the carboxylic acid is soluble in water, while the phenol remains in the organic phase.
- This allows for the separation of the two compounds into different layers.
3. Extraction:
- The aqueous layer containing the sodium salt can be separated from the organic layer containing phenol.
- The salt can then be converted back to the carboxylic acid by acidifying the aqueous layer.
Conclusion
Using sodium bicarbonate is a selective method for separating lighter phenols from aromatic carboxylic acids. The ability of sodium bicarbonate to convert the carboxylic acid into a soluble salt while leaving the phenol untouched is the key to this separation process.
Separating a mixture of a lighter phenol (A) and an aromatic carboxylic acid (B) can effectively be achieved using a sodium bicarbonate solution. Here's a detailed explanation of why this method works.
Properties of the Compounds
- Phenol (A):
- A weak acid but does not react significantly with sodium bicarbonate.
- Remains soluble in organic solvents and retains its original form in aqueous solutions.
- Aromatic Carboxylic Acid (B):
- A stronger acid compared to phenol.
- Reacts with sodium bicarbonate to form a water-soluble sodium salt and carbon dioxide gas.
Separation Process
1. Dissolution in Sodium Bicarbonate:
- When the mixture is treated with sodium bicarbonate solution, the aromatic carboxylic acid (B) reacts to form a sodium salt of the acid.
- This reaction generates carbon dioxide, which can be observed as effervescence.
2. Formation of Layers:
- The sodium salt of the carboxylic acid is soluble in water, while the phenol remains in the organic phase.
- This allows for the separation of the two compounds into different layers.
3. Extraction:
- The aqueous layer containing the sodium salt can be separated from the organic layer containing phenol.
- The salt can then be converted back to the carboxylic acid by acidifying the aqueous layer.
Conclusion
Using sodium bicarbonate is a selective method for separating lighter phenols from aromatic carboxylic acids. The ability of sodium bicarbonate to convert the carboxylic acid into a soluble salt while leaving the phenol untouched is the key to this separation process.
Camphor is often used in molecular mass determination because- a)it is readily available
- b)it has a very high cryoscopic constant
- c)it is volatile
- d)it is solvent for organic substances
Correct answer is option 'C'. Can you explain this answer?
Camphor is often used in molecular mass determination because
a)
it is readily available
b)
it has a very high cryoscopic constant
c)
it is volatile
d)
it is solvent for organic substances
 | Top Rankers answered |
Camphor is used in molecular mass determination due to its volatile nature.
The method is called Rast’s camphor method. Camphor acts as a solid solvent which is volatile, hence can be removed easily.
The method is called Rast’s camphor method. Camphor acts as a solid solvent which is volatile, hence can be removed easily.
In steam distillation of toluene, the pressure of toluene in vapour is- a)equal to the pressure of barometer
- b)less than the pressure of barometer
- c)equal to vapour pressure of toluene in simple distillation
- d)more than vapour pressure of toluene in simple distillation
Correct answer is option 'B'. Can you explain this answer?
In steam distillation of toluene, the pressure of toluene in vapour is
a)
equal to the pressure of barometer
b)
less than the pressure of barometer
c)
equal to vapour pressure of toluene in simple distillation
d)
more than vapour pressure of toluene in simple distillation
 | Lead Academy answered |
In steam distillation of toluene, the pressure of toluene in vapour is less than pressure of barometer, because it is carried out when a solid or liquid is insoluble in water and is volatile with steam but the impurities are non-volatile.
Which of the statements is not true?- a)On passing H2S through acidified K2Cr2O7 solution, a milky colour is observed
- b)Na2Cr2O7 is preferred over K2Cr2O7 in volumetric analysis
- c)K2Cr2O7 solution in acidic medium is orange
- d)K2Cr2O7 solution becomes yellow on increasing the pH beyond 7
Correct answer is option 'B'. Can you explain this answer?
Which of the statements is not true?
a)
On passing H2S through acidified K2Cr2O7 solution, a milky colour is observed
b)
Na2Cr2O7 is preferred over K2Cr2O7 in volumetric analysis
c)
K2Cr2O7 solution in acidic medium is orange
d)
K2Cr2O7 solution becomes yellow on increasing the pH beyond 7
 | Ciel Knowledge answered |
Being hygroscopic, sodium dichromate, Na2Cr2O7 cannot be used in volumetric analysis.
All other given statements are true.
All other given statements are true.
The most suitable method of separation of 1:1 mixture of ortho and para-nitrophenols is- a)sublimation
- b)chromatography
- c)crystallisation
- d)steam distillation
Correct answer is option 'D'. Can you explain this answer?
The most suitable method of separation of 1:1 mixture of ortho and para-nitrophenols is
a)
sublimation
b)
chromatography
c)
crystallisation
d)
steam distillation
 | Stepway Academy answered |
Steam distillation is used to purify the substances which
(i) are volatile in steam but are immiscible with water.
(ii) possess sufficiently high vapour pressure at the boiling point of water.
(iii) contain non- volatile impurities.
The process of steam distillation can also be used to separate a mixture of two organic compounds one of which is steam volatile while the other is not.
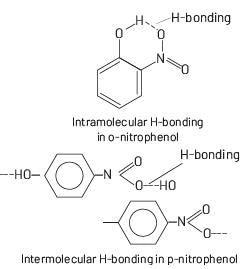
In ortho and para-nitrophenol, ortho-nitrophenol has intramolecular H-bonding. So, it has lower boiling point.
Intermolecular H-Bonding more strong then intramolecular H-bonding. Whereas para-nitrophenol has intermolecular H-bonding. So, it has higher boiling point.
Due to difference in boiling points ortho and para-nitrophenol can be separated from each other by distillation.
(i) are volatile in steam but are immiscible with water.
(ii) possess sufficiently high vapour pressure at the boiling point of water.
(iii) contain non- volatile impurities.
The process of steam distillation can also be used to separate a mixture of two organic compounds one of which is steam volatile while the other is not.
In ortho and para-nitrophenol, ortho-nitrophenol has intramolecular H-bonding. So, it has lower boiling point.
Intermolecular H-Bonding more strong then intramolecular H-bonding. Whereas para-nitrophenol has intermolecular H-bonding. So, it has higher boiling point.
Due to difference in boiling points ortho and para-nitrophenol can be separated from each other by distillation.

Which of the following techniques is most suitable for purification of cyclohexanone from a mixture containing benzoic acid, isoamyl alcohol, cyclohexane and cyclohexanone?- a)Crystallisation
- b)IR spectroscopy
- c)Sublimation
- d)Evaporation
Correct answer is option 'B'. Can you explain this answer?
Which of the following techniques is most suitable for purification of cyclohexanone from a mixture containing benzoic acid, isoamyl alcohol, cyclohexane and cyclohexanone?
a)
Crystallisation
b)
IR spectroscopy
c)
Sublimation
d)
Evaporation
 | Lead Academy answered |
IR spectroscopy is used for the purification of cyclohexanone from a mixture of benzoic acid, isoamyl alcohol, cyclohexane and cyclohexanone because in IR spectroscopy each functional group appears at a certain peak. IR spectroscopy exploits the fact that molecules absorb specific frequencies that are characteristic of their structure.
Prussian blue is formed when- a)ferrous sulphate reacts with FeCl3
- b)ferric sulphate reacts with Na4[Fe(CN)6]
- c)ferrous ammonium sulphate reacts with FeCl3
- d)ammonium sulphate reacts with FeCl3
Correct answer is option 'B'. Can you explain this answer?
Prussian blue is formed when
a)
ferrous sulphate reacts with FeCl3
b)
ferric sulphate reacts with Na4[Fe(CN)6]
c)
ferrous ammonium sulphate reacts with FeCl3
d)
ammonium sulphate reacts with FeCl3
 | Bs Academy answered |
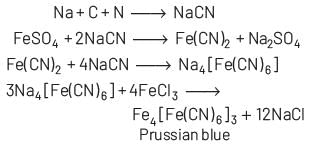
When the sodium fusion extract is added with FeCl3 and then the resulting solution is acidified with dilute hydrochloric acid, the appearance of Prussian blue colouration confirms the presence of nitrogen in the organic compound.

Lassaigne’s test for the detection of nitrogen fails in- a)NH2CONHNH2⋅HCl
- b)NH2NH2⋅HCl
- c)NH2CONH2
- d)C6H5NHNH2⋅HCl
Correct answer is option 'B'. Can you explain this answer?
Lassaigne’s test for the detection of nitrogen fails in
a)
NH2CONHNH2⋅HCl
b)
NH2NH2⋅HCl
c)
NH2CONH2
d)
C6H5NHNH2⋅HCl
 | Ambition Institute answered |
Lassaigne’s test is given by only those compounds which contain both carbon and nitrogen. When compounds containing C and N heated with sodium, then it form NaCN which is easily detected byFeCl3.
Or
Some compounds live hydrazine (NH2⋅ NH2) although contain nitrogen but they do not respond Lassaigne’s test because they do not have any carbon and hence, NaCN is not formed.
Or
Some compounds live hydrazine (NH2⋅ NH2) although contain nitrogen but they do not respond Lassaigne’s test because they do not have any carbon and hence, NaCN is not formed.
The Lassaigne’s extract is boiled with conc. HNO3 while testing for halogens. By doing so it- a)helps in the precipitation of AgCl
- b)increases the solubility product of AgCl
- c)increases the concentration of NO3–ions
- d)decomposes Na2S and NaCN, if formed
Correct answer is option 'D'. Can you explain this answer?
The Lassaigne’s extract is boiled with conc. HNO3 while testing for halogens. By doing so it
a)
helps in the precipitation of AgCl
b)
increases the solubility product of AgCl
c)
increases the concentration of NO3–ions
d)
decomposes Na2S and NaCN, if formed
 | Bs Academy answered |
Na2S and NaCN, if present in the extract, will be decomposed to H2S and HCN by HNO3.
NaCN + HNO3 → NaNO3 + HCN
Na2S + 2HNO3 → 2NaNO3 + H2S
These will escape from the solution and will not interfere with the test for halogens.
NaCN + HNO3 → NaNO3 + HCN
Na2S + 2HNO3 → 2NaNO3 + H2S
These will escape from the solution and will not interfere with the test for halogens.
Chapter doubts & questions for Purification and Characterisation of Organic Compounds - 30-Day Revision Course for NEET 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Purification and Characterisation of Organic Compounds - 30-Day Revision Course for NEET in English & Hindi are available as part of NEET exam. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
;
Signup to see your scores go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup

