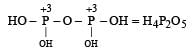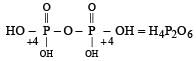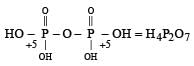All Exams > NEET > Chemistry Class 12 > All Questions
All questions of p-Block Elements (Only in NEET) for NEET Exam
Which is the strongest acid in the following : [NEET 2013]- a)HClO3
- b)HClO4
- c)H2SO3
- d)H2SO4
Correct answer is option 'B'. Can you explain this answer?
Which is the strongest acid in the following : [NEET 2013]
a)
HClO3
b)
HClO4
c)
H2SO3
d)
H2SO4
 | Surbhi Das answered |
HClO4 is the strongest acid amongst all because the oxidation state or Cl is maximum (+7).
Which of the following is a polar molecule ? [NEET 2013]- a)SF4
- b)SiF4
- c)XeF4
- d)BF3
Correct answer is option 'A'. Can you explain this answer?
Which of the following is a polar molecule ? [NEET 2013]
a)
SF4
b)
SiF4
c)
XeF4
d)
BF3
 | Rajesh Datta answered |
SF4 has 4 bond pairs and 1 lone pair of electrons, sp3d hybridisation leads to irregular shape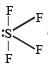 and resultant
and resultant
 and resultant
and resultantμ ≠ 0.
Among the following compounds, the number of compounds which have oxidation states of S is +4 ?PbS, SO2, SF6, Na2S2O3, H2SO3
Correct answer is '2'. Can you explain this answer?
Among the following compounds, the number of compounds which have oxidation states of S is +4 ?
PbS, SO2, SF6, Na2S2O3, H2SO3
| | Shreya Singh answered |
SO2...x+2(-2)=0.x=4..H2SO3.2(1)+x+3(-2)=0.2+x-6=0.-4+x=0.x=4...
In the MOT of F2 molecule, number of electrons occupying antibonding orbitals are
Correct answer is '8'. Can you explain this answer?
In the MOT of F2 molecule, number of electrons occupying antibonding orbitals are
| | Nandita Ahuja answered |
Fluorine atom have 2+7 electrons so an F2 molecule contain 18 electrons.
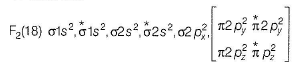
Hence, 8 electrons occupy the antibonding orbitals.
Hence, 8 electrons occupy the antibonding orbitals.
The correct statements among the following are- a)Bond lengths in O2 ,
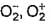 are 121 , 134, 149 pm
are 121 , 134, 149 pm - b)Ozone is stronger oxidising agent than dioxygen
- c)O2 acts as reducing agent when it reacts with powerful oxidising agents like PtF6
- d)Ozone is much more stable than oxygen
Correct answer is option 'A,B,C'. Can you explain this answer?
The correct statements among the following are
a)
Bond lengths in O2 , 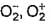 are 121 , 134, 149 pm
are 121 , 134, 149 pm
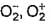 are 121 , 134, 149 pm
are 121 , 134, 149 pmb)
Ozone is stronger oxidising agent than dioxygen
c)
O2 acts as reducing agent when it reacts with powerful oxidising agents like PtF6
d)
Ozone is much more stable than oxygen
 | Srishti Kaur answered |
The correct option is Option A, B and C.
Bond length is inversely proportional to bond order. O2+ has the highest bond order among these three, so it should have the shortest bond length.
Ozone is a powerful oxidizing agent as compared to oxygen. This is due to the unstable nature of ozone and the nascent oxygen that is released during the reaction.
O2 when gas makes others like H2 gas to lose electrons, therefore, O2 gas is an oxidizing agent and H2 when gas loses electrons in redox reaction, therefore H2 gas is a reducing agent.
Oxygen is more stable than ozone. On heating, ozone readily dissociates and forms oxygen and free radicals of oxygen known as nascent oxygen which take part in reaction, thus ozone is more reactive than oxygen
Which of the following statement is incorrect ?- a)SRP values of halogens F2 > Cl2 > Br2 > I2
- b)Bond dissociation enthalpy of Br2 > F2 > Cl2 > I2
- c)Boiling points of I2 > Br2 > Cl2 > F2
- d)Reducing power of I- > Br- > Cl- > F-
Correct answer is option 'B'. Can you explain this answer?
Which of the following statement is incorrect ?
a)
SRP values of halogens F2 > Cl2 > Br2 > I2
b)
Bond dissociation enthalpy of Br2 > F2 > Cl2 > I2
c)
Boiling points of I2 > Br2 > Cl2 > F2
d)
Reducing power of I- > Br- > Cl- > F-
| | Preeti Khanna answered |
The correct order of bond dissociation enthalpy is
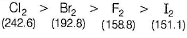
Which of the following is thermally the most stable?- a)H2Te
- b)H2S
- c)H2O
- d)H2Se
Correct answer is option 'C'. Can you explain this answer?
Which of the following is thermally the most stable?
a)
H2Te
b)
H2S
c)
H2O
d)
H2Se
 | Ujwal Patel answered |
Stability of hydrides decreases down the group so most stable is H2O
Direction (Q. Nos. 8-12) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.Q. Which of the following can act as dehydrating agent ?- a)P4O10
- b)POCI3
- c)Cone.H2SO4
- d)P4O6
Correct answer is option 'A,B,C'. Can you explain this answer?
Direction (Q. Nos. 8-12) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q.
Which of the following can act as dehydrating agent ?
a)
P4O10
b)
POCI3
c)
Cone.H2SO4
d)
P4O6
 | Keshav Bhatheja answered |
Conc h2so4 is a very good dehydrating agent as extract h2o from reaction very easily...by breaking into hso4- and h+ and the rest of two i dont know i think we have to learn them
The number of P – O bonds and lone pairs of electron present in P4O6 molecule respectively - a)12 and 4
- b)8 and 8
- c)12 and 16
- d)12 and 12
Correct answer is option 'C'. Can you explain this answer?
The number of P – O bonds and lone pairs of electron present in P4O6 molecule respectively
a)
12 and 4
b)
8 and 8
c)
12 and 16
d)
12 and 12
 | Sushil Kumar answered |
Number of P – O bonds = 12
Number of pair of electron = 16
Number of pair of electron = 16
In the case of alkali metals, the covalent character decreases in the order: [2009]- a)MF > MCl > MBr > MI
- b)MF > MCl > MI > MBr
- c)MI > MBr > MCl > MF
- d)MCl > MI > MBr > MF
Correct answer is option 'C'. Can you explain this answer?
In the case of alkali metals, the covalent character decreases in the order: [2009]
a)
MF > MCl > MBr > MI
b)
MF > MCl > MI > MBr
c)
MI > MBr > MCl > MF
d)
MCl > MI > MBr > MF
 | Ruchi Chopra answered |
MI > MBr > MCl > MF. As the size of the anion decreases covalency decreases.
When chlorine reacts with hot, cone. NaOH, the products formed are- a)NaCI
- b)NaOCI
- c)NaCIO3
- d)HCI
Correct answer is option 'A,C'. Can you explain this answer?
When chlorine reacts with hot, cone. NaOH, the products formed are
a)
NaCI
b)
NaOCI
c)
NaCIO3
d)
HCI
 | Anupama Nair answered |
When Cl2 reacts with hot and concentrated NaOH, then....6NaOH+3Cl2→5NaCl +NaClO3+3H2O...When Cl2 reacts with cold and dilute NaOH then ...2NaOH+Cl2→NaCl+NaOCl+H2O
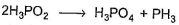 in this equivalent weight of the acid in the reactant side is obtained by dividing molecular weight with
in this equivalent weight of the acid in the reactant side is obtained by dividing molecular weight with
Correct answer is '2'. Can you explain this answer?
| | Abc Bcd answered |
Because there are two molecules of acid in balanced equation.
The total number of positive oxidation states shown by fluorine is
Correct answer is '1'. Can you explain this answer?
The total number of positive oxidation states shown by fluorine is
| | Puja Gupta answered |
Fluorine is an element that belongs to the halogen group in the periodic table. It has an atomic number of 9, and its electron configuration is 1s^2 2s^2 2p^5. Fluorine is highly electronegative, meaning it has a strong tendency to attract electrons towards itself when it forms chemical bonds. This property is due to its relatively small atomic size and high effective nuclear charge.
Fluorine has a total of 7 valence electrons, which are electrons in its outermost energy level (2s^2 2p^5). In order to achieve a stable electron configuration, fluorine tends to gain one electron to complete its octet. By gaining one electron, fluorine achieves a stable electron configuration similar to the nearest noble gas, neon (1s^2 2s^2 2p^6).
Fluorine's strong electronegativity and its tendency to gain electrons result in it having only one common oxidation state, which is -1. In this oxidation state, fluorine gains one electron to achieve a stable configuration of 1s^2 2s^2 2p^6. This oxidation state is commonly observed in compounds where fluorine acts as an anion, such as in the compound sodium fluoride (NaF). In NaF, fluorine gains an electron from sodium to form the F- ion.
It is important to note that although fluorine is highly electronegative and tends to gain electrons, it does not have the capability to lose electrons easily and form positive oxidation states. This is because fluorine's valence shell is almost full, and losing electrons would require a significant amount of energy.
In conclusion, fluorine has only one common oxidation state, which is -1. This is due to its strong electronegativity and its tendency to gain one electron to achieve a stable electron configuration.
Fluorine has a total of 7 valence electrons, which are electrons in its outermost energy level (2s^2 2p^5). In order to achieve a stable electron configuration, fluorine tends to gain one electron to complete its octet. By gaining one electron, fluorine achieves a stable electron configuration similar to the nearest noble gas, neon (1s^2 2s^2 2p^6).
Fluorine's strong electronegativity and its tendency to gain electrons result in it having only one common oxidation state, which is -1. In this oxidation state, fluorine gains one electron to achieve a stable configuration of 1s^2 2s^2 2p^6. This oxidation state is commonly observed in compounds where fluorine acts as an anion, such as in the compound sodium fluoride (NaF). In NaF, fluorine gains an electron from sodium to form the F- ion.
It is important to note that although fluorine is highly electronegative and tends to gain electrons, it does not have the capability to lose electrons easily and form positive oxidation states. This is because fluorine's valence shell is almost full, and losing electrons would require a significant amount of energy.
In conclusion, fluorine has only one common oxidation state, which is -1. This is due to its strong electronegativity and its tendency to gain one electron to achieve a stable electron configuration.
Which of the following is the strongest Lewis base?- a)NBr3
- b)NF3
- c)NCl3
- d)NI3
Correct answer is option 'D'. Can you explain this answer?
Which of the following is the strongest Lewis base?
a)
NBr3
b)
NF3
c)
NCl3
d)
NI3
| | Shanaya Choudhary answered |
Correct Answer :- D
- Lewis bases need to be able to donate electrons. Fluorine is the most electronegative element in the halogens followed by chlorine, bromine and iodine.
- Due to fluorine being strongly electronegative, it draws the electron density towards itself which makes it difficult for nitrogen atom to donate its lone pair of electrons. So, NF is the least basic. This trend follows the strength of electronegativity of the halides.
- Since iodine is least electronegative, it is the most basic trihalide of nitrogen.
So, we have the trend, in decreasing order of basic strength:
NF3 < NCl3 < NBr3 < NI3
Among the following which is the strongest oxidising agent? [2009]- a)Br2
- b)I2
- c)Cl2
- d)F2
Correct answer is option 'D'. Can you explain this answer?
Among the following which is the strongest oxidising agent? [2009]
a)
Br2
b)
I2
c)
Cl2
d)
F2
 | Rajesh Datta answered |
Standard reduction potential of halogens are positive and decreases from fluorine to iodine. Therefore halogens act as strong oxidising agent and their oxidising power decreases from fluorine to iodine.
Direction (Q. Nos. 16 and 17) This section contains a paragraph, each describing theory, experiments, data, etc. Two questions related to the paragraphs have been given. Each question has only one correct answer among the four given options (a), (b), (c) and (d).PassageSulphur and the rest of the elements of 16th group are less electronegative than oxygen. They can acquire ns2np6 by sharing two electrons with the atoms of other elements and thus, exhibit +2 oxidation state in their compounds, in addition to this, atoms have vacant d-orbitals in their valence shell due to which electrons I can be promoted from the s- and p-orbitals of the same shell. As a result, they can show +4 and +6 oxidation states. The oxidation states of elements never exceed +6.Q.Oxidation states of sulphur in H2S2O7 and H2S2O8 respectively are- a)+ 6 and + 7
- b)+ 6 and + 6
- c)+ 5 and + 6
- d)+ 5 and + 7
Correct answer is option 'B'. Can you explain this answer?
Direction (Q. Nos. 16 and 17) This section contains a paragraph, each describing theory, experiments, data, etc. Two questions related to the paragraphs have been given. Each question has only one correct answer among the four given options (a), (b), (c) and (d).
Passage
Sulphur and the rest of the elements of 16th group are less electronegative than oxygen. They can acquire ns2np6 by sharing two electrons with the atoms of other elements and thus, exhibit +2 oxidation state in their compounds, in addition to this, atoms have vacant d-orbitals in their valence shell due to which electrons I can be promoted from the s- and p-orbitals of the same shell. As a result, they can show +4 and +6 oxidation states. The oxidation states of elements never exceed +6.
Q.
Oxidation states of sulphur in H2S2O7 and H2S2O8 respectively are
a)
+ 6 and + 7
b)
+ 6 and + 6
c)
+ 5 and + 6
d)
+ 5 and + 7
| | Om Desai answered |
H2S2O7 has no peroxy bond while H2S2O8 has one peroxy bond.
Which has lowest bond energy (single bond)?- a)O—H
- b)O—O
- c)S—H
- d)S—S
Correct answer is option 'B'. Can you explain this answer?
Which has lowest bond energy (single bond)?
a)
O—H
b)
O—O
c)
S—H
d)
S—S
| | Rahul Bansal answered |
Sulfur atoms are larger than oxygen atoms.
Pi bonds are formed by overlapping of two parallel p orbitals. The further the distance between atoms, the lesser the overlapping and weaker the bond.
But sigma bonds in case of Oxygen and Nitrogen are not strong enough because you are bringing two very small atoms (with large no. of electrons in the outer shell) too close which makes the sigma bond comparatively unstable than that of S-S bond where sigma bond is more stable due to lesser electro static repulsion of non-bonding electrons.
Which of the following statements is not correct about XeF2?- a)It can be obtained by direct reaction between F2 and Xe at high pressure
- b)XeF2 undergoes alkaline hydrolysis to give O2 and Xe
- c)XeF2 is a powerful reducing agent
- d)XeF2 contains two bond pairs and three lone pairs
Correct answer is option 'C'. Can you explain this answer?
Which of the following statements is not correct about XeF2?
a)
It can be obtained by direct reaction between F2 and Xe at high pressure
b)
XeF2 undergoes alkaline hydrolysis to give O2 and Xe
c)
XeF2 is a powerful reducing agent
d)
XeF2 contains two bond pairs and three lone pairs
| | Raghav Bansal answered |
XeF2 is obtained by direct reaction of Xe and F2 at high pressure. It undergoes alkali hydrolysis to produce Xe and O2. XeF2 is a powerful reducing agent where Xe+2 can change to +6 state.
At what temperature white phosphorous changes to red phosphorous?- a)300° C
- b)450° C
- c)50° C
- d)400° C
Correct answer is option 'A'. Can you explain this answer?
At what temperature white phosphorous changes to red phosphorous?
a)
300° C
b)
450° C
c)
50° C
d)
400° C
| | Ananya Singh answered |
According to NCERT, red phosphorus is obtained by heating white phosphorus at 573 K in an inert atmosphere for several days. In degree celius, temperature = 300⁰ C (573K - 273).
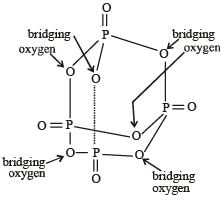
In the all oxyacids of phosphorus, each phosphorus atom is in sp3-hybridised state. All these acids contain P—OH bonds, the hydrogen atom of which are ionisable imparting acidic nature to the compound. The ‘ous’ acids (oxidation state of P is + 1 or + 3) also have P—H bonds in which hydrogens are not ionisable.
The presence of P—H bonds in these acids imparts reducing properties. The structure of some oxyacids are drawn below: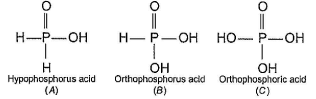
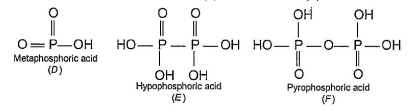 Q. Which of the acids show reducing properties?
Q. Which of the acids show reducing properties?- a)A and C
- b)A and B
- c)A , B and D
- d)C, D, E and F
Correct answer is option 'B'. Can you explain this answer?
In the all oxyacids of phosphorus, each phosphorus atom is in sp3-hybridised state. All these acids contain P—OH bonds, the hydrogen atom of which are ionisable imparting acidic nature to the compound. The ‘ous’ acids (oxidation state of P is + 1 or + 3) also have P—H bonds in which hydrogens are not ionisable.
The presence of P—H bonds in these acids imparts reducing properties. The structure of some oxyacids are drawn below:
The presence of P—H bonds in these acids imparts reducing properties. The structure of some oxyacids are drawn below:
Q.
Which of the acids show reducing properties?
a)
A and C
b)
A and B
c)
A , B and D
d)
C, D, E and F
 | Kirti Choudhary answered |
Reducing property is due to P...H bond
Harmful UV radiations emitted from the sun are prevented from reaching the earth by the presence of ozone in the- a)mesosphere
- b)thermosphere
- c)stratosphere
- d)troposphere
Correct answer is option 'C'. Can you explain this answer?
Harmful UV radiations emitted from the sun are prevented from reaching the earth by the presence of ozone in the
a)
mesosphere
b)
thermosphere
c)
stratosphere
d)
troposphere
 | Pooja Pillai answered |
Stratosphere layer strongly absorb harmful UV-radiations (λ. 255 nm).
Ozone can be detected by using- a)Silver
- b)Sodium
- c)Mercury
- d)None of these
Correct answer is option 'C'. Can you explain this answer?
Ozone can be detected by using
a)
Silver
b)
Sodium
c)
Mercury
d)
None of these
 | Aarya Dasgupta answered |
Ozone is detected by using Hg.
Which of the following is the most basic oxide?- a)Sb2O3
- b)Bi2O3 [2006]
- c)SeO2
- d)Al2O3
Correct answer is option 'B'. Can you explain this answer?
Which of the following is the most basic oxide?
a)
Sb2O3
b)
Bi2O3 [2006]
c)
SeO2
d)
Al2O3
 | Nilotpal Gupta answered |
More the oxidation state of the central atom (metal) more is its acidity. Hence SeO2 (O. S. of Se = +4) is acidic. Further for a given O.S., the basic character of the oxides increases with the increasing size of the central atom.
Thus Al2O3 and Sb2O3 are amphoteric and Bi2O3 is basic.
Thus Al2O3 and Sb2O3 are amphoteric and Bi2O3 is basic.
Which of the following is not a use of noble gases?- a)Argon is widely used for filling incandescent electric bulbs
- b)Neon is used in safety devices for protecting electrical instruments
- c)Radon is used in radiotherapy of cancer
- d)Helium is filled in tubes of cycles and scooters tyres
Correct answer is option 'D'. Can you explain this answer?
Which of the following is not a use of noble gases?
a)
Argon is widely used for filling incandescent electric bulbs
b)
Neon is used in safety devices for protecting electrical instruments
c)
Radon is used in radiotherapy of cancer
d)
Helium is filled in tubes of cycles and scooters tyres
| | Arindam Chaudhary answered |
Helium is filled in tubes of cycles and scooters tyres
Helium is not used for filling tubes of cycles and scooters tyres. It is primarily used in other applications such as:
Argon:
- Widely used for filling incandescent electric bulbs to prevent the filament from oxidizing.
- Used in welding and metal fabrication to shield the weld area from atmospheric gases.
Neon:
- Used in safety devices for protecting electrical instruments due to its ability to produce a bright light when an electric current passes through it.
- Also commonly used in neon signs for advertising.
Radon:
- Used in radiotherapy of cancer as a radiation source to destroy cancer cells.
- It can also be used in some geological research applications to trace the movement of underground gases.
In contrast, helium is typically used in applications such as:
- Cryogenics to achieve low temperatures
- In filling balloons for various purposes
- In cooling nuclear reactors and MRI machines
Therefore, while noble gases have a variety of important uses, filling tubes of cycles and scooters tyres with helium is not one of them.
Helium is not used for filling tubes of cycles and scooters tyres. It is primarily used in other applications such as:
Argon:
- Widely used for filling incandescent electric bulbs to prevent the filament from oxidizing.
- Used in welding and metal fabrication to shield the weld area from atmospheric gases.
Neon:
- Used in safety devices for protecting electrical instruments due to its ability to produce a bright light when an electric current passes through it.
- Also commonly used in neon signs for advertising.
Radon:
- Used in radiotherapy of cancer as a radiation source to destroy cancer cells.
- It can also be used in some geological research applications to trace the movement of underground gases.
In contrast, helium is typically used in applications such as:
- Cryogenics to achieve low temperatures
- In filling balloons for various purposes
- In cooling nuclear reactors and MRI machines
Therefore, while noble gases have a variety of important uses, filling tubes of cycles and scooters tyres with helium is not one of them.
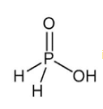 is the structure of
is the structure of- a)Phosphorous acid
- b)Hypophosphorus Acid
- c)Phosphoric acid
- d)Pyrophosphoric acid
Correct answer is option 'B'. Can you explain this answer?
a)
Phosphorous acid
b)
Hypophosphorus Acid
c)
Phosphoric acid
d)
Pyrophosphoric acid
| | Baby Ghosh answered |
Of course..this is a structure of hypo phosphorus acid. It is a mineral acid with formula H4P2O6.In hypophosphorus acid,the phosphorus bonds are identical and joined with p-p bond .there is also joined oxygen and hydrogen bonds as the structure follows.
Most metal oxides are- a)Covalent in nature
- b)Acidic in nature
- c)Basic in nature
- d)None of these
Correct answer is option 'C'. Can you explain this answer?
Most metal oxides are
a)
Covalent in nature
b)
Acidic in nature
c)
Basic in nature
d)
None of these
 | Rashi Bose answered |
Most metal oxides are ionic crystals with high melting and boiling points. They are compounds that contain at least one metal and one oxygen atom, and are generally basic in nature. Metal oxides are basic because they react with acids to form salt and water, neutralizing the acids.
Which one of the following arrangements does not truly represent the property indicated against it?[2000]- a)Br2 < Cl2 < F2 : Electronegativity
- b)Br2 < F2 < Cl2 : Electron affinity
- c)Br2 < Cl2 < F2 : Bond energy
- d)Br2 < Cl2 < F2 : Oxidising power
Correct answer is option 'C'. Can you explain this answer?
Which one of the following arrangements does not truly represent the property indicated against it?[2000]
a)
Br2 < Cl2 < F2 : Electronegativity
b)
Br2 < F2 < Cl2 : Electron affinity
c)
Br2 < Cl2 < F2 : Bond energy
d)
Br2 < Cl2 < F2 : Oxidising power
 | Krish Khanna answered |
The bond energy of F2 < Cl2 due to more repulsion in between non-bonding electrons (2p) of F2 in comparision to non- bonding pair (3p) repulsion in Cl2, the bond energy of F2 < Cl2.
Bond energy (kJ mol–1) :
Bond energy (kJ mol–1) :

Solid iodine is an example of- a)covalent solid
- b)ionic solid
- c)molecular solid
- d)metallic solid
Correct answer is option 'C'. Can you explain this answer?
Solid iodine is an example of
a)
covalent solid
b)
ionic solid
c)
molecular solid
d)
metallic solid
 | Vaishnavi Dasgupta answered |
Solid iodine is a molecular solid.
When Cl2 gas reacts with hot and concentrated sodium hydroxide solution, the oxidation number of chlorine changes from- a)zero to +1 and zero to -5
- b)zero to -1 and zero to +5
- c)zero to -1 and zero to +5
- d)zero to +1 and zero to -3
Correct answer is option 'B'. Can you explain this answer?
When Cl2 gas reacts with hot and concentrated sodium hydroxide solution, the oxidation number of chlorine changes from
a)
zero to +1 and zero to -5
b)
zero to -1 and zero to +5
c)
zero to -1 and zero to +5
d)
zero to +1 and zero to -3
 | Shail Chakraborty answered |
When chlorine gas reacts with hot and concentrated NaOH solution, it disproportionates into Chloride (Cl-) and Chlorate (ClO3-) ions.
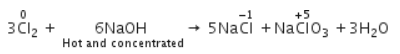
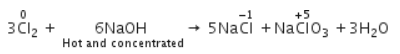
Direction (Q. No. 25) This section is based on Statement I and Statement II. Select the correct answer from the codes given below.Q. Statement I : All halogens are coloured.Statement II : Halogens absorbs part of the light in the visible region.- a)Both Statement I and Statement II are correct and Statement II is the correct explanation of Statement I
- b)Both Statement I and Statement II are correct but Statement II is not the correct explanation of Statement I
- c)Statement I is correct but Statement II is incorrect
- d)Statement II is correct but Statement I is incorrect
Correct answer is option 'A'. Can you explain this answer?
Direction (Q. No. 25) This section is based on Statement I and Statement II. Select the correct answer from the codes given below.
Q.
Statement I : All halogens are coloured.
Statement II : Halogens absorbs part of the light in the visible region.
a)
Both Statement I and Statement II are correct and Statement II is the correct explanation of Statement I
b)
Both Statement I and Statement II are correct but Statement II is not the correct explanation of Statement I
c)
Statement I is correct but Statement II is incorrect
d)
Statement II is correct but Statement I is incorrect
 | Gauri Kaur answered |
Halogen molecule absorbs light in the visible region as a result of which their electrons are excited to higher energy level while the remaining light is transmitted.
The colour of halogens is actually the colour of this transmitted light, i.e. halogens have complementary colours.
The colour of halogens is actually the colour of this transmitted light, i.e. halogens have complementary colours.
Direction (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.Q. The fluoride ores are- a)carnalite
- b)cryolite
- c)fluorspar
- d)chile saltpetre
Correct answer is option 'B,C'. Can you explain this answer?
Direction (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q.
The fluoride ores are
a)
carnalite
b)
cryolite
c)
fluorspar
d)
chile saltpetre
 | Gauri Kaur answered |
The cryolite (Na3AIF6) and fluorspar (CaF2) are two ores of fluoride.
Number of moles of NaOH needed to neutralise one mole each of H3PO2, H3PO3 and H3PO4 respectively are- a)3, 3 and 3
- b)1, 2 and 3
- c)2, 2 and 3
- d)2, 1 and 3
Correct answer is option 'B'. Can you explain this answer?
Number of moles of NaOH needed to neutralise one mole each of H3PO2, H3PO3 and H3PO4 respectively are
a)
3, 3 and 3
b)
1, 2 and 3
c)
2, 2 and 3
d)
2, 1 and 3
 | Gauri Sharma answered |
H3PO2, H3PO3 and H3PO4 are mono, di and tribasic acids respectively.
Which is the correct order of basic strength ?- a) NH2OH < N2H4 < NH3
- b) N2H4 < NH2OH < NH3
- c) NH3 < N2H4 < NH2OH
- d) NH2OH < NH3 < N2H4
Correct answer is option 'A'. Can you explain this answer?
Which is the correct order of basic strength ?
a)
NH2OH < N2H4 < NH3
b)
N2H4 < NH2OH < NH3
c)
NH3 < N2H4 < NH2OH
d)
NH2OH < NH3 < N2H4
 | Ashish Nambiar answered |
NH2OH and NH2—NH2 may be considered as NH3 derivatives in which H is replaced but — OH and NH2 respectively. Due to their electron withdrawing nature, these groups decreases electron density over nitrogen making them less basic. The effect of — OH group is stronger than —NH2
The formula for calcium chlorite is [1994]- a)Ca(ClO4)2
- b)Ca(ClO3)2
- c)CaClO2
- d)Ca(ClO2)2
Correct answer is option 'D'. Can you explain this answer?
The formula for calcium chlorite is [1994]
a)
Ca(ClO4)2
b)
Ca(ClO3)2
c)
CaClO2
d)
Ca(ClO2)2
| | Shraddha Choudhury answered |
The formula for calcium chlorite is Ca(ClO2)2. Let's break down the components of this compound to understand why this is the correct formula.
Calcium:
- Calcium is a metal with a +2 charge. It is represented by the symbol Ca in the periodic table.
Chlorite:
- Chlorite is a polyatomic ion with the formula ClO2-. It consists of one chlorine atom (Cl) bonded to two oxygen atoms (O), and it carries a -1 charge.
Formation of calcium chlorite:
- When calcium reacts with chlorite, the calcium ion (Ca2+) combines with two chlorite ions (ClO2-) to form calcium chlorite (Ca(ClO2)2).
- The calcium ion has a +2 charge, while each chlorite ion has a -1 charge. To balance the charges, two chlorite ions are needed for every calcium ion.
Explanation of options:
a) Ca(ClO4)2:
- This formula represents calcium perchlorate, not calcium chlorite. Perchlorate is a different polyatomic ion that consists of one chlorine atom and four oxygen atoms.
b) Ca(ClO3)2:
- This formula represents calcium chlorate, not calcium chlorite. Chlorate is another polyatomic ion that consists of one chlorine atom and three oxygen atoms.
c) CaClO2:
- This formula represents calcium hypochlorite, not calcium chlorite. Hypochlorite is a different polyatomic ion that consists of one chlorine atom and two oxygen atoms.
d) Ca(ClO2)2:
- This formula represents calcium chlorite, which is the correct compound. It consists of one calcium ion (Ca2+) bonded to two chlorite ions (ClO2-).
In summary, the correct formula for calcium chlorite is Ca(ClO2)2, as it consists of one calcium ion bonded to two chlorite ions.
Calcium:
- Calcium is a metal with a +2 charge. It is represented by the symbol Ca in the periodic table.
Chlorite:
- Chlorite is a polyatomic ion with the formula ClO2-. It consists of one chlorine atom (Cl) bonded to two oxygen atoms (O), and it carries a -1 charge.
Formation of calcium chlorite:
- When calcium reacts with chlorite, the calcium ion (Ca2+) combines with two chlorite ions (ClO2-) to form calcium chlorite (Ca(ClO2)2).
- The calcium ion has a +2 charge, while each chlorite ion has a -1 charge. To balance the charges, two chlorite ions are needed for every calcium ion.
Explanation of options:
a) Ca(ClO4)2:
- This formula represents calcium perchlorate, not calcium chlorite. Perchlorate is a different polyatomic ion that consists of one chlorine atom and four oxygen atoms.
b) Ca(ClO3)2:
- This formula represents calcium chlorate, not calcium chlorite. Chlorate is another polyatomic ion that consists of one chlorine atom and three oxygen atoms.
c) CaClO2:
- This formula represents calcium hypochlorite, not calcium chlorite. Hypochlorite is a different polyatomic ion that consists of one chlorine atom and two oxygen atoms.
d) Ca(ClO2)2:
- This formula represents calcium chlorite, which is the correct compound. It consists of one calcium ion (Ca2+) bonded to two chlorite ions (ClO2-).
In summary, the correct formula for calcium chlorite is Ca(ClO2)2, as it consists of one calcium ion bonded to two chlorite ions.
Noble gases do not react with other elements because [1994]- a)They are mono atomic
- b)They are found in abundance
- c)The size of their atoms is very small
- d)They are completely paired up and stable electron shells
Correct answer is option 'D'. Can you explain this answer?
Noble gases do not react with other elements because [1994]
a)
They are mono atomic
b)
They are found in abundance
c)
The size of their atoms is very small
d)
They are completely paired up and stable electron shells
 | Abhishek Desai answered |
On account of highly stable ns2 np6 configuration in the valence shell. These elements have no tendency either to lose gain or share electrons with atoms of other elements i.e., their combining capacity or valency is zero. Further all the orbitals in the atoms of these elements are doubly occupied i.e electrons are not available for sharing.
The correct order of thermal stability of the hydrides of group 16 elements is- a)H2Po> H2Fe> H2Se> H2S > H2O
- b)H2O<H2S>H2Se>H2Te>H2Po
- c)H2O=H2S=H2Se-H2Te=H2Po
- d)H2O>H2S>H2SE>H2Te>H2Po
Correct answer is option 'D'. Can you explain this answer?
The correct order of thermal stability of the hydrides of group 16 elements is
a)
H2Po> H2Fe> H2Se> H2S > H2O
b)
H2O<H2S>H2Se>H2Te>H2Po
c)
H2O=H2S=H2Se-H2Te=H2Po
d)
H2O>H2S>H2SE>H2Te>H2Po
 | Preethi Kaur answered |
The correct order of thermal stability of the hydrides of group 16 elements is:
a) H2O > H2S > H2Se > H2Te > H2Po
a) H2O > H2S > H2Se > H2Te > H2Po
Which one of the following arrangements does not give the correct picture of the trends indicated against it ? [2008]- a)F2 > Cl2 > Br2 > I2 : Oxidizing power
- b)F2 > Cl2 > Br2 > I2 : Electron gain enthalpy
- c)F2 > Cl2 > Br2 > I2 : Bond dissociation energy
- d)F2 > Cl2 > Br2 > I2 : Electronegativity.
Correct answer is option 'B,C'. Can you explain this answer?
Which one of the following arrangements does not give the correct picture of the trends indicated against it ? [2008]
a)
F2 > Cl2 > Br2 > I2 : Oxidizing power
b)
F2 > Cl2 > Br2 > I2 : Electron gain enthalpy
c)
F2 > Cl2 > Br2 > I2 : Bond dissociation energy
d)
F2 > Cl2 > Br2 > I2 : Electronegativity.
| | Nisha Pillai answered |
From the given options we find option (a) is correct. The oxidising power of halogens follow the order F2 > Cl2 > Br2 > I2 . Option (b) is incorrect because it in not the correct order of electron gain enthalpy of halogens. The correct order is Cl2 > F2 > Br2 > I2 . The low value of F2 than Cl2 is due to its small size. Option (c) is incorrect. The correct order of bond dissociation energies of halogens is Cl2 > Br2 > F2 > I2 . Option (d) is correct. It is the correct order of electronegativity values of halogens. Thus option (b) and (c) are incorrect.
Direction (Q. No. 20) This section is bassed on Statment I and Statment II. II. Select the correct answer from the codes given below.Q. Statement I : NF3 is a weaker ligand than N(CH3)3Statement II : NF3 ionises to give F- ions in aqueous solutio- a)Both Statement I and Statement II are correct and Statement II is the correct explanation of Statemen I
- b)Both Statement I and Statement II are correct but Statement II is not the correct. explanation of Statment I
- c)Statement I is correct but Statement II is incorrect
- d)Statement II is correct but Statement I is incorrect
Correct answer is option 'B'. Can you explain this answer?
Direction (Q. No. 20) This section is bassed on Statment I and Statment II. II. Select the correct answer from the codes given below.
Q.
Statement I : NF3 is a weaker ligand than N(CH3)3
Statement II : NF3 ionises to give F- ions in aqueous solutio
a)
Both Statement I and Statement II are correct and Statement II is the correct explanation of Statemen I
b)
Both Statement I and Statement II are correct but Statement II is not the correct. explanation of Statment I
c)
Statement I is correct but Statement II is incorrect
d)
Statement II is correct but Statement I is incorrect
 | Rashi Bose answered |
Due to e- withdrawing capacity of fluorideion, it withdraws. from nitrogen in NF3 make it weakerlig and while presence of e- donating methyl group makes the nitrogen in N(CH3)3 a strong ligand. In aqueous medium, NF3 furnishes fluorideion.
Which of the following statements is not correct?- a)Helium has the lowest boiling point among the noble gases
- b)Argon is used in electric bulbs
- c)Krypton is obtained during radioactive disintegration
- d)Xe forms XeF6
Correct answer is option 'C'. Can you explain this answer?
Which of the following statements is not correct?
a)
Helium has the lowest boiling point among the noble gases
b)
Argon is used in electric bulbs
c)
Krypton is obtained during radioactive disintegration
d)
Xe forms XeF6
| | Swati Bose answered |
Explanation:
The correct statement is option 'C': Krypton is obtained during radioactive disintegration.
Explanation:
- Statement a: Helium has the lowest boiling point among the noble gases.
- This statement is correct. Helium has the lowest boiling point (-268.93°C) among all the noble gases. It is a colorless and odorless gas that remains in a gaseous state at room temperature and pressure.
- Statement b: Argon is used in electric bulbs.
- This statement is correct. Argon is commonly used in electric bulbs to maintain an inert atmosphere and prevent oxidation of the filament. It is an abundant noble gas and is chemically inert.
- Statement c: Krypton is obtained during radioactive disintegration.
- This statement is incorrect. Krypton is not obtained during radioactive disintegration. Krypton (Kr) is a noble gas that is chemically unreactive and does not undergo radioactive disintegration. It is obtained through the fractional distillation of liquid air.
- Statement d: Xe forms XeF6.
- This statement is correct. Xenon (Xe) can form compounds, and one of them is xenon hexafluoride (XeF6). XeF6 is a strong oxidizing agent and is used in chemical reactions and synthesis.
Therefore, the correct statement is option 'C': Krypton is obtained during radioactive disintegration. Krypton is not obtained through radioactive disintegration, but rather through fractional distillation of liquid air.
The correct statement is option 'C': Krypton is obtained during radioactive disintegration.
Explanation:
- Statement a: Helium has the lowest boiling point among the noble gases.
- This statement is correct. Helium has the lowest boiling point (-268.93°C) among all the noble gases. It is a colorless and odorless gas that remains in a gaseous state at room temperature and pressure.
- Statement b: Argon is used in electric bulbs.
- This statement is correct. Argon is commonly used in electric bulbs to maintain an inert atmosphere and prevent oxidation of the filament. It is an abundant noble gas and is chemically inert.
- Statement c: Krypton is obtained during radioactive disintegration.
- This statement is incorrect. Krypton is not obtained during radioactive disintegration. Krypton (Kr) is a noble gas that is chemically unreactive and does not undergo radioactive disintegration. It is obtained through the fractional distillation of liquid air.
- Statement d: Xe forms XeF6.
- This statement is correct. Xenon (Xe) can form compounds, and one of them is xenon hexafluoride (XeF6). XeF6 is a strong oxidizing agent and is used in chemical reactions and synthesis.
Therefore, the correct statement is option 'C': Krypton is obtained during radioactive disintegration. Krypton is not obtained through radioactive disintegration, but rather through fractional distillation of liquid air.
Which is incorrect among the following options?- a)Acidic Character of H3PO4 > H3PO3 > H3PO2
- b)Oxidation state of nitrogen N2O < NO < N2O3 < N2O5
- c) Basicity NH3 > PH3 > AsH3 > SbH3
- d)Boiling point of SbH3 > AsH3 > NH3 > PH3
Correct answer is option 'A'. Can you explain this answer?
Which is incorrect among the following options?
a)
Acidic Character of H3PO4 > H3PO3 > H3PO2
b)
Oxidation state of nitrogen N2O < NO < N2O3 < N2O5
c)
Basicity NH3 > PH3 > AsH3 > SbH3
d)
Boiling point of SbH3 > AsH3 > NH3 > PH3
 | Saptarshi Ghoshal answered |
Acidic Character of P oxyacids is due to P—H bonds in the acid. Correct reducing property order is
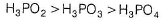
When carnallite is dissolved in water the number of ions formed are
Correct answer is '5'. Can you explain this answer?
When carnallite is dissolved in water the number of ions formed are
 | Vaishnavi Dasgupta answered |
Carnallite is a double salt. It will dissociate into simple substances or ions completely when dissolved in water.
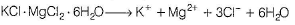
Hence, 5 ions are produced.
Hence, 5 ions are produced.
Direction (Q. Nos. 1-10) This section contains 10 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.Q. Which of the following property decreases down the group in the halogens?- a)Electropositive nature
- b)Density
- c)Boiling point
- d)Ionisation enthalp
Correct answer is option 'D'. Can you explain this answer?
Direction (Q. Nos. 1-10) This section contains 10 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q.
Which of the following property decreases down the group in the halogens?
a)
Electropositive nature
b)
Density
c)
Boiling point
d)
Ionisation enthalp
 | Amar Jain answered |
Due to increase in atomic size, ionisation enthalpy decreases down the group.
Chapter doubts & questions for p-Block Elements (Only in NEET) - Chemistry Class 12 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of p-Block Elements (Only in NEET) - Chemistry Class 12 in English & Hindi are available as part of NEET exam. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
;
Signup to see your scores go up
within 7 days!
within 7 days!
Takes less than 10 seconds to signup