All Exams > NEET > 30-Day Revision Course for NEET > All Questions
All questions of Nuclei for NEET Exam
A radioactive material decays by simultaneous emission of two particles with respective half lives 1620 and 810 years. The time in years, after which one fourth of the material remains is- a)4860
- b)2340
- c)1080.0
- d)3240
Correct answer is option 'C'. Can you explain this answer?
A radioactive material decays by simultaneous emission of two particles with respective half lives 1620 and 810 years. The time in years, after which one fourth of the material remains is
a)
4860
b)
2340
c)
1080.0
d)
3240
| | Nikita Singh answered |
Since, from Rutherford-Soddy law, the number of atoms left after half-lives is given by
N=N0(1/2)n
where, N0 is the original number of atoms.
The number of half-lives, n= time of decay/effective half−life
Relation between effective disintegration constant (λ) and half-life (T)
λ=ln2/T
∴λ1+λ2= (ln2/ T1)+ (ln2/ T2)
Effective half-life,
1/T=1/T1+1/T2=(1/1620)+(1/810)
1/T=1+2/1620 ⇒T=540yr
∴n=T/540
∴N=N0(1/2)t/540⇒N/N0=(1/2)2=(1/2)t/540
⇒t/540=2⇒t=2×540=1080yr
N=N0(1/2)n
where, N0 is the original number of atoms.
The number of half-lives, n= time of decay/effective half−life
Relation between effective disintegration constant (λ) and half-life (T)
λ=ln2/T
∴λ1+λ2= (ln2/ T1)+ (ln2/ T2)
Effective half-life,
1/T=1/T1+1/T2=(1/1620)+(1/810)
1/T=1+2/1620 ⇒T=540yr
∴n=T/540
∴N=N0(1/2)t/540⇒N/N0=(1/2)2=(1/2)t/540
⇒t/540=2⇒t=2×540=1080yr
Cadmium rods are used in a nuclear reactor for- a)absorbing neutrons
- b)speeding up slow neutrons
- c)regulating the power level of the reactor.
- d)slowing down fast neutrons
Correct answer is option 'A'. Can you explain this answer?
Cadmium rods are used in a nuclear reactor for
a)
absorbing neutrons
b)
speeding up slow neutrons
c)
regulating the power level of the reactor.
d)
slowing down fast neutrons
 | Awantika Gupta answered |
Cadmium and boron rod both are used in cotroling the reactivity of uranium means slow down the rate of fission...
All nuclides with same mass number A are called- a)isobars
- b)isoclines
- c)isotones
- d)isotopes
Correct answer is option 'A'. Can you explain this answer?
All nuclides with same mass number A are called
a)
isobars
b)
isoclines
c)
isotones
d)
isotopes
| | Rocky Handsome answered |
Isobars are atoms of different elements with the same mass number but different atomic numbers.
• Isotones are atomic nuclei with the same number of neutrons (N) and different number of protons(Z)
• Isotones are atomic nuclei with the same number of neutrons (N) and different number of protons(Z)
90% of a radioactive sample is left undisintegrated after time τ has elapsed, what percentage of initial sample will decay in a total time2τ?- a)9%
- b)38%
- c)19%
- d)62%
Correct answer is option 'C'. Can you explain this answer?
90% of a radioactive sample is left undisintegrated after time τ has elapsed, what percentage of initial sample will decay in a total time2τ?
a)
9%
b)
38%
c)
19%
d)
62%
| | Krishna Iyer answered |
Given that 90% is left un-decayed after time 't'.
Hence, 10% decays in time 't'.
Initially assume that the amount of substance is 'x'
After time 't' 10% is decayed.
i.e. Amount of substance left =0.9x
After further time 't' another 10% is decayed.
i.e. 0.1×0.9x is decayed
Leaving behind 0.81x.
Hence after time 2t we see that 0.19x has decayed, which is 19%.
Hence, 10% decays in time 't'.
Initially assume that the amount of substance is 'x'
After time 't' 10% is decayed.
i.e. Amount of substance left =0.9x
After further time 't' another 10% is decayed.
i.e. 0.1×0.9x is decayed
Leaving behind 0.81x.
Hence after time 2t we see that 0.19x has decayed, which is 19%.
α-rays are- a)helium nuclei
- b)heavy nuclei
- c)lithium nuclei
- d)hydrogen nuclei
Correct answer is option 'A'. Can you explain this answer?
α-rays are
a)
helium nuclei
b)
heavy nuclei
c)
lithium nuclei
d)
hydrogen nuclei
| | Ræjû Bhæï answered |
Alpha particles, also called alpha rays or alpha radiation, consist of two protons and two neutrons bound together into a particle identical to a helium-4 nucleus. They are generally produced in the process of alpha decay, but may also be produced in other ways.
At a given time there are 25% undecayed nuclei in a sample. After 10 seconds number of undecayed nuclei reduces to 12.5%. Then mean life of the nuclei will be about- a)22 sec
- b)10 sec
- c)12 sec
- d)15 sec
Correct answer is option 'D'. Can you explain this answer?
At a given time there are 25% undecayed nuclei in a sample. After 10 seconds number of undecayed nuclei reduces to 12.5%. Then mean life of the nuclei will be about
a)
22 sec
b)
10 sec
c)
12 sec
d)
15 sec
| | Lavanya Menon answered |
Half-life of radioactive sample, i.e., the time in which the number of undecayed nuclei becomes half (T) is 10 s.
Mean life, τ=T/loge2=10s/0.693=1.443×10=14.43s ≈ 15s
Mean life, τ=T/loge2=10s/0.693=1.443×10=14.43s ≈ 15s
The half life of radon is 3.8 days. After how many days will only one twentieth of a radon sample be left over?
a)10.00 daysb)5.45 daysc)15.45 daysd)16.45 daysCorrect answer is option 'D'. Can you explain this answer?
| | Suresh Iyer answered |
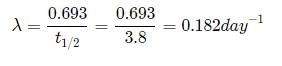 Let the initial amount of radon be N0 and the amount left after t days be N which is equal to N0/2
Let the initial amount of radon be N0 and the amount left after t days be N which is equal to N0/2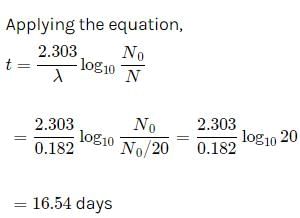
What is the main source of energy of the sun?- a)Nuclear fission of heavier unstable elements in the sun
- b)Combustion of pure carbon present in the sun
- c)Gravitational energy liberated during the slow contraction of the sun.
- d)Nuclear fusion of lighter elements in the sun.
Correct answer is option 'D'. Can you explain this answer?
What is the main source of energy of the sun?
a)
Nuclear fission of heavier unstable elements in the sun
b)
Combustion of pure carbon present in the sun
c)
Gravitational energy liberated during the slow contraction of the sun.
d)
Nuclear fusion of lighter elements in the sun.
 | Kanika S answered |
Option d
When H and He in sun undergoes fusion large amount of heat is released.
When H and He in sun undergoes fusion large amount of heat is released.
Nuclear mass M is found to be- a)always greater than total mass of its individual protons and neutrons
- b)always equal to the total mass of its individual neutrons
- c)always equal to the total mass of its individual protons and neutrons
- d)always less than total mass of its individual protons and neutrons
Correct answer is option 'D'. Can you explain this answer?
Nuclear mass M is found to be
a)
always greater than total mass of its individual protons and neutrons
b)
always equal to the total mass of its individual neutrons
c)
always equal to the total mass of its individual protons and neutrons
d)
always less than total mass of its individual protons and neutrons
| | Ritu Singh answered |
The actual mass is always less than the sum of the individual masses of the constituent protons and neutrons because energy is removed when the nucleus is formed. This energy has mass, which is removed from the total mass of the original particles.
The average number of neutrons released by the fission of one uranium atom is
a)3.0b)2c)2.5d)1Correct answer is option 'C'. Can you explain this answer?
| | Bhanu Saini answered |
Fission result in the production of typically 2 or 3 neutron so on the average about 2.5 neutron released per unit. so correct answer is option a
for option c one uranium atom split into one barium and one krypton atom releasing 3 neutron.
but in this question average is asking so according to me and books 2.5 is correct
for option c one uranium atom split into one barium and one krypton atom releasing 3 neutron.
but in this question average is asking so according to me and books 2.5 is correct
What percentage of the mass of an atom is concentrated in the nucleus?- a)79.9%
- b)99.9%
- c)66.9%
- d)50.9%
Correct answer is option 'B'. Can you explain this answer?
What percentage of the mass of an atom is concentrated in the nucleus?
a)
79.9%
b)
99.9%
c)
66.9%
d)
50.9%
| | Jyoti Kapoor answered |
More than 99.99% of the mass of any atom is concentrated in its nucleus. If the mass of protons and neutrons (which are in the nucleus of every atom) is approximately one (1) atomic mass unit, then the relative mass of an electron is 0.0005 atomic mass units.
Plutonium decays with a half-life of 24000 years. If the plutonium is stored for 72000 years, then the fraction of plutonium that remains is - a)1 /3
- b)1 /2
- c)1/8
- d)1 /4
Correct answer is option 'C'. Can you explain this answer?
Plutonium decays with a half-life of 24000 years. If the plutonium is stored for 72000 years, then the fraction of plutonium that remains is
a)
1 /3
b)
1 /2
c)
1/8
d)
1 /4
| | Mira Sharma answered |
The amount of plotinium after a time period of 72000 if the half life is 24000 will be
the initial amount x would be reduced to x/2 , in 24000 yrs
then it would lessen to x/4 in the next 24000yrs
and then to x/8 in the next 24000 yrs
that is it will reduce to x/8 in the next 72000yrs starting from x .
In the mass number range A = 30 to 170, the binding energy per nucleon is- a)decreases with increasing A
- b)increases linearly with A
- c)decreases linearly with A
- d)nearly constant
Correct answer is option 'D'. Can you explain this answer?
In the mass number range A = 30 to 170, the binding energy per nucleon is
a)
decreases with increasing A
b)
increases linearly with A
c)
decreases linearly with A
d)
nearly constant
| | Shraddha Choudhury answered |
Binding energy per nucleon in the mass number range A = 30 to 170
The binding energy per nucleon is the energy required to separate a nucleus into its constituent nucleons. It is a measure of the stability of the nucleus, and it depends on the mass number of the nucleus. In the mass number range A = 30 to 170, the binding energy per nucleon is nearly constant. This means that the stability of the nucleus is nearly constant in this range.
Explanation:
The binding energy per nucleon is given by the formula:
BE/A = (ZmH + NmN - M)/A
where BE is the binding energy, Z is the atomic number, N is the number of neutrons, mH is the mass of a hydrogen atom, mN is the mass of a neutron, and M is the mass of the nucleus.
In the mass number range A = 30 to 170, the binding energy per nucleon is nearly constant because the nuclear force between nucleons is nearly constant. This means that the energy required to separate a nucleon from the nucleus is nearly constant in this range.
The nuclear force between nucleons is a strong force that holds the nucleus together. It is a short-range force that depends on the distance between nucleons. In the mass number range A = 30 to 170, the distance between nucleons is nearly constant, and so the nuclear force is nearly constant.
Therefore, the binding energy per nucleon is nearly constant in this range because the nuclear force is nearly constant. This means that the stability of the nucleus is nearly constant in this range.
The binding energy per nucleon is the energy required to separate a nucleus into its constituent nucleons. It is a measure of the stability of the nucleus, and it depends on the mass number of the nucleus. In the mass number range A = 30 to 170, the binding energy per nucleon is nearly constant. This means that the stability of the nucleus is nearly constant in this range.
Explanation:
The binding energy per nucleon is given by the formula:
BE/A = (ZmH + NmN - M)/A
where BE is the binding energy, Z is the atomic number, N is the number of neutrons, mH is the mass of a hydrogen atom, mN is the mass of a neutron, and M is the mass of the nucleus.
In the mass number range A = 30 to 170, the binding energy per nucleon is nearly constant because the nuclear force between nucleons is nearly constant. This means that the energy required to separate a nucleon from the nucleus is nearly constant in this range.
The nuclear force between nucleons is a strong force that holds the nucleus together. It is a short-range force that depends on the distance between nucleons. In the mass number range A = 30 to 170, the distance between nucleons is nearly constant, and so the nuclear force is nearly constant.
Therefore, the binding energy per nucleon is nearly constant in this range because the nuclear force is nearly constant. This means that the stability of the nucleus is nearly constant in this range.
In what units is mass measured on the atomic scale?- a)kilogram
- b)atomic mass units (u)
- c)milligram
- d)gram
Correct answer is option 'B'. Can you explain this answer?
In what units is mass measured on the atomic scale?
a)
kilogram
b)
atomic mass units (u)
c)
milligram
d)
gram
| | Lavanya Menon answered |
The atomic mass of an element is the average mass of the atoms of an element measured in atomic mass unit (amu, also known as daltons, D). The atomic mass is a weighted average of all of the isotopes of that element, in which the mass of each isotope is multiplied by the abundance of that particular isotope.
The nuclei of isotopes of a given element contain the same number of- a)neutrinos
- b)protons
- c)neutrons
- d)positrons
Correct answer is option 'B'. Can you explain this answer?
The nuclei of isotopes of a given element contain the same number of
a)
neutrinos
b)
protons
c)
neutrons
d)
positrons
| | Varanasi Sai Srinivasa K answered |
Atom of same element, contain same number of protons, they differ in number of neutrons .
This is known as isotope .
Therefore we can conclude that answer is [ B ]
This is known as isotope .
Therefore we can conclude that answer is [ B ]
The chemical behavior of an atom depends upon- a)the number of neutrons in its nucleus
- b)the number of nucleons in its nucleus.
- c)the number of electrons orbiting around its nucleus
- d)the number of protons in its nucleus
Correct answer is option 'C'. Can you explain this answer?
The chemical behavior of an atom depends upon
a)
the number of neutrons in its nucleus
b)
the number of nucleons in its nucleus.
c)
the number of electrons orbiting around its nucleus
d)
the number of protons in its nucleus
 | Kuheli Sengupta answered |
This is because in most chemical reactions only electrons participate. the nucleons have little role in chemical reactions
Nuclear fusion is possible- a)only between light nuclei
- b)only between heavy nuclei
- c)between both light and heavy nuclei
- d)only between nuclei which are stable against β-decay
Correct answer is option 'A'. Can you explain this answer?
Nuclear fusion is possible
a)
only between light nuclei
b)
only between heavy nuclei
c)
between both light and heavy nuclei
d)
only between nuclei which are stable against β-decay
| | Harshit Agrawal answered |
In nuclear fusion, two or more small nuclei combine to form a single larger nucleus, a neutron, and a tremendous amount of energy. Nuclear fusion of hydrogen to form helium occurs naturally in the sun and other stars. It takes place only at extremely high temperatures.
A free neutron decays into- a)a proton, a positron and a antineutrino
- b)a proton, a positron and a neutrino
- c)a proton, an electron and an antineutrino
- d)a proton, an electron and a neutrino
Correct answer is option 'C'. Can you explain this answer?
A free neutron decays into
a)
a proton, a positron and a antineutrino
b)
a proton, a positron and a neutrino
c)
a proton, an electron and an antineutrino
d)
a proton, an electron and a neutrino
 | Knowledge Hub answered |
Neutron is unstable outside the nucleus.
It's decaying equation is given below.
0n1⇒11H+0−1e+v
It's decaying equation is given below.
0n1⇒11H+0−1e+v
B210has a half life of 5 days. The time taken for seven-eighth of a sample to decay is- a)10 days
- b)20 days
- c)3.4 days
- d)15 days
Correct answer is option 'D'. Can you explain this answer?
B210has a half life of 5 days. The time taken for seven-eighth of a sample to decay is
a)
10 days
b)
20 days
c)
3.4 days
d)
15 days
| | Srishti Chavan answered |
Half-life of Bi210=5 days
∴k= 0.693/(t1/2) =(0.693/5) day−1
Using k=(2.303/t) log (a/a-x)
(where a = a0, (let) ⇒x=7/8 a0, t is time taken in decay and k is rate constant)
We get, t=(2.303×5/0.693)log a0/(1/8)a0
= (2.303×5/0.693) log8=15days
∴k= 0.693/(t1/2) =(0.693/5) day−1
Using k=(2.303/t) log (a/a-x)
(where a = a0, (let) ⇒x=7/8 a0, t is time taken in decay and k is rate constant)
We get, t=(2.303×5/0.693)log a0/(1/8)a0
= (2.303×5/0.693) log8=15days
Choose the WRONG statement. A thermonuclear fusion reactor is better than a fission reactor for the following reason:- a)For the same mass of substances involved, a fusion reaction releases much more energy than a fission reaction.
- b)The fuel required for fusion is readily available in abundance from seawater.
- c)A fusion reaction can be much more easily controlled than a fission
- d)A fusion reaction produces almost no radioactive waste.
Correct answer is option 'B'. Can you explain this answer?
Choose the WRONG statement. A thermonuclear fusion reactor is better than a fission reactor for the following reason:
a)
For the same mass of substances involved, a fusion reaction releases much more energy than a fission reaction.
b)
The fuel required for fusion is readily available in abundance from seawater.
c)
A fusion reaction can be much more easily controlled than a fission
d)
A fusion reaction produces almost no radioactive waste.
| | Nisha Kulkarni answered |
Explanation:
The wrong statement among the given options is option B: "The fuel required for fusion is readily available in abundance from seawater."
Reason:
- While it is correct that a thermonuclear fusion reactor is better than a fission reactor for several reasons, including higher energy release and less radioactive waste production, the availability of fuel from seawater is not accurate.
- Fusion reactions require isotopes of hydrogen, such as deuterium and tritium, as fuel. Deuterium can be extracted from seawater, but tritium is a radioactive isotope that is not naturally abundant and needs to be produced artificially.
- Tritium can be produced by exposing lithium to neutron radiation, which can be generated by a fission reactor or a fusion reactor itself. However, the process of producing tritium is not as straightforward as extracting deuterium from seawater.
- Tritium is also highly radioactive and has a short half-life, which means it requires careful handling and containment. It cannot be easily stored or transported.
- Therefore, the fuel required for fusion reactions is not readily available in abundance from seawater, as stated in option B.
Correct statements:
a) For the same mass of substances involved, a fusion reaction releases much more energy than a fission reaction.
- This is true. Fusion reactions release a tremendous amount of energy, several times more than fission reactions. The fusion of hydrogen atoms into helium is the same process occurring in the Sun and other stars, which produces immense amounts of energy.
c) A fusion reaction can be much more easily controlled than a fission reaction.
- This is true. Fusion reactions require extremely high temperatures and pressures to sustain, and if these conditions are not maintained, the reaction will cease. This inherent stability makes fusion reactions more easily controllable than fission reactions, which can lead to runaway chain reactions if not properly regulated.
d) A fusion reaction produces almost no radioactive waste.
- This is true. Fusion reactions do not produce long-lived radioactive waste like fission reactions. The only radioactive byproduct of fusion is tritium, which has a relatively short half-life and can be managed safely.
In summary, option B is the wrong statement because the fuel required for fusion reactions is not readily available in abundance from seawater.
The wrong statement among the given options is option B: "The fuel required for fusion is readily available in abundance from seawater."
Reason:
- While it is correct that a thermonuclear fusion reactor is better than a fission reactor for several reasons, including higher energy release and less radioactive waste production, the availability of fuel from seawater is not accurate.
- Fusion reactions require isotopes of hydrogen, such as deuterium and tritium, as fuel. Deuterium can be extracted from seawater, but tritium is a radioactive isotope that is not naturally abundant and needs to be produced artificially.
- Tritium can be produced by exposing lithium to neutron radiation, which can be generated by a fission reactor or a fusion reactor itself. However, the process of producing tritium is not as straightforward as extracting deuterium from seawater.
- Tritium is also highly radioactive and has a short half-life, which means it requires careful handling and containment. It cannot be easily stored or transported.
- Therefore, the fuel required for fusion reactions is not readily available in abundance from seawater, as stated in option B.
Correct statements:
a) For the same mass of substances involved, a fusion reaction releases much more energy than a fission reaction.
- This is true. Fusion reactions release a tremendous amount of energy, several times more than fission reactions. The fusion of hydrogen atoms into helium is the same process occurring in the Sun and other stars, which produces immense amounts of energy.
c) A fusion reaction can be much more easily controlled than a fission reaction.
- This is true. Fusion reactions require extremely high temperatures and pressures to sustain, and if these conditions are not maintained, the reaction will cease. This inherent stability makes fusion reactions more easily controllable than fission reactions, which can lead to runaway chain reactions if not properly regulated.
d) A fusion reaction produces almost no radioactive waste.
- This is true. Fusion reactions do not produce long-lived radioactive waste like fission reactions. The only radioactive byproduct of fusion is tritium, which has a relatively short half-life and can be managed safely.
In summary, option B is the wrong statement because the fuel required for fusion reactions is not readily available in abundance from seawater.
Which of the following particles can be added to the nucleus of an atom without changing its chemical properties?- a)Alpha Particles
- b)Protons
- c)Neutrons
- d)Electrons
Correct answer is option 'C'. Can you explain this answer?
Which of the following particles can be added to the nucleus of an atom without changing its chemical properties?
a)
Alpha Particles
b)
Protons
c)
Neutrons
d)
Electrons
| | Rajat Kapoor answered |
Adding a neutron to the nucleus will make no change in the chemical properties of the atom. The atom will have the same number of protons and therefore the same number of electrons. It is the number of electrons that determines chemical properties.
Actually, with hydrogen the addition of a neutron will almost double its mass and thus cause it to behave a little differently chemically.
Of course if the added neutron causes the nucleus to fission, decay, or otherwise change, that will change the chemistry.
When a hydrogen bomb explodes, which of the following is used?- a)fission
- b)both
- c)neither of two
- d)fusion
Correct answer is option 'B'. Can you explain this answer?
When a hydrogen bomb explodes, which of the following is used?
a)
fission
b)
both
c)
neither of two
d)
fusion
| | Naina Bansal answered |
Hydrogen bomb or H-bomb, weapon deriving a large portion of its energy from the nuclear fusion of hydrogen isotopes. In an atomic bomb, uranium or plutonium is split into lighter elements that together weigh less than the original atoms, the remainder of the mass appearing as energy. Unlike this fission bomb, the hydrogen bomb functions by the fusion, or joining together, of lighter elements into heavier elements. The end product again weighs less than its components, the difference once more appearing as energy. Because extremely high temperatures are required in order to initiate fusion reactions, the hydrogen bomb is also known as a thermonuclear bomb.
The disintegration energy is- a)the difference between the initial energy and the total energy of the decay products
- b)the difference between the initial mass and the total mass of the decay products
- c)the difference between the initial mass energy and the total mass energy of the decay products
- d)none of the above
Correct answer is option 'C'. Can you explain this answer?
The disintegration energy is
a)
the difference between the initial energy and the total energy of the decay products
b)
the difference between the initial mass and the total mass of the decay products
c)
the difference between the initial mass energy and the total mass energy of the decay products
d)
none of the above
| | Rakibul Halsana answered |
CCCC
β -rays and γ-rays are respectively- a)neutrons and electromagnetic radiation of wavelengths shorter than X-rays
- b)protons and neutrons of wavelengths shorter than X-rays
- c)protons and electromagnetic radiation of wavelengths shorter than X-rays
- d)electrons and electromagnetic radiation of wavelengths shorter than X-rays
Correct answer is option 'D'. Can you explain this answer?
β -rays and γ-rays are respectively
a)
neutrons and electromagnetic radiation of wavelengths shorter than X-rays
b)
protons and neutrons of wavelengths shorter than X-rays
c)
protons and electromagnetic radiation of wavelengths shorter than X-rays
d)
electrons and electromagnetic radiation of wavelengths shorter than X-rays
| | Divyansh Kulkarni answered |
Beta radiation ~ Stream of electrons (unit negative charge). Beta positive radiation is when a positron is emitted rather than an electron.
Gamma radiation ~ Electromagnetic radiation of very short wavelength = high photon energy.
Gamma radiation ~ Electromagnetic radiation of very short wavelength = high photon energy.
When a hydrogen bomb explodes, which of the following is used?- a)fission
- b)fusion
- c)neither of two
- d)both
Correct answer is option 'D'. Can you explain this answer?
When a hydrogen bomb explodes, which of the following is used?
a)
fission
b)
fusion
c)
neither of two
d)
both
 | Dr Manju Sen answered |
The hydrogen bomb is a nuclear weapon that uses a mixture of fission and fusion to produce a massive explosion.
The nuclear fission generates enough heat to initiate the nuclear fusion reaction. After that, the nuclear fusion releases enormous amounts of energy, making the hydrogen bomb a lot more powerful than an atomic bomb.
The nuclear fission generates enough heat to initiate the nuclear fusion reaction. After that, the nuclear fusion releases enormous amounts of energy, making the hydrogen bomb a lot more powerful than an atomic bomb.
Nuclear forces are- a)spin dependent and have no non-central part
- b)spin dependent and have a non-central part
- c)spin independent and have no non-central part
- d)spin independent and have a non-central part
Correct answer is option 'D'. Can you explain this answer?
Nuclear forces are
a)
spin dependent and have no non-central part
b)
spin dependent and have a non-central part
c)
spin independent and have no non-central part
d)
spin independent and have a non-central part
 | EduRev JEE answered |
Nuclear forces are fundamental interactions that govern the behaviour of protons and neutrons within an atomic nucleus. Here are some key characteristics:
- The nuclear force is spin independent, meaning it does not rely on the spin orientation of nucleons.
- It has a non-central part, which implies that the force varies with the spatial arrangement of nucleons.
- This force is significantly stronger than the Coulomb force, which acts between charged particles.
- The nuclear force operates over a very short range, typically around a few femtometres (fm).
In summary, the nuclear force is essential for holding the nucleus together, overcoming the repulsive forces between positively charged protons, and ensuring the stability of atomic nuclei.
Energies associated with nuclear processes are- a)about a 100 times larger than chemical process
- b)about a million times larger than chemical process
- c)about a thousand times larger than chemical process
- d)about a ten times larger than chemical process
Correct answer is option 'B'. Can you explain this answer?
Energies associated with nuclear processes are
a)
about a 100 times larger than chemical process
b)
about a million times larger than chemical process
c)
about a thousand times larger than chemical process
d)
about a ten times larger than chemical process
| | Keerthana Chakraborty answered |
Unlike a chemical reaction, a nuclear reaction results in a significant change in mass and an associated change of energy, as described by Einstein’s equation. Nuclear reactions are accompanied by large changes in energy, which result in detectable changes in mass. The change in mass is related to the change in energy according to Einstein’s equation: ΔE = (Δm)c2. Large changes in energy are usually reported in kiloelectronvolts or megaelectronvolts (thousands or millions of electronvolts). With the exception of 1H, the experimentally determined mass of an atom is always less than the sum of the masses of the component particles (protons, neutrons, and electrons) by an amount called the mass defect of the nucleus. The energy corresponding to the mass defect is the nuclear binding energy, the amount of energy released when a nucleus forms from its component particles. In nuclear fission, nuclei split into lighter nuclei with an accompanying release of multiple neutrons and large amounts of energy. The critical mass is the minimum mass required to support a self-sustaining nuclear chain reaction. Nuclear fusion is a process in which two light nuclei combine to produce a heavier nucleus plus a great deal of energy.
Chapter doubts & questions for Nuclei - 30-Day Revision Course for NEET 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Nuclei - 30-Day Revision Course for NEET in English & Hindi are available as part of NEET exam. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
;
Signup to see your scores go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup


