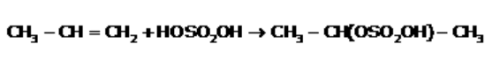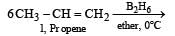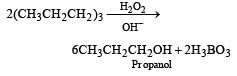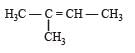All Exams > NEET > Topic-wise MCQ Tests for NEET > All Questions
All questions of Hydrocarbons for NEET Exam
What is the major bromination product in the following reaction? 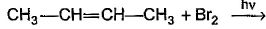
- a)
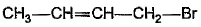
- b)
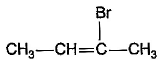
- c)
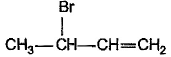
- d)
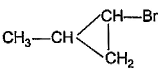
Correct answer is option 'C'. Can you explain this answer?
What is the major bromination product in the following reaction?
a)
b)
c)
d)
 | Mohit Rajpoot answered |
The correct answer is option c
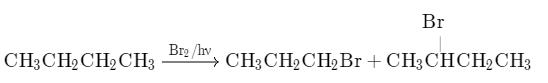
Thus, a racemic mixture is obtained. A racemic mixture is one that has an equal amount of left and right handed enantiomers of a chiral molecule.
Thus, a racemic mixture is obtained. A racemic mixture is one that has an equal amount of left and right handed enantiomers of a chiral molecule.
Organic compounds can be classified even based upon the function groups. Identify the one which is not a functional group- a)Isocyanide
- b)Isocyano
- c)Carboxyl
- d)Carbonyl
Correct answer is option 'A'. Can you explain this answer?
Organic compounds can be classified even based upon the function groups. Identify the one which is not a functional group
a)
Isocyanide
b)
Isocyano
c)
Carboxyl
d)
Carbonyl
| | Krishna Iyer answered |
Isocyanide is a compound and it is not a functional group.
Ethene and ethyne can be distinguished by:- a)Bromine water
- b)KMnO4 solution
- c)Ammoniacal Cuprous chloride solution
- d)Any of the above
Correct answer is option 'C'. Can you explain this answer?
Ethene and ethyne can be distinguished by:
a)
Bromine water
b)
KMnO4 solution
c)
Ammoniacal Cuprous chloride solution
d)
Any of the above
| | Nandini Patel answered |
The two hydrocarbons can be easily distinguished by simple chemical tests, as ethyne molecule is supposed to have acidic hydrogen.
1. When ethyne is bubbled through ammoniacal silver nitrate solution , a yellow-white precipitate of silver acetylide would be formed.
C2H2 + 2AgNO3 = Ag2C2 + 2HNO3
2. Similarly, ethyne forms a red precipitate of copper acetylide (Cu2C2) when it is passed through ammoniacal cuprous chloride solution.
Ethene does not react with AgNO3 or Cu2Cl2 solution.
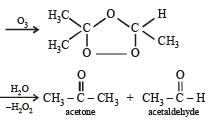
Propene on ozonolysis forms:- a)Acetaldehyde
- b)Formaldehyde
- c)Both acetaldehyde and formaldehyde
- d)Acetone
Correct answer is option 'C'. Can you explain this answer?
Propene on ozonolysis forms:
a)
Acetaldehyde
b)
Formaldehyde
c)
Both acetaldehyde and formaldehyde
d)
Acetone
| | Raghav Bansal answered |
When propene on ozonolysis it yields a new structure called ozonide
and there cleavage takes place and it yields two products namely
1.acetaldehyde
2.formaldehyde
and there cleavage takes place and it yields two products namely
1.acetaldehyde
2.formaldehyde
Direction (Q. Nos. 16 - 18) This section contains a paragraph, describing theory, experiments, data, etc.
Three questions related to the paragraph have been given. Each question has only one correct answer among the four given options (a), (b), (cj and (d).PassageA hydrocarbon with molecular formula C10H18, upon catalytic hydrogenation gives C10H20 (X). X on free radical chlorination gives two monochloro derivatives with their molecular formula C10H19CI that are constitutional isomers.Q. Which of the following satisfy the criteria of X ?- a)
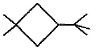
- b)

- c)

- d)
Correct answer is option 'C'. Can you explain this answer?
Direction (Q. Nos. 16 - 18) This section contains a paragraph, describing theory, experiments, data, etc.
Three questions related to the paragraph have been given. Each question has only one correct answer among the four given options (a), (b), (cj and (d).
Three questions related to the paragraph have been given. Each question has only one correct answer among the four given options (a), (b), (cj and (d).
Passage
A hydrocarbon with molecular formula C10H18, upon catalytic hydrogenation gives C10H20 (X). X on free radical chlorination gives two monochloro derivatives with their molecular formula C10H19CI that are constitutional isomers.
Q. Which of the following satisfy the criteria of X ?
a)
b)
c)
d)
 | EduRev JEE answered |
In option c, there are only two different position which can be chlorinated,
So, option c is correct.
So, option c is correct.
Ethylene reacts with HBr to give:- a)Acetylene
- b)Ethyl alcohol
- c)Acetaldehyde
- d)Ethyl bromide
Correct answer is option 'D'. Can you explain this answer?
Ethylene reacts with HBr to give:
a)
Acetylene
b)
Ethyl alcohol
c)
Acetaldehyde
d)
Ethyl bromide
| | Preeti Iyer answered |
Ethylene reacts with HBr to form Ethyl bromide. The reaction propagates as follow:-
H2C=CH2 + HBr → H2C+-CH3 →H2BrC-CH3
Since π cloud is electron rich, so HBr dissociates into H+ and Br-. H+ attacks on alkene to give a carbocation and then Br- attacks to get ethyl bromide.
H2C=CH2 + HBr → H2C+-CH3 →H2BrC-CH3
Since π cloud is electron rich, so HBr dissociates into H+ and Br-. H+ attacks on alkene to give a carbocation and then Br- attacks to get ethyl bromide.
Direction (Q. Nos. 12 -15) This section contains 4 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.Q. In the following free radical bromination reaction, the im portant product(s) is/are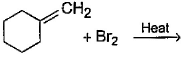
- a)
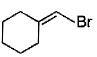
- b)
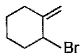
- c)
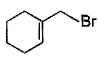
- d)
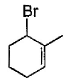
Correct answer is option 'B,C'. Can you explain this answer?
Direction (Q. Nos. 12 -15) This section contains 4 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q. In the following free radical bromination reaction, the im portant product(s) is/are
a)
b)
c)
d)
 | Knowledge Hub answered |
This is the case of allylic substitution. The radical will delocalise itself to two locations. And so, we will get two different positions for radical substitution.

There is also a possibility at C6, but it is not in our option.

There is also a possibility at C6, but it is not in our option.
Cis isomer have:- a)High boiling point than trans isomer
- b)Lower boiling point than trans isomer
- c)Same boiling point
- d)None of the above
Correct answer is option 'A'. Can you explain this answer?
Cis isomer have:
a)
High boiling point than trans isomer
b)
Lower boiling point than trans isomer
c)
Same boiling point
d)
None of the above
| | Rajat Kapoor answered |
The cis isomer in this case has a boiling point of 60.3 degC, while the trans isomer has a boiling point of 47.5 degC. In the cis isomer the two polar C-Cl bond dipole moments combine to give an overall molecular dipole, so that there are intermolecular dipole–dipole forces (or Keesom forces), which add to the London.
Which of the following reactions can bring about chlorination of cyclohexane?- a)
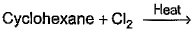
- b)
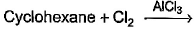
- c)
- d)
Correct answer is option 'A,C'. Can you explain this answer?
Which of the following reactions can bring about chlorination of cyclohexane?
a)
b)
c)
d)
 | Knowledge Hub answered |
For SO2Cl2: The reactivity patterns of SO2Cl2 and SOCl2 are quite different. SOCl2 is a good electrophile, and can be thought of as a source of Cl− ions. These ions can go on to react in their typical nucleophilic fashion. SO2Cl2 however is often a Cl2 source, as it readily decomposes giving off sulfur dioxide. Usually, much easier/safer to use this than measuring out (and getting into solution) chlorine gas. The chlorination of simple alkanes by Cl2 gas (or something that makes it in solution) happens by a radical mechanism i.e. Cl⋅ not Cl
For Cl2 and heat/light:
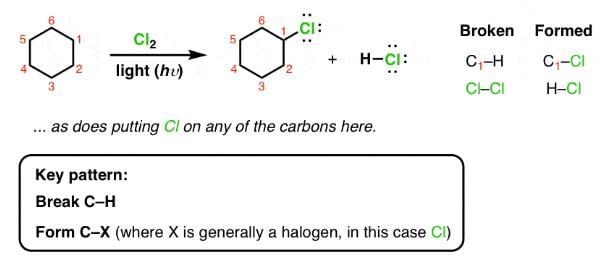
For Cl2 and heat/light:

For Cl with AlCl3: It is used for chlorination of compounds like benzene
For HCl: It is used for halogenations of a double bond.
For HCl: It is used for halogenations of a double bond.
How many monobromo derivatives exists for anthracene?
- a)2
- b)3
- c)4
- d)5
Correct answer is option 'B'. Can you explain this answer?
How many monobromo derivatives exists for anthracene?
a)
2
b)
3
c)
4
d)
5
| | Suresh Iyer answered |
The correct answer is Option B.
There are 3 monobromo derivatives exists for anthracene:
1-Chloroanthracene
2-Chloroanthracene
and 9-Chloroanthracene
1-Chloroanthracene
2-Chloroanthracene
and 9-Chloroanthracene
Which of the following is not a possible termination step in the free radical chlorination of methane?- a)
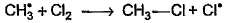
- b)

- c)
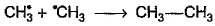
- d)
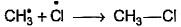
Correct answer is option 'A'. Can you explain this answer?
Which of the following is not a possible termination step in the free radical chlorination of methane?
a)
b)
c)
d)
| | Preeti Iyer answered |
Termination is the last step. So there shouldn't be any free radical atom remaining. In first option there is Cl• remaining it can't be termination step.The steps in free radical halogenation are as
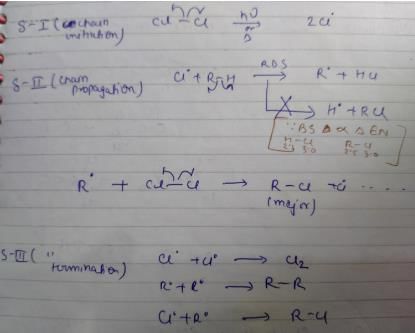
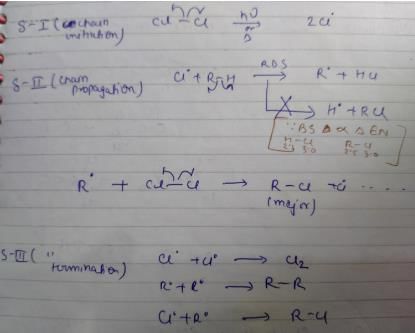
Direction (Q. Nos, 19 - 22) This section contains 4 questions. When worked out will result in an integer from 0 to 9 (both inclusive).Q. How many different isomers of alkenes {including stereoisomers) exist that all upon catalytic hydrogenation adds one mole of H2 to give the same 2, 2, 3,5-tetramethyl hexane?
Correct answer is '7'. Can you explain this answer?
Direction (Q. Nos, 19 - 22) This section contains 4 questions. When worked out will result in an integer from 0 to 9 (both inclusive).
Q. How many different isomers of alkenes {including stereoisomers) exist that all upon catalytic hydrogenation adds one mole of H2 to give the same 2, 2, 3,5-tetramethyl hexane?
 | Infinity Academy answered |
To determine the number of different isomers of alkenes (including stereoisomers) that upon catalytic hydrogenation yield the same 2,2,3,5-tetramethylhexane,
Follow these steps:
1. Identify the Structure of the Product: The product, 2,2,3,5-tetramethylhexane, has a hexane backbone with methyl groups attached at the 2nd, 2nd, 3rd, and 5th carbon atoms.
2. Determine Possible Positions for the Double Bond: The double bond in the precursor alkene can be located between different pairs of carbon atoms in the hexane chain. Possible positions are: - Between C1-C2 - Between C2-C3 - Between C3-C4 - Between C4-C5
3. Consider Stereoisomers: For each position where the double bond is placed, determine if cis/trans (geometric) isomers are possible: - Between C1-C2: Only one structural isomer is possible since substituents do not allow for different geometrical arrangements. - Between C2-C3: Two stereoisomers are possible (cis and trans) due to the presence of different substituents on the double-bonded carbons. - Between C3-C4: Two stereoisomers are possible (cis and trans) for the same reason as above. - Between C4-C5: Only one structural isomer is possible.
4. Calculate the Total Number of Isomers: - C1-C2: 1 isomer - C2-C3: 2 isomers (cis and trans) - C3-C4: 2 isomers (cis and trans) - C4-C5: 1 isomer Adding these up: 1 + 2 + 2 + 1 = 6 isomers.
5. Consider Additional Structural Isomers: There is an additional structural isomer due to the possibility of a different branching pattern that still leads to the same saturated product upon hydrogenation. This increases the total count to 7 isomers. Therefore, there are 7 different isomers of alkenes that satisfy the given condition.
Addition of halogens to alkenes is an example of:- a)Nucleophilic addition reaction
- b)Electrophilic addition reaction
- c)Electrophilic substitution reaction
- d)Nucleophilic substitution reaction
Correct answer is option 'B'. Can you explain this answer?
Addition of halogens to alkenes is an example of:
a)
Nucleophilic addition reaction
b)
Electrophilic addition reaction
c)
Electrophilic substitution reaction
d)
Nucleophilic substitution reaction
 | Avantika Chakraborty answered |
Halogens can act as electrophiles to attack a double bond in alkene. Double bond represents a region of electron density and therefore functions as a nucleophile.
Direction (Q. Nos. 1 - 11) This section contains 11 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.Q. When light is shined on a mixture of chlorine and ethane, chloroethane is formed besides dichloroethane, trichloroethane and several other products. What reaction condition can optimise the yield of chloroethane?- a)Higher reaction temperature
- b)High concentration of chlorine gas
- c)Excess of ethane reactan
- d)Low reaction temperature
Correct answer is option 'C'. Can you explain this answer?
Direction (Q. Nos. 1 - 11) This section contains 11 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Q. When light is shined on a mixture of chlorine and ethane, chloroethane is formed besides dichloroethane, trichloroethane and several other products. What reaction condition can optimise the yield of chloroethane?
a)
Higher reaction temperature
b)
High concentration of chlorine gas
c)
Excess of ethane reactan
d)
Low reaction temperature
| | Pooja Shah answered |
When chlorine and Ethane are taken with chlorine in excess only then we have more than one product like chloroethane, dichloroethane, trichloroethane etc. To avoid this we should take Ethane in excess because when we will take it then in excess then we will have only single time chlorination and we will get monochloroethane.
A hydrocarbon with molecular formula C10H18, upon catalytic hydrogenation gives C10H20 (X). X on free radical chlorination gives two monochloro derivatives with their molecular formula C10H19CI that are constitutional isomers.Q. How many different alkenes on hydrogenation, can gives X ?- a)1
- b)2
- c)3
- d)4
Correct answer is option 'A'. Can you explain this answer?
A hydrocarbon with molecular formula C10H18, upon catalytic hydrogenation gives C10H20 (X). X on free radical chlorination gives two monochloro derivatives with their molecular formula C10H19CI that are constitutional isomers.
Q. How many different alkenes on hydrogenation, can gives X ?
a)
1
b)
2
c)
3
d)
4
| | Gaurav Kumar answered |
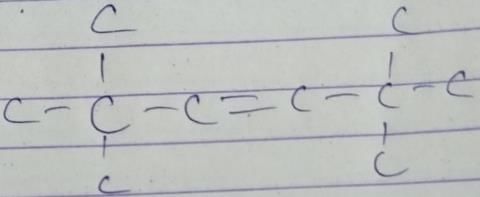
Select the true statement about benzene amongst the following [1992]- a)Because of unsatur a tion benzene easily undergoes addition
- b)There are two types of C – C bon ds in benzene molecule
- c)There is cyclic delocalisation of pi-electrons in benzene
- d)Monosubstitution of benzene gives three isomeric products.
Correct answer is option 'C'. Can you explain this answer?
Select the true statement about benzene amongst the following [1992]
a)
Because of unsatur a tion benzene easily undergoes addition
b)
There are two types of C – C bon ds in benzene molecule
c)
There is cyclic delocalisation of pi-electrons in benzene
d)
Monosubstitution of benzene gives three isomeric products.
 | Anirudh Datta answered |
Benzene do not show addition reaction like other unsaturated hydrocarbons. However it show substitution reactions. Due to resonance all the C – C bonds have the same nature, which is possible because of the cycli c del oca lisati on of π-electr on s in benzene. Monosubstitution will give only a single product.
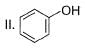
What is the correct order of increasing acidic strength of the following?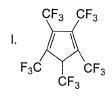
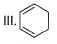
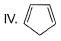
- a)Ill < I < IV < II
- b)Ill < IV < II < I
- c) Ill < II < IV < I
- d)III<IV<I<II
Correct answer is option 'B'. Can you explain this answer?
What is the correct order of increasing acidic strength of the following?




a)
Ill < I < IV < II
b)
Ill < IV < II < I
c)
Ill < II < IV < I
d)
III<IV<I<II
| | Lavanya Menon answered |
Compound I is having the highest acidic strength due to the -I effect of five CF3 substituents.
Compound II is having less acidic strength than I but more than the rest due to the extremely stable conjugate anion formed after deprrotonation.
So, Option B is correct.
Direction (Q. Nos. 21 - 24) This section contains 4 questions. When worked out will result in an integer from 0 to 9 (both inclusive).Q. How many different isomeric alkynes on catalytic hydrogenation can give 2, 3, 4 -trimethyl heptane?
Correct answer is '8'. Can you explain this answer?
Direction (Q. Nos. 21 - 24) This section contains 4 questions. When worked out will result in an integer from 0 to 9 (both inclusive).
Q. How many different isomeric alkynes on catalytic hydrogenation can give 2, 3, 4 -trimethyl heptane?
 | Ashish Mishra answered |
8 is correct.
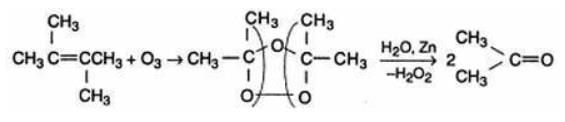
The alkene which on ozonolysis gives only acetone is:- a)(CH3)2C = CH2
- b)CH3CH = CHCH3
- c)CH3CH = C(CH3)2
- d)(CH3)2C = C(CH3)2
Correct answer is option 'D'. Can you explain this answer?
The alkene which on ozonolysis gives only acetone is:
a)
(CH3)2C = CH2
b)
CH3CH = CHCH3
c)
CH3CH = C(CH3)2
d)
(CH3)2C = C(CH3)2
 | Pioneer Academy answered |
The reductive ozaonalysis of 2,3 -Dimethyl-2-butene yields acetone.The reaction is as follows: 2,3 -Dimethyl-2-butene acetone
During free radical bromination of isobutane, an alkene is produced as by product via disproportionation of the intermediate alkyl free radical. What is this alkene?- a)1-butene
- b)2-butene
- c)2-methyl propene
- d)Both ‘b’ and ‘c’
Correct answer is option 'C'. Can you explain this answer?
During free radical bromination of isobutane, an alkene is produced as by product via disproportionation of the intermediate alkyl free radical. What is this alkene?
a)
1-butene
b)
2-butene
c)
2-methyl propene
d)
Both ‘b’ and ‘c’
 | Aquib Anwer answered |
3
Which of the following statem ents regarding free radical halogenation o f alkane is not true?- a)Hydrogen abstraction by halogen radical in the propagation step is exothermic in both chlorination and bromination
- b)Hydrogen abstraction by halogen radical in propagation step is exothermic in chlorination but endothermic in bromination
- c)A single halogen radical may bring about halogenation of thousands of alkane molecules
- d)Continuous source of energy is required to continue the free radical halogenation reaction
Correct answer is option 'A'. Can you explain this answer?
Which of the following statem ents regarding free radical halogenation o f alkane is not true?
a)
Hydrogen abstraction by halogen radical in the propagation step is exothermic in both chlorination and bromination
b)
Hydrogen abstraction by halogen radical in propagation step is exothermic in chlorination but endothermic in bromination
c)
A single halogen radical may bring about halogenation of thousands of alkane molecules
d)
Continuous source of energy is required to continue the free radical halogenation reaction
| | Geetika Shah answered |
Hydrogen abstraction by halogen radical in the propagation step is exothermic in chlorination but endothermic in bromination. Hence, the option (A) is an incorrect statement.
When H+ attacks CH3 – CH = CH2 , carbonation which is more stable is- a)CH3 – CH2 – CH2
- b)CH2+ – CH2 – CH3
- c)CH3 – CH+ – CH2
- d)CH3 – CH2 – CH2+
Correct answer is option 'C'. Can you explain this answer?
When H+ attacks CH3 – CH = CH2 , carbonation which is more stable is
a)
CH3 – CH2 – CH2
b)
CH2+ – CH2 – CH3
c)
CH3 – CH+ – CH2
d)
CH3 – CH2 – CH2+
| | Nitin Khanna answered |
CH3 – CH = CH2 → CH3 – CH+ – CH2
The reason for this is only that carbocation is formed which has maximum stability. In this case, we have 6 α-H while for option a, b and d; we have 0, 2 and 2 α-H respectively. So only carbocation in option c forms.
The reason for this is only that carbocation is formed which has maximum stability. In this case, we have 6 α-H while for option a, b and d; we have 0, 2 and 2 α-H respectively. So only carbocation in option c forms.
Consider the following bromination reaction.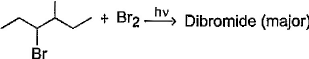 If a pure enantiomer of reactant is taken in the above reaction, the correct statement concerning product dibromide is/are
If a pure enantiomer of reactant is taken in the above reaction, the correct statement concerning product dibromide is/are - a)A racemic mixture is formed
- b)Two optically active isomers are formed
- c)A pair of diastereomers in equal amount is formed
- d)A pair of enantiomers but in unequal amounts is formed
Correct answer is option 'B,C'. Can you explain this answer?
Consider the following bromination reaction.
If a pure enantiomer of reactant is taken in the above reaction, the correct statement concerning product dibromide is/are
a)
A racemic mixture is formed
b)
Two optically active isomers are formed
c)
A pair of diastereomers in equal amount is formed
d)
A pair of enantiomers but in unequal amounts is formed
 | Ashish Mishra answered |
B and C is correct
Arrange the following in increasing order of boiling points.I. 3 -methyl pentane
II. 3-chloropentane
III. 3-bromopentane
IV. 3,3-dichloropentane- a)IV < III < II < I
- b)I < II < IV < III
- c)I < II < III < IV
- d)II < I < IV < III
Correct answer is option 'C'. Can you explain this answer?
Arrange the following in increasing order of boiling points.
I. 3 -methyl pentane
II. 3-chloropentane
III. 3-bromopentane
IV. 3,3-dichloropentane
II. 3-chloropentane
III. 3-bromopentane
IV. 3,3-dichloropentane
a)
IV < III < II < I
b)
I < II < IV < III
c)
I < II < III < IV
d)
II < I < IV < III
| | Om Desai answered |
For boiling point, we have to consider both branching and Molecular mass. In 4 bromopentane molecular mass is nearly the same as compared to 3 chloro pentane but we have 3,3-dichloropentane extended into 2 directions so the boiling point of 3,3-dichloropentane will be more and the other order will be followed by option C.
A hydrocarbon A on chlorination gives B which on heating with alcoholic potassium hydroxide changes into another hydrocarbon C. The latter decolourises Baeyer's reagent and on ozonolysis forms formaldehyde only. A is [1998]- a)Ethane
- b)Butane
- c)Methane
- d)Ethene
Correct answer is option 'A'. Can you explain this answer?
A hydrocarbon A on chlorination gives B which on heating with alcoholic potassium hydroxide changes into another hydrocarbon C. The latter decolourises Baeyer's reagent and on ozonolysis forms formaldehyde only. A is [1998]
a)
Ethane
b)
Butane
c)
Methane
d)
Ethene
 | Prisha Singh answered |
The hydrocarbon 'a' is ethane.
Ethane on chlorination, gives 1-chloroethane, which reacts with alcoholic KOH to give a dehydro-halogenation product (Ethene). The ethene formed from this undergoes ozonolysis to break the double bond and form two molecules of formaldehyde.
'a' - Ethane
'b' - 1-chloroethane
'c' - Ethene
Ethane on chlorination, gives 1-chloroethane, which reacts with alcoholic KOH to give a dehydro-halogenation product (Ethene). The ethene formed from this undergoes ozonolysis to break the double bond and form two molecules of formaldehyde.
'a' - Ethane
'b' - 1-chloroethane
'c' - Ethene
Which of the following systems are aromatic?- a)
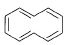
- b)
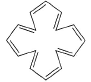
- c)

- d)
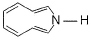
Correct answer is option 'C,D'. Can you explain this answer?
Which of the following systems are aromatic?
a)
b)
c)
d)
| | Pooja Shah answered |
The correct answers are Options C and D.
Aromatic compounds are those which follow Huckel's rule i.e, they have (4n+2) π electrons, n must be an integer.
In option C, there are 5 π bonds which means 10 π electrons; so 4n+2 = 10 i.e, n= 2 which is an integer.
In option D, Nitrogen has a lone pair which contains 2 electrons therefore this compound also have 10 π electrons; so n= 2.
Aromatic compounds are those which follow Huckel's rule i.e, they have (4n+2) π electrons, n must be an integer.
In option C, there are 5 π bonds which means 10 π electrons; so 4n+2 = 10 i.e, n= 2 which is an integer.
In option D, Nitrogen has a lone pair which contains 2 electrons therefore this compound also have 10 π electrons; so n= 2.
A hydrocarbon X (C14H22)o n treatm ent with H2/Pt gives C14H26. Also X on treatm ent with alkaline KMnO4 followed by hydrolysis of products yields C7H12O2 which on further heating with soda lime gives cyclohexane. Hence, X is- a)
- b)
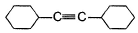
- c)
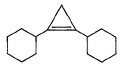
- d)
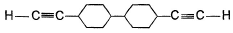
Correct answer is option 'B'. Can you explain this answer?
A hydrocarbon X (C14H22)o n treatm ent with H2/Pt gives C14H26. Also X on treatm ent with alkaline KMnO4 followed by hydrolysis of products yields C7H12O2 which on further heating with soda lime gives cyclohexane. Hence, X is
a)
b)
c)
d)
 | Sushil Kumar answered |
The correct answers are option B.
As only 4 hydrogen atoms are increased after hydrogenation, there should be only 1 3× bond. So option B is correct.
As only 4 hydrogen atoms are increased after hydrogenation, there should be only 1 3× bond. So option B is correct.
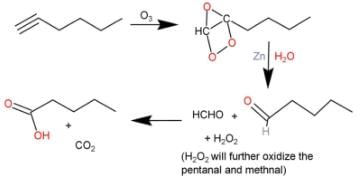
Direction (Q. Nos. 9 - 12) This section contains 4 multiple choice questions. Each question has fo ur choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.Q. What is/are true about ozonolysis of 1-hexyne followed by aqueous work-up?- a)Pentanoic acid and CO2 are formed
- b)Hydrolysis of ozonide of alkyne form H2O2 by product that oxidises the intermediate product into acids
- c)The ozonide has a C— C bond
- d)Pentanal and methanal are formed
Correct answer is option 'A,B,C'. Can you explain this answer?
Direction (Q. Nos. 9 - 12) This section contains 4 multiple choice questions. Each question has fo ur choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q. What is/are true about ozonolysis of 1-hexyne followed by aqueous work-up?
a)
Pentanoic acid and CO2 are formed
b)
Hydrolysis of ozonide of alkyne form H2O2 by product that oxidises the intermediate product into acids
c)
The ozonide has a C— C bond
d)
Pentanal and methanal are formed
| | Pooja Shah answered |
The correct answers are Options A, B and C.

Photochemical chlorination of alkane is initiated by process called:- a)Homolysis
- b)Pyrolysis
- c)Substitution
- d)Peroxidation
Correct answer is option 'A'. Can you explain this answer?
Photochemical chlorination of alkane is initiated by process called:
a)
Homolysis
b)
Pyrolysis
c)
Substitution
d)
Peroxidation
| | Rahul Bansal answered |
Photochemical chlorination of alkane take place by free radical mechanism which are possible by Homolysis of C - C bond
hat is relative reactivity of secondary versus primary hydrogens in free radical bromination of n-butane if the ratio of 1-bromo to 2-bromobutane formed is 7 : 39?- a)200 :1
- b)20 :1
- c)5 :1
- d)1 : 20
Correct answer is option 'B'. Can you explain this answer?
hat is relative reactivity of secondary versus primary hydrogens in free radical bromination of n-butane if the ratio of 1-bromo to 2-bromobutane formed is 7 : 39?
a)
200 :1
b)
20 :1
c)
5 :1
d)
1 : 20
| | Riya Banerjee answered |
Correct Answer :- b
Explanation : The 2o hydrogens are 20 times more reactive than the 1o ones.
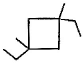
How many different monochlorination products would be obtained on free radical chlorination of methyl cyclobutane?
Correct answer is '8'. Can you explain this answer?
How many different monochlorination products would be obtained on free radical chlorination of methyl cyclobutane?
| | Riya Banerjee answered |
After free radical halogenation of methyl cyclobutane, we have its 8different isomers. They are as follow:-
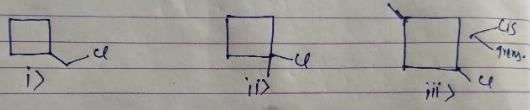

From i) and ii), we get only positional isomers. From iii) we will have 2 isomers, cis and Trans. They won't show a chiral centre.
In iv) we have 2 chiral centres which will give us 4 isomers.So, in total there would be 4+2+1+1 = 8 isomers.


From i) and ii), we get only positional isomers. From iii) we will have 2 isomers, cis and Trans. They won't show a chiral centre.
In iv) we have 2 chiral centres which will give us 4 isomers.So, in total there would be 4+2+1+1 = 8 isomers.
Direction (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.Q. Consider the following compounds. The correct statement regarding properties of above mentioned compounds is/are
The correct statement regarding properties of above mentioned compounds is/are- a)Both have all their C—C bonds of equal length
- b)I does not decolourises brown colour of bromine water solution but II does
- c)Both are planar molecules
- d)I is not absorbed in cold, dil. H2SO4 but II is absorbed
Correct answer is option 'A,B,D'. Can you explain this answer?
Direction (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q. Consider the following compounds.
The correct statement regarding properties of above mentioned compounds is/are
a)
Both have all their C—C bonds of equal length
b)
I does not decolourises brown colour of bromine water solution but II does
c)
Both are planar molecules
d)
I is not absorbed in cold, dil. H2SO4 but II is absorbed
 | Pioneer Academy answered |
- Both have all their C—C bonds of equal length due to conjugation.
- I does not decolorises brown colour of bromine water solution but II does as The π bonds in Cyclooctatetraene (Compound II) react as usual for olefins, rather than as aromatic ring systems.
- I is planar but II is not as it adopts a tub conformation.
- Cyclooctatetraene shows various other addition reactions including Sulfonation.
Hence, Option A, B and D are correct.
Which reagent(s) below results in a visible change withl-butyne and can be used to differentiate it from 2-butyne?- a)

- b)

- c)
- d)
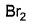
Correct answer is option 'A,B'. Can you explain this answer?
Which reagent(s) below results in a visible change withl-butyne and can be used to differentiate it from 2-butyne?
a)
b)
c)
d)
 | Knowledge Hub answered |
The correct answer is option A,B.
(a) Tollen's reagent produces a white precipitate of acetylide with terminal alkyne.
(b) Fehling solution produces a red precipitate with terminal alkyne.
So $$1-butyne$$ is terminal alkyne.
(a) Tollen's reagent produces a white precipitate of acetylide with terminal alkyne.
(b) Fehling solution produces a red precipitate with terminal alkyne.
So $$1-butyne$$ is terminal alkyne.
The major monobromination product which results when ethyl cyclohexane is subjected to free radical bromination, is- a)
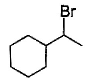
- b)
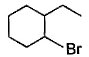
- c)
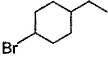
- d)

Correct answer is option 'D'. Can you explain this answer?
The major monobromination product which results when ethyl cyclohexane is subjected to free radical bromination, is
a)
b)
c)
d)
 | Ambition Institute answered |
Free radical bromination reaction is highly selective, occurs mainly at the carbon where most stable free radical is formed.
We know that the stability of free radical is in the order,
Tertiary radical > Secondary radical > Primary radical
In (a), (b) and (c), the bromination occurs at secondary carbon whereas in (d) the bromination occurs at tertiary carbon. Since, tertiary radicals are more stable than secondary radical the major product of monobromination of ethyl cyclohexane is (d).
The stability of tertiary radical is due to the higher number of α−Hygrogens which give more hyperconjugation effect than secondary.
When hydrochloric acid gas is treated with propene in presence of benzoyl peroxide, it gives [1993]- a)2-Chloropropane
- b)Allyl chloride
- c)No reaction
- d)n-Propyl chloride.
Correct answer is option 'A'. Can you explain this answer?
When hydrochloric acid gas is treated with propene in presence of benzoyl peroxide, it gives [1993]
a)
2-Chloropropane
b)
Allyl chloride
c)
No reaction
d)
n-Propyl chloride.
 | Aniket Chawla answered |
Peroxide effect is observed only in case of HBr. Therefore, addition of HCl to propene even in the presence of benzyoyl peroxide occurs according to Markovnikov’s rule :
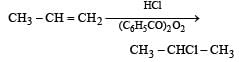
What is true regarding the following compound?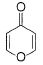
- a)It is aromatic compound
- b)It is a heterocyclic compound
- c)It has very high affinity for BF3
- d)It undergo very rapid reduction with NaBH4
Correct answer is option 'A,B,C'. Can you explain this answer?
What is true regarding the following compound?
a)
It is aromatic compound
b)
It is a heterocyclic compound
c)
It has very high affinity for BF3
d)
It undergo very rapid reduction with NaBH4
| | Lavanya Menon answered |
The given compound will turn itself to
To gain aromaticity, it will transfer the electrons as follow:-
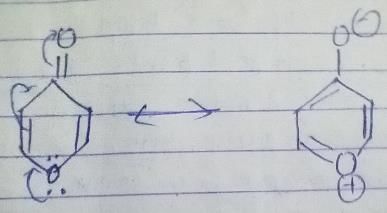
It is clear that the compound is heterocyclic(the ring constitutes other than C and H). Due to -ve charge on outer O atom, it has high affinity for BF3. However, NaBH4 has no reaction with this. As the compound will turn itself to latter, there is no aldehyde or ketone group present in the compound.
To gain aromaticity, it will transfer the electrons as follow:-

It is clear that the compound is heterocyclic(the ring constitutes other than C and H). Due to -ve charge on outer O atom, it has high affinity for BF3. However, NaBH4 has no reaction with this. As the compound will turn itself to latter, there is no aldehyde or ketone group present in the compound.
In preparation of alkene from alcohol using Al2O3 which is effective factor? [2001]- a)Temperature
- b)Concentration
- c)Surface area of Al2O3
- d)Porosity of Al2O3
Correct answer is option 'A'. Can you explain this answer?
In preparation of alkene from alcohol using Al2O3 which is effective factor? [2001]
a)
Temperature
b)
Concentration
c)
Surface area of Al2O3
d)
Porosity of Al2O3
 | Aniket Chawla answered |
Temperature
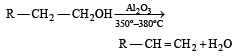
While at 220º – 250ºC it forms ether
What is true about the 1,3,5,7-cyclooctatetraene?- a)It is an aromatic compound
- b)It is an anti-aromatic compound
- c)It on reaction with K metal absorbs two electrons
- d)On refluxing with K metal, it gives off H2 gas
Correct answer is option 'B,C'. Can you explain this answer?
What is true about the 1,3,5,7-cyclooctatetraene?
a)
It is an aromatic compound
b)
It is an anti-aromatic compound
c)
It on reaction with K metal absorbs two electrons
d)
On refluxing with K metal, it gives off H2 gas
| | Tanishq Unni answered |
1-3-5-7-cyclooctatetraene it has 8 pi electrons, and like stated above, fits the criteria of 4n, to be antiaromatic. to avoid this state of anti-aromaticity (less stable then expected), it becomes non-planar, so it can be more stable then it would be in the antiaromatic state. cyclooctatetraene can do this because it can fold, however other 6 carbon compounds that have 4n electrons and are planar can not and result in an antiaromatic compound.
Potassium cyclooctatetraene is formed by the reaction of cyclooctatetraene with potassium metal:
2 K + C8H8 → K2C8H8
The reaction entails 2-electron reduction of the polyene and is accompanied by a color change from colorless to brown.
Potassium cyclooctatetraene is formed by the reaction of cyclooctatetraene with potassium metal:
2 K + C8H8 → K2C8H8
The reaction entails 2-electron reduction of the polyene and is accompanied by a color change from colorless to brown.
Which of the following compounds has the lowest boiling point ? [1994]- a)CH3CH2CH2CH2CH3
- b)CH3CH = CHCH2CH3
- c)CH3CH = CH – CH = CH2
- d)CH3CH2CH2CH3
Correct answer is option 'D'. Can you explain this answer?
Which of the following compounds has the lowest boiling point ? [1994]
a)
CH3CH2CH2CH2CH3
b)
CH3CH = CHCH2CH3
c)
CH3CH = CH – CH = CH2
d)
CH3CH2CH2CH3
 | Ishani Nambiar answered |
Of all the options listed CH3CH2CH2CH3 has the least number of C-atoms and hence has the lowest b.p.
Hydrogenation of alkenes can be carried out in the presence of- a)copper
- b)zinc
- c)aluminium
- d)nickel
Correct answer is option 'D'. Can you explain this answer?
Hydrogenation of alkenes can be carried out in the presence of
a)
copper
b)
zinc
c)
aluminium
d)
nickel
 | Stepway Academy answered |
- Unsaturated hydrocarbons such as alkenes add Hydrogen in the presence of catalysts such as Ni to form saturated hydrocarbons. This is called an addition reaction.
- The reaction is commonly used in the hydrogenation of vegetable oils using a Nickel catalyst.
7-bromo-1,3,5-cycloheptatriene exists as ionic species in aqueous solution while 5-bromo-1,3-cyclopentadiene does not ionise even in presence of AgNO3(aq) because- a)C—Br bond has partial double bond character in 5-bromo-1,3-cyclopentadiene
- b)7-bromo-1,3,5-cyclopheptatriene ionises to form an aromatic anion while 5-bromo-1,3-cyclopentadiene ionises to form an anti-aromatic cation
- c)7-bromo-1,3,5-cycloheptatriene ionises into an aromatic cation while 5-bromo-1,3 cyclopentadiene ionises into a highly unstable anti-aromatic cation
- d)Both ionises to form anions but one form aromatic while other form anti-aromatic anion
Correct answer is option 'C'. Can you explain this answer?
7-bromo-1,3,5-cycloheptatriene exists as ionic species in aqueous solution while 5-bromo-1,3-cyclopentadiene does not ionise even in presence of AgNO3(aq) because
a)
C—Br bond has partial double bond character in 5-bromo-1,3-cyclopentadiene
b)
7-bromo-1,3,5-cyclopheptatriene ionises to form an aromatic anion while 5-bromo-1,3-cyclopentadiene ionises to form an anti-aromatic cation
c)
7-bromo-1,3,5-cycloheptatriene ionises into an aromatic cation while 5-bromo-1,3 cyclopentadiene ionises into a highly unstable anti-aromatic cation
d)
Both ionises to form anions but one form aromatic while other form anti-aromatic anion
| | Akash Chakraborty answered |
The correct answer is Option C.
The C-Br bond in the case of 7-bromo-1, 3, 5-cycloheptatriene is broken easily because the intermediate carbocation formed is very stable (aromatic as it contains (4n + 2)π e- ie, follows Huckle rule) while it does not break easily in the case of S-bromo-1, 3-cyclopentadiene because carbocation formed here is highly unstable as it is anti aromatic ie, does not follow Huckel rule. (It contains 4π electrons).
The C-Br bond in the case of 7-bromo-1, 3, 5-cycloheptatriene is broken easily because the intermediate carbocation formed is very stable (aromatic as it contains (4n + 2)π e- ie, follows Huckle rule) while it does not break easily in the case of S-bromo-1, 3-cyclopentadiene because carbocation formed here is highly unstable as it is anti aromatic ie, does not follow Huckel rule. (It contains 4π electrons).
From following list, how many of them upon catalytic hydrogenation would produce more heat than that produced in catalytic hydrogenation of frans-2-butene?
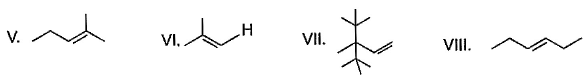
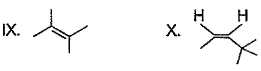
Correct answer is '4'. Can you explain this answer?
From following list, how many of them upon catalytic hydrogenation would produce more heat than that produced in catalytic hydrogenation of frans-2-butene?
| | Preeti Iyer answered |
Correct answer is 4 because heat of hydrogenation is directly proportional to number of carbon atoms present in it
Which compound below has maximum tendency to form a salt when treated with HBr?- a)
- b)

- c)
- d)
Correct answer is option 'C'. Can you explain this answer?
Which compound below has maximum tendency to form a salt when treated with HBr?
a)
b)
c)
d)
 | Gaurav Saini answered |
Due formation of more resonance structure.
Step1: Take the radius of the molecule
Step2: molecular formula,v =(4/3)πr3N
Where N is AVOGADRO’S Number=
6.0221409e+23
Step 3: putting values
Step5: using percentage formula
The IUPAC name of the compound CH3CH = CHC = CH is [2010]- a)Pent-l-yn-3-ene
- b)Pent-4-yn-2-ene
- c)Pent-3-en-1-yne
- d)Pent-2-en-4-yne
Correct answer is option 'C'. Can you explain this answer?
The IUPAC name of the compound CH3CH = CHC = CH is [2010]
a)
Pent-l-yn-3-ene
b)
Pent-4-yn-2-ene
c)
Pent-3-en-1-yne
d)
Pent-2-en-4-yne
 | Snehal Shah answered |
When both double and triple bonds are present, then triple bond is considered as the principal group.
The reaction of C6H5CH = CHCH3 with HBr produces :- a)
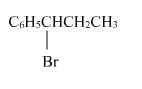
- b)
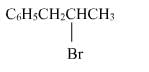
- c)
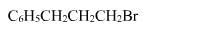
- d)
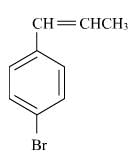
Correct answer is option 'A'. Can you explain this answer?
The reaction of C6H5CH = CHCH3 with HBr produces :
a)
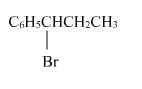
b)

c)
d)

| | Raghav Bansal answered |
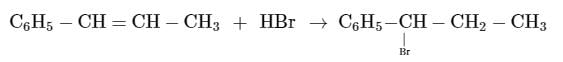
This is the electrophilic addition reaction in which addition takes place via more stable carbocation according to the Markovnikov's rule.
Chapter doubts & questions for Hydrocarbons - Topic-wise MCQ Tests for NEET 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Hydrocarbons - Topic-wise MCQ Tests for NEET in English & Hindi are available as part of NEET exam. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
;
Signup to see your scores go up
within 7 days!
within 7 days!
Takes less than 10 seconds to signup