All Exams > NEET > NCERT Based Tests for NEET > All Questions
All questions of Biomolecules for NEET Exam
Synthesis of protein is controlled by:- a)Nucleotide
- b)RNA
- c)DNA
- d)Nucleoside
Correct answer is option 'B'. Can you explain this answer?
Synthesis of protein is controlled by:
a)
Nucleotide
b)
RNA
c)
DNA
d)
Nucleoside
| | Lavanya Menon answered |
RNA can directly control the synthesis of proteins, hence can easily express the characters. DNA, however, is dependent on RNA for synthesis of proteins. The protein synthesising machinery has evolved around RNA.
The compound which gives red colour with Fehling’s solution?- a)Cellulose
- b)Benzaldehyde
- c)Cane sugar
- d)Glucose
Correct answer is option 'D'. Can you explain this answer?
The compound which gives red colour with Fehling’s solution?
a)
Cellulose
b)
Benzaldehyde
c)
Cane sugar
d)
Glucose
 | Knowledge Hub answered |
Fehling's solution to make difference between carbohydrate and ketone functional grp and also used for differentiate reducing and non reducing sugar and as u know glucose is a reducing sugar so it will give red colour with fehling's solution (all monosachharides are reducing sugar).
The essential amino acid is:- a)Serine
- b)Arginine
- c)Alanine
- d)Glycine
Correct answer is option 'B'. Can you explain this answer?
The essential amino acid is:
a)
Serine
b)
Arginine
c)
Alanine
d)
Glycine
| | Naina Bansal answered |
In humans, arginine is classified as a semiessential or conditionally essential amino acid, depending on the developmental stage and health status of the individual. Preterm infants are unable to synthesize or create arginine internally, making the amino acid nutritionally essential for them.
Alpha helix is found in- a)RNA
- b)Lipid
- c)Carbohydrates
- d)secondary proteins
Correct answer is option 'D'. Can you explain this answer?
Alpha helix is found in
a)
RNA
b)
Lipid
c)
Carbohydrates
d)
secondary proteins
| | Ræjû Bhæï answered |
The alpha helix (α-helix) is a common motif in the secondary structure of proteins and is a right hand-helix conformation in which every backbone N−H group hydrogen bonds to the backbone C=O. group of the amino acid located three or four residues earlier along the protein sequence.
The enzyme pepsin hydrolyses:- a)polysaccharides to monosaccharides
- b)glucose to ethyl alcohol
- c)proteins to amino acids
- d)fats to fatty acids
Correct answer is option 'C'. Can you explain this answer?
The enzyme pepsin hydrolyses:
a)
polysaccharides to monosaccharides
b)
glucose to ethyl alcohol
c)
proteins to amino acids
d)
fats to fatty acids
| | Krishna Iyer answered |
The correct answer is Option C
Pepsin hydrolyses proteins to amino acid(polypeptide chains) and initiates protein digestion in the digestion process in humans.
Pepsin hydrolyses proteins to amino acid(polypeptide chains) and initiates protein digestion in the digestion process in humans.
Haemoglobin is an example of:- a)Quaternary Structure of protein
- b)Tertiary Structure of protein
- c)Primary Structure of protein
- d)Seconary Structure of protein
Correct answer is option 'A'. Can you explain this answer?
Haemoglobin is an example of:
a)
Quaternary Structure of protein
b)
Tertiary Structure of protein
c)
Primary Structure of protein
d)
Seconary Structure of protein
| | Om Desai answered |
The correct answer is A
Haemoglobin is a Quaternary protein because it has 4 polypeptide structures - 2alpha and 2 beta.
Haemoglobin is a Quaternary protein because it has 4 polypeptide structures - 2alpha and 2 beta.
Glucose + Tollen’s reagent → Silver mirror. The process shows:- a)The presence of -CHO group
- b)The presence of alkaline group
- c)The presence of acidic group
- d)The presence of keto group
Correct answer is option 'A'. Can you explain this answer?
Glucose + Tollen’s reagent → Silver mirror. The process shows:
a)
The presence of -CHO group
b)
The presence of alkaline group
c)
The presence of acidic group
d)
The presence of keto group
| | Swati Verma answered |
Glucose gives silver mirror test with Tollens reagent. It shows the presence of aldehyde group.
Also, mild oxidation of glucose with bromine water gives gluconic acid which shows the presence of aldehyde group.
Also, mild oxidation of glucose with bromine water gives gluconic acid which shows the presence of aldehyde group.
The active form of vitamin D is:- a)Calcidiol
- b)Calcitriol
- c)Cholecalciferol
- d)Any of the above
- e)None of the above
Correct answer is option 'B'. Can you explain this answer?
The active form of vitamin D is:
a)
Calcidiol
b)
Calcitriol
c)
Cholecalciferol
d)
Any of the above
e)
None of the above
 | Rajdeep Saini answered |
The active form of vitamin D is calcitriol.
Calcitriol, also known as 1,25-dihydroxyvitamin D3, is the active form of vitamin D in the body. It is the biologically active metabolite that is synthesized from calcidiol, also known as 25-hydroxyvitamin D3, which is the storage form of vitamin D.
Explanation:
1. Vitamin D and its forms:
Vitamin D is a fat-soluble vitamin that plays a crucial role in maintaining bone health and calcium homeostasis. It exists in several forms, including vitamin D2 (ergocalciferol) and vitamin D3 (cholecalciferol), which are obtained from dietary sources or synthesized in the skin upon exposure to sunlight.
2. Conversion of vitamin D to its active form:
When vitamin D2 or D3 is ingested or synthesized, it undergoes a series of metabolic conversions in the liver and kidneys to become the active form of vitamin D. The first step involves hydroxylation in the liver, where vitamin D is converted to calcidiol (25-hydroxyvitamin D3) through the action of the enzyme 25-hydroxylase.
3. Activation of calcidiol to calcitriol:
The second and final step in the activation of vitamin D occurs in the kidneys. Calcidiol undergoes further hydroxylation by the enzyme 1-alpha-hydroxylase, resulting in the formation of calcitriol (1,25-dihydroxyvitamin D3). This conversion is tightly regulated by various factors, including parathyroid hormone (PTH), calcium levels, and phosphate levels.
4. Biological functions of calcitriol:
Calcitriol acts as a hormone and binds to the vitamin D receptor (VDR) in target tissues, such as the intestines, bones, and kidneys. It regulates the absorption of calcium and phosphate in the intestines, promotes bone mineralization by increasing calcium and phosphate uptake, and helps maintain normal blood calcium levels. Calcitriol also has non-skeletal functions, including modulation of immune responses and cell growth.
Conclusion:
In conclusion, the active form of vitamin D is calcitriol, which is synthesized from calcidiol through the action of 1-alpha-hydroxylase in the kidneys. Calcitriol plays a crucial role in maintaining calcium homeostasis and bone health, as well as other physiological processes in the body.
Calcitriol, also known as 1,25-dihydroxyvitamin D3, is the active form of vitamin D in the body. It is the biologically active metabolite that is synthesized from calcidiol, also known as 25-hydroxyvitamin D3, which is the storage form of vitamin D.
Explanation:
1. Vitamin D and its forms:
Vitamin D is a fat-soluble vitamin that plays a crucial role in maintaining bone health and calcium homeostasis. It exists in several forms, including vitamin D2 (ergocalciferol) and vitamin D3 (cholecalciferol), which are obtained from dietary sources or synthesized in the skin upon exposure to sunlight.
2. Conversion of vitamin D to its active form:
When vitamin D2 or D3 is ingested or synthesized, it undergoes a series of metabolic conversions in the liver and kidneys to become the active form of vitamin D. The first step involves hydroxylation in the liver, where vitamin D is converted to calcidiol (25-hydroxyvitamin D3) through the action of the enzyme 25-hydroxylase.
3. Activation of calcidiol to calcitriol:
The second and final step in the activation of vitamin D occurs in the kidneys. Calcidiol undergoes further hydroxylation by the enzyme 1-alpha-hydroxylase, resulting in the formation of calcitriol (1,25-dihydroxyvitamin D3). This conversion is tightly regulated by various factors, including parathyroid hormone (PTH), calcium levels, and phosphate levels.
4. Biological functions of calcitriol:
Calcitriol acts as a hormone and binds to the vitamin D receptor (VDR) in target tissues, such as the intestines, bones, and kidneys. It regulates the absorption of calcium and phosphate in the intestines, promotes bone mineralization by increasing calcium and phosphate uptake, and helps maintain normal blood calcium levels. Calcitriol also has non-skeletal functions, including modulation of immune responses and cell growth.
Conclusion:
In conclusion, the active form of vitamin D is calcitriol, which is synthesized from calcidiol through the action of 1-alpha-hydroxylase in the kidneys. Calcitriol plays a crucial role in maintaining calcium homeostasis and bone health, as well as other physiological processes in the body.
Name the RNA molecules which is used to carry genetic information copied from DNA?- a)tRNA
- b)mRNA
- c)rRNA
- d)snRNA
Correct answer is option 'B'. Can you explain this answer?
Name the RNA molecules which is used to carry genetic information copied from DNA?
a)
tRNA
b)
mRNA
c)
rRNA
d)
snRNA
| | Raushani Praween answered |
Because mRNA is called messanger RNA, which carries the information from DNA to synthesis of protein and other essential products in cell.
Glucose and fructose are:- a)Position isomers
- b)Functional isomers
- c)Chain isomers
- d)Optical isomers
Correct answer is option 'B'. Can you explain this answer?
Glucose and fructose are:
a)
Position isomers
b)
Functional isomers
c)
Chain isomers
d)
Optical isomers
| | Tanvi Bose answered |
Glucose and fructose are functional isomers of each other Because they have same molecular formula that is C6H12O6 But different functional group in their chemical formula. Glucose has aldehyde group while fructose has ketone as functional group.
Process which leads to the synthesis of RNA is called- a)Termination
- b)Replication
- c)Transcription
- d)Translation
Correct answer is option 'C'. Can you explain this answer?
Process which leads to the synthesis of RNA is called
a)
Termination
b)
Replication
c)
Transcription
d)
Translation
 | Srishti Raghuwanshi answered |
DNA TO DNA - replication
DNA TO RNA-transcription
RNA TO PROTEIN-translation
DNA TO RNA-transcription
RNA TO PROTEIN-translation
Purine derivative among the following bases is:- a)Uracil
- b)Cytosine
- c)Guanine
- d)Thymine
Correct answer is option 'C'. Can you explain this answer?
Purine derivative among the following bases is:
a)
Uracil
b)
Cytosine
c)
Guanine
d)
Thymine
| | Shail Chawla answered |
Purine is a type of nitrogenous base found in DNA and RNA molecules. The purine bases are adenine and guanine. Among the given options, the purine derivative is:
c) Guanine
Explanation:
- Purine and pyrimidine are two types of nitrogenous bases found in DNA and RNA molecules.
- Purine bases are larger and have a double-ring structure, while pyrimidine bases are smaller and have a single-ring structure.
- Adenine and guanine are purine bases, while cytosine, thymine, and uracil are pyrimidine bases.
- Therefore, among the given options, the purine derivative is guanine.
c) Guanine
Explanation:
- Purine and pyrimidine are two types of nitrogenous bases found in DNA and RNA molecules.
- Purine bases are larger and have a double-ring structure, while pyrimidine bases are smaller and have a single-ring structure.
- Adenine and guanine are purine bases, while cytosine, thymine, and uracil are pyrimidine bases.
- Therefore, among the given options, the purine derivative is guanine.
Nucleoside differs from nucleotide with the absence of:- a)Pentose sugar and Nitrogenous base
- b)Nitrogenous base
- c)Phosphoric acid
- d)Pentose sugar
Correct answer is option 'C'. Can you explain this answer?
Nucleoside differs from nucleotide with the absence of:
a)
Pentose sugar and Nitrogenous base
b)
Nitrogenous base
c)
Phosphoric acid
d)
Pentose sugar
| | Arun Khanna answered |
The structure of nucleotide and nucleoside units are distinguished primarily by the presence (or lack thereof) of this phosphate group. Deoxyribose in DNA differs from the ribose found in RNA in that it has only a hydrogen atom in the same position that ribose has a hydroxyl (-OH) group.
Which of the following carbohydrate is an example of an oligosaccharide?- a)Cellulose
- b)Lactose
- c)Mannose
- d)Glucose
Correct answer is option 'B'. Can you explain this answer?
Which of the following carbohydrate is an example of an oligosaccharide?
a)
Cellulose
b)
Lactose
c)
Mannose
d)
Glucose
| | Anjali Iyer answered |
Lactose intolerance is the inability to break down a type of natural sugar called lactose. Lactose is commonly found in dairy products, such as milk and yogurt. A person becomes lactose intolerant when his or her small intestine stops making enough of the enzyme lactase to digest and break down the lactose.
Hydrolysis of sucrose is called:- a)Hydration
- b)Inversion
- c)Esterification
- d)Saponification
Correct answer is option 'B'. Can you explain this answer?
Hydrolysis of sucrose is called:
a)
Hydration
b)
Inversion
c)
Esterification
d)
Saponification
 | Sahana Savalagi answered |
Hydrolysis of sucrose is inversion because the angle of specific rotation of the plane polarized light changes from positive to negative value due to the presence of optical isomers of mixture of glucose and fructose sugar...
On hydrolysis, proteins give:- a)Amides
- b)Amino acids
- c)Nucleotides
- d)Nucleosides
Correct answer is option 'B'. Can you explain this answer?
On hydrolysis, proteins give:
a)
Amides
b)
Amino acids
c)
Nucleotides
d)
Nucleosides
 | Aiims answered |
Proteins are polymers of amino acids therefore their hydrolysis gives amino acids
Carbohydrates are classified on the basis of their behaviour on hydrolysis and also as reducing or non-reducing sugar. Sucrose is a __________.- a)monosaccharide
- b)disaccharide
- c)non-reducing sugar
- d)Both b and c
Correct answer is option 'D'. Can you explain this answer?
Carbohydrates are classified on the basis of their behaviour on hydrolysis and also as reducing or non-reducing sugar. Sucrose is a __________.
a)
monosaccharide
b)
disaccharide
c)
non-reducing sugar
d)
Both b and c
 | Divey Sethi answered |
The correct answer is option B & D.
One of the common disaccharides is sucrose which on hydrolysis gives an equimolar mixture of D−(+)− glucose and D−(−) fructose. These two monosaccharides are held together by a glycosidic linkage between Cl of α- glucose and C2 of β- fructose. Since the reducing groups of glucose and fructose are involved in glycosidic bond formation, sucrose is a non-reducing sugar.
One of the common disaccharides is sucrose which on hydrolysis gives an equimolar mixture of D−(+)− glucose and D−(−) fructose. These two monosaccharides are held together by a glycosidic linkage between Cl of α- glucose and C2 of β- fructose. Since the reducing groups of glucose and fructose are involved in glycosidic bond formation, sucrose is a non-reducing sugar.
Nucleotides are joined together by 5′ and 3′ carbon atoms of pentose sugar. The linkage is known as: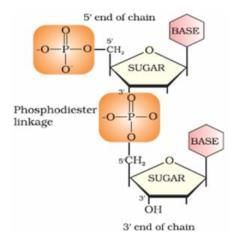
- a)Glycosidic
- b)Peptide
- c)Hydrogen
- d)Phoshodiester
Correct answer is option 'D'. Can you explain this answer?
Nucleotides are joined together by 5′ and 3′ carbon atoms of pentose sugar. The linkage is known as:

a)
Glycosidic
b)
Peptide
c)
Hydrogen
d)
Phoshodiester
| | Preeti Iyer answered |
Nucleotides are joined together by phosphodiester linkage between 5′ and 3′ carbon atoms of pentose sugar.
Which of the following base is not found in DNA?- a)Uracil
- b)Guanine
- c)Cytosine
- d)Adenine
Correct answer is option 'A'. Can you explain this answer?
Which of the following base is not found in DNA?
a)
Uracil
b)
Guanine
c)
Cytosine
d)
Adenine
| | Ritu Pal answered |
Uracil is not found in DNA , as it Uracil has more base pair affinity to adenine , guanine and cytosine. Instead thymine is present in DNA.
RNA is different from DNA because RNA contains:- a)Ribose sugar and thymine
- b)Deoxyribose sugar and thymine
- c)Ribose sugar and uracil
- d)Deoxyribose sugar and uracil
Correct answer is option 'C'. Can you explain this answer?
RNA is different from DNA because RNA contains:
a)
Ribose sugar and thymine
b)
Deoxyribose sugar and thymine
c)
Ribose sugar and uracil
d)
Deoxyribose sugar and uracil
| | Priyanka Sharma answered |
RNA is ribonucliec acid, it contains ribose sugar and uracil. In DNA molecules, the sugar is 2-deoxyribose. In DNA, four bases have been found. They are adenine (A), guanine (G), cytosine (C) and thymine (T). The first three of these bases are found in RNA also but the fourth is uracil (U).
Three structures are given below in which two glucose units are linked. Which of these linkages between glucose units are between C1 and C4 and which linkages are between C1 and C6?
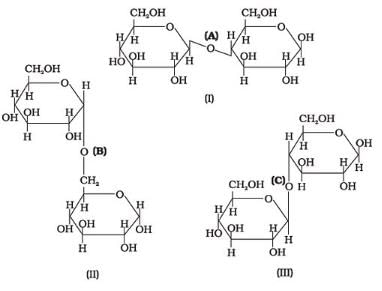
- a)(A) is between C1 and C4, (B) and (C) are between C1 and C6
- b)(A) and (B) are between C1 and C4, (C) is between C1 and C6
- c)(A) and (C) are between C1 and C4, (B) is between C1 and C6
- d)(A) and (C) are between C1 and C6, (B) is between C1 and C4
Correct answer is option 'C'. Can you explain this answer?
Three structures are given below in which two glucose units are linked. Which of these linkages between glucose units are between C1 and C4 and which linkages are between C1 and C6?


a)
(A) is between C1 and C4, (B) and (C) are between C1 and C6
b)
(A) and (B) are between C1 and C4, (C) is between C1 and C6
c)
(A) and (C) are between C1 and C4, (B) is between C1 and C6
d)
(A) and (C) are between C1 and C6, (B) is between C1 and C4
| | Preeti Iyer answered |
The correct answer is Option C.
(i) The first structure is lactose, the linkage is (A) C1−C4 glycosidic linkage.
(ii) The 2nd structure has C1−C6 linkages.
(iii) The 3rd structure has C1−C4 linkages.
(ii) The 2nd structure has C1−C6 linkages.
(iii) The 3rd structure has C1−C4 linkages.

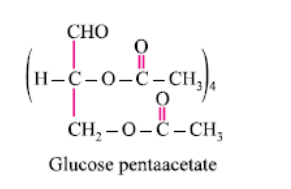
Glucose reacts with acetic anhydride to form:- a)Monoacetate
- b)Diacetate
- c)Pentaacetate
- d) Hexaacetate
Correct answer is option 'C'. Can you explain this answer?
Glucose reacts with acetic anhydride to form:
a)
Monoacetate
b)
Diacetate
c)
Pentaacetate
d)
Hexaacetate
| | Pooja Mehta answered |
It forms glucose pentaacetate. The acetic anhydride esterifies with all the alcohol groups on the glucose ring.

Which of the following statements is correct about Enzymes?- a)Enzymes have optimum activity at body temperature.
- b)Enzymes are carbohydrates.
- c)Enzymes have all these properties.
- d)Enzymes consist of nucleic acid.
Correct answer is option 'A'. Can you explain this answer?
Which of the following statements is correct about Enzymes?
a)
Enzymes have optimum activity at body temperature.
b)
Enzymes are carbohydrates.
c)
Enzymes have all these properties.
d)
Enzymes consist of nucleic acid.
 | Honey answered |
Yes. option a is correct as enzymes are proteins and contain amino acids and the correct option is only they have optimum activity at body temperature... hope you got it...
In which structure of protein, the polypeptide chain forms all possible hydrogen bonds by twisting into right handed screw?- a)Primary structure
- b)Tertiary structure
- c)β-pleated sheet structure
- d)α-helix structure
Correct answer is option 'D'. Can you explain this answer?
In which structure of protein, the polypeptide chain forms all possible hydrogen bonds by twisting into right handed screw?
a)
Primary structure
b)
Tertiary structure
c)
β-pleated sheet structure
d)
α-helix structure
| | Swara Sharma answered |
Alpha helix in secondary structure.
The oxide linkage formed by the loss of a water molecule when two monosaccharides are joined together through oxygen atom is called:- a)Carboxylic linkage
- b)Carbonyl linkage
- c)Peptide linkage
- d)Glycosidic linkage
Correct answer is option 'D'. Can you explain this answer?
The oxide linkage formed by the loss of a water molecule when two monosaccharides are joined together through oxygen atom is called:
a)
Carboxylic linkage
b)
Carbonyl linkage
c)
Peptide linkage
d)
Glycosidic linkage
 | Aleena Mathew answered |
All sacchrides /carbohydrates form glycosidic bond by eliminating water molecule
Sucrose is a non-reducing sugar because:- a)The -CHO group of glucose is not involved in glycosidic bond formation.
- b)Two monosaccharide units are held together by a glycosidic linkage between C1 of α-glucose and C2 of β-fructose.
- c)On hydrolysis, sucrose gives dextrorotatory and laevorotatory and the mixture is laevorotatory.
- d)Sucrose is dextrorotatory.
Correct answer is option 'B'. Can you explain this answer?
Sucrose is a non-reducing sugar because:
a)
The -CHO group of glucose is not involved in glycosidic bond formation.
b)
Two monosaccharide units are held together by a glycosidic linkage between C1 of α-glucose and C2 of β-fructose.
c)
On hydrolysis, sucrose gives dextrorotatory and laevorotatory and the mixture is laevorotatory.
d)
Sucrose is dextrorotatory.
| | Gowri Menon answered |
Glucose and C2 of fructose, which does not have a free aldehyde or ketone group to undergo oxidation and reduction reactions.
c)Sucrose does not react with Benedict's reagent, which is used to detect the presence of reducing sugars.
d)Sucrose cannot be hydrolyzed by acid or enzyme into its constituent monosaccharides.
c)Sucrose does not react with Benedict's reagent, which is used to detect the presence of reducing sugars.
d)Sucrose cannot be hydrolyzed by acid or enzyme into its constituent monosaccharides.
The Molisch test is a chemical test that determines the presence of:- a)All carbohydrates
- b)Sucrose
- c)Fructose
- d)Glucose
Correct answer is option 'A'. Can you explain this answer?
The Molisch test is a chemical test that determines the presence of:
a)
All carbohydrates
b)
Sucrose
c)
Fructose
d)
Glucose
| | Anjali Iyer answered |
The Molisch test uses concentrated sulphuric acid as the dehydrating acid. This acid dehydrates all carbohydrates, so the test is used to distinguish between carbohydrates and non-carbohydrates.
What is the relation between the following pairs of monosaccharides?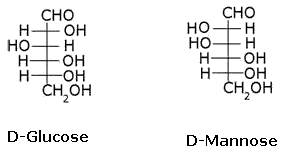
- a)Epimers
- b)Enatiomers
- c)Structural isomers
- d)Anomers
Correct answer is option 'A'. Can you explain this answer?
What is the relation between the following pairs of monosaccharides?
a)
Epimers
b)
Enatiomers
c)
Structural isomers
d)
Anomers
 | Infinity Academy answered |
The pairs of optical isomers which differ in configuration around any other C atom other than C1 atom are called epimers. D-glucose and D-mannose are C2 epimers.
In fibrous proteins, polypeptide chains are held together by ___________.- a)van der Waals forces
- b)disulphide linkage & hydrogen bonds
- c)electrostatic forces of attraction
- d)None of these
Correct answer is option 'B'. Can you explain this answer?
In fibrous proteins, polypeptide chains are held together by ___________.
a)
van der Waals forces
b)
disulphide linkage & hydrogen bonds
c)
electrostatic forces of attraction
d)
None of these
| | Neha Sharma answered |
The correct answer are options B & D,
In fibrous proteins, polypeptide chains are held together by hydrogen and disulphide bonds, in parallel manner. Due to which fibre-like structure is obtained. Such proteins are generally known as fibrous proteins. These proteins are generally insoluble in water e.g., keratin, myosin.
In fibrous proteins, polypeptide chains are held together by hydrogen and disulphide bonds, in parallel manner. Due to which fibre-like structure is obtained. Such proteins are generally known as fibrous proteins. These proteins are generally insoluble in water e.g., keratin, myosin.
On hydrolysis maltose gives:- a)One molecule of glucose and one molecule of galactose
- b)Two molecules of fructose
- c)One molecule of glucose and one molecule of fructose
- d)Two molecules of glucose
Correct answer is option 'D'. Can you explain this answer?
On hydrolysis maltose gives:
a)
One molecule of glucose and one molecule of galactose
b)
Two molecules of fructose
c)
One molecule of glucose and one molecule of fructose
d)
Two molecules of glucose
| | Alok Mehta answered |
On hydrolysis, maltose gives glucose with the help of maltase enzyme which works as a catalysis of the hydrolysis of the glycoside bond.
Maltase is a disaccharide which will reduce sugar giving two molecules of glucose on hydrolysis.
It will give alpha-D-glucose and alpha-D-glucose.
Which among the following is a non-reducing sugar?- a)Sucrose
- b)Maltose
- c)Lactose
- d)Glucose
Correct answer is option 'A'. Can you explain this answer?
Which among the following is a non-reducing sugar?
a)
Sucrose
b)
Maltose
c)
Lactose
d)
Glucose
| | Rajeev Saxena answered |
Non-reducing sugars do not have an OH group attached to the anomeric carbon so they cannot reduce other compounds. All monosaccharides such as glucose are reducing sugars. A disaccharide can be a reducing sugar or a non-reducing sugar. Maltose and lactose are reducing sugars, while sucrose is a non-reducing sugar.
The commonest disaccharide have the molecular formula:- a)C10H20O10
- b)C12H22O11
- c)C10H18O9
- d)C11H22O11
Correct answer is option 'B'. Can you explain this answer?
The commonest disaccharide have the molecular formula:
a)
C10H20O10
b)
C12H22O11
c)
C10H18O9
d)
C11H22O11
 | Sanjeev Kumar answered |
Because it is in the ratio of 1:2:1
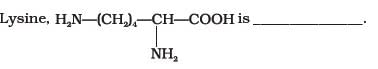
- a)α-Amino acid
- b)Basic amino acid
- c)Amino acid synthesised in body
- d)β-Amino acid
Correct answer is option 'A,B'. Can you explain this answer?
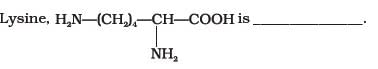
a)
α-Amino acid
b)
Basic amino acid
c)
Amino acid synthesised in body
d)
β-Amino acid
 | Sadiya Siddique answered |
In lysine,the amino group is attached to alpha carbon which makes it Alpha amino acid and it has more amino groups as compared to -COOH groups ,thus basic . so, op A,B.
What does the following reaction shows about the structure of glucose?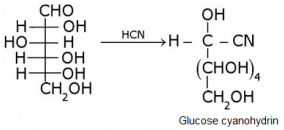
- a)It shows the presence of carbonyl group in glucose.
- b)It shows the presence of primary alcoholic group in glucose.
- c)It shows the presence of ring structure in glucose.
- d)none
Correct answer is option 'A'. Can you explain this answer?
What does the following reaction shows about the structure of glucose?
a)
It shows the presence of carbonyl group in glucose.
b)
It shows the presence of primary alcoholic group in glucose.
c)
It shows the presence of ring structure in glucose.
d)
none
| | Geetika Shah answered |
The correct answer is Option A.
Glucose gets oxidized to cyanohydrin in reaction with HCN . This indicates that the carbonyl group is present as an aldehydic group.
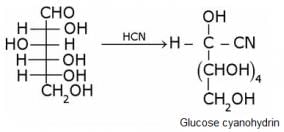
Glucose gets oxidized to cyanohydrin in reaction with HCN . This indicates that the carbonyl group is present as an aldehydic group.

Read the passage given below and answer the following questions:
When a protein in its native form, is subjected to physical changes like change in temperature or chemical changes like change in pH, the hydrogen bonds are disturbed. Due to this, globules unfold and helix get uncoiled and protein loses its biological activity. This is called denaturation of protein.
The denaturation causes change in secondary and tertiary structures but primary structures remains intact.
Examples of denaturation of protein are coagulation of egg white on boiling, curding of milk, formation of cheese when an acid is added to milk.
Q. Mark the wrong statement about denaturation of proteins.- a)The primary structure of the protein does not change.
- b)Globular proteins are converted into fibrous proteins.
- c)Fibrous proteins are converted into globular proteins.
- d)The biological activity of the protein is destroyed.
Correct answer is option 'C'. Can you explain this answer?
Read the passage given below and answer the following questions:
When a protein in its native form, is subjected to physical changes like change in temperature or chemical changes like change in pH, the hydrogen bonds are disturbed. Due to this, globules unfold and helix get uncoiled and protein loses its biological activity. This is called denaturation of protein.
The denaturation causes change in secondary and tertiary structures but primary structures remains intact.
Examples of denaturation of protein are coagulation of egg white on boiling, curding of milk, formation of cheese when an acid is added to milk.
Q. Mark the wrong statement about denaturation of proteins.
When a protein in its native form, is subjected to physical changes like change in temperature or chemical changes like change in pH, the hydrogen bonds are disturbed. Due to this, globules unfold and helix get uncoiled and protein loses its biological activity. This is called denaturation of protein.
The denaturation causes change in secondary and tertiary structures but primary structures remains intact.
Examples of denaturation of protein are coagulation of egg white on boiling, curding of milk, formation of cheese when an acid is added to milk.
Q. Mark the wrong statement about denaturation of proteins.
a)
The primary structure of the protein does not change.
b)
Globular proteins are converted into fibrous proteins.
c)
Fibrous proteins are converted into globular proteins.
d)
The biological activity of the protein is destroyed.
 | Abhishek Mishraclasses answered |
Due to denaturation, globules unfold and helix get uncoiled and protein loses its biological activity. Thus globular proteins are converted into fibrous proteins.
Hence, (C) is wrong.
Hence, (C) is wrong.
Which of the following are purine bases?- a)Guanine
- b)Adenine
- c)Thymine
- d)Uracil
Correct answer is option 'A,B'. Can you explain this answer?
Which of the following are purine bases?
a)
Guanine
b)
Adenine
c)
Thymine
d)
Uracil
| | Tanvi Bose answered |
Explanation:
Purine bases:
Purine bases are nitrogen-containing molecules that are formed by a pyrimidine ring fused to an imidazole ring. The purine bases are guanine and adenine.
Pyrimidine bases:
Pyrimidine bases consist of a single ring structure and include thymine, cytosine, and uracil.
Answer:
Therefore, the correct answer is option A and B, which are guanine and adenine. These bases are found in DNA and RNA and are important for the genetic code. Thymine is a pyrimidine base found only in DNA, while uracil is a pyrimidine base found only in RNA.
Purine bases:
Purine bases are nitrogen-containing molecules that are formed by a pyrimidine ring fused to an imidazole ring. The purine bases are guanine and adenine.
Pyrimidine bases:
Pyrimidine bases consist of a single ring structure and include thymine, cytosine, and uracil.
Answer:
Therefore, the correct answer is option A and B, which are guanine and adenine. These bases are found in DNA and RNA and are important for the genetic code. Thymine is a pyrimidine base found only in DNA, while uracil is a pyrimidine base found only in RNA.
Structure of a disaccharide formed by glucose and fructose is given below. Identify anomeric carbon atoms in monosaccharide units.
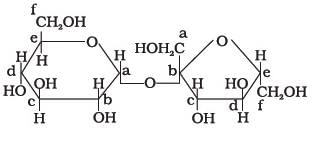
- a)‘a’ carbon of glucose and ‘a’ carbon of fructose.
- b)‘a’ carbon of glucose and ‘e’ carbon of fructose.
- c)‘a’ carbon of glucose and ‘b’ carbon of fructose.
- d)‘f ’ carbon of glucose and ‘f ’ carbon of fructose.
Correct answer is option 'C'. Can you explain this answer?
Structure of a disaccharide formed by glucose and fructose is given below. Identify anomeric carbon atoms in monosaccharide units.


a)
‘a’ carbon of glucose and ‘a’ carbon of fructose.
b)
‘a’ carbon of glucose and ‘e’ carbon of fructose.
c)
‘a’ carbon of glucose and ‘b’ carbon of fructose.
d)
‘f ’ carbon of glucose and ‘f ’ carbon of fructose.
 | Nirupama Kumari answered |
Ans - because in this case both are counted in Right Side (glucose in "a" & fructose in "b") but only single in figure fructose always counted in left side & glucose right side.
Amino acid are connected to each other by:- a)Hydrogen bond
- b)Carbon bond
- c)Polydiester bond
- d)Peptide bond
Correct answer is option 'D'. Can you explain this answer?
Amino acid are connected to each other by:
a)
Hydrogen bond
b)
Carbon bond
c)
Polydiester bond
d)
Peptide bond
| | Vijay Bansal answered |
A peptide bond is formed (-CONH) between the –NH2 group and the –COOH group of any two adjacent amino acids and it leads to the elimination of a water molecule. The resultant product formed is an amide.
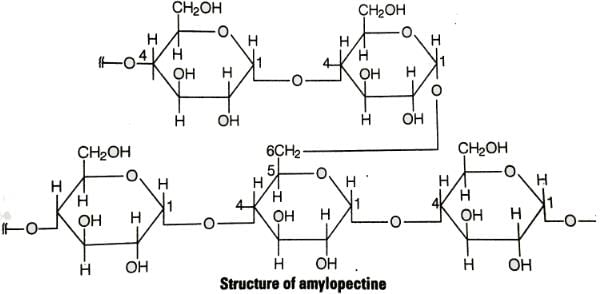
Glycogen is a branched chain polymer of α-D-glucose units in which chain is formed by C1—C4 glycosidic linkage whereas branching occurs by the formation of C1-C6 glycosidic linkage. Structure of glycogen is similar to ______.- a)Amylose
- b)Amylopectin
- c)Cellulose
- d)Glucose
Correct answer is option 'B'. Can you explain this answer?
Glycogen is a branched chain polymer of α-D-glucose units in which chain is formed by C1—C4 glycosidic linkage whereas branching occurs by the formation of C1-C6 glycosidic linkage. Structure of glycogen is similar to ______.
a)
Amylose
b)
Amylopectin
c)
Cellulose
d)
Glucose
| | Ritu Singh answered |
Glycogen is a branched chain polymer of αD glucose units in which chain is formed by C1−C4 glycosidic linkage whereas branchin occurs by the formation of C1−C6 glycosidic linkage. Structure of glycogen can be shown below similar to the sturcture amplopectin.

The following is a reaction of glucose with Fehling’s reagent. What will happen in the reaction?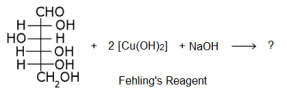
- a)Glucose will not react with Fehling’s reagent.
- b)Grey precipitate will be formed on warming glucose with Fehling’s reagent.
- c)Red precipitate will be formed on warming glucose with Fehling’s reagent.
- d)Saccharic acid will be formed when glucose will react with Fehling’s reagent.
Correct answer is option 'C'. Can you explain this answer?
The following is a reaction of glucose with Fehling’s reagent. What will happen in the reaction?
a)
Glucose will not react with Fehling’s reagent.
b)
Grey precipitate will be formed on warming glucose with Fehling’s reagent.
c)
Red precipitate will be formed on warming glucose with Fehling’s reagent.
d)
Saccharic acid will be formed when glucose will react with Fehling’s reagent.
 | Ciel Knowledge answered |
Because it is the chemical property of aliphatic aldehyde to give red precipitate with fehling solution.
RCHO + 2 CuO -----------> Cu2O. + RCOOH
Fehling reagents is aq. alkaline CuSO4 solution along with Rochelle salt Na-K tartrate.
And glucose molecule consists of aldehydic group at first position and hence it is also a aldehyde and thus give this characteristic test.
RCHO + 2 CuO -----------> Cu2O. + RCOOH
Fehling reagents is aq. alkaline CuSO4 solution along with Rochelle salt Na-K tartrate.
And glucose molecule consists of aldehydic group at first position and hence it is also a aldehyde and thus give this characteristic test.
Which of the following carbohydrates is called an invert sugar?- a)Sucrose
- b)Fructose
- c)Glucose
- d)Cellulose
Correct answer is option 'A'. Can you explain this answer?
Which of the following carbohydrates is called an invert sugar?
a)
Sucrose
b)
Fructose
c)
Glucose
d)
Cellulose
 | Sanjeevini Angadi answered |
It is called invert sugar because the angle of the specific rotation of the plain polarized light changes from a positive to a negative value due to the presence of the optical isomers of the mixture of glucose and fructosesugars.
Which of the following is a test for proteins?- a)Molish’s test
- b)Benedict’s test
- c)Biuret test
- d)Beilstein test
Correct answer is option 'C'. Can you explain this answer?
Which of the following is a test for proteins?
a)
Molish’s test
b)
Benedict’s test
c)
Biuret test
d)
Beilstein test
 | Savita Soni answered |
Yes biuret is a blue solution,when it reacts with protein ,its color become pink - purple. molish test & Benedict both are for carbohydrate where as beilstein is for halides
Dinucleotide is obtained by joining two nucleotides together by phosphodiester linkage. Between which carbon atoms of pentose sugars of nucleotides are these linkages present?- a)5′ and 3′
- b)1′ and 5′
- c)5′ and 5′
- d)3′ and 3′
Correct answer is option 'A'. Can you explain this answer?
Dinucleotide is obtained by joining two nucleotides together by phosphodiester linkage. Between which carbon atoms of pentose sugars of nucleotides are these linkages present?
a)
5′ and 3′
b)
1′ and 5′
c)
5′ and 5′
d)
3′ and 3′
| | Devanshi Mehta answered |
B)3
When more than ten amino acids react, then the product formed is called:- a)Polypeptide
- b)Hexapeptide
- c)Tripeptide
- d)Dipeptide
Correct answer is option 'A'. Can you explain this answer?
When more than ten amino acids react, then the product formed is called:
a)
Polypeptide
b)
Hexapeptide
c)
Tripeptide
d)
Dipeptide
| | Nabanita Pillai answered |
Polypeptide is the correct answer.
Explanation:
When more than ten amino acids react, the product formed is called a polypeptide. A polypeptide is a chain of amino acids joined together by peptide bonds. Peptide bonds are formed through a condensation reaction between the carboxyl group of one amino acid and the amino group of another amino acid, resulting in the release of a water molecule.
Key points:
- Polypeptides are formed by the polymerization of amino acids.
- Amino acids are organic compounds that contain an amino group (-NH2) and a carboxyl group (-COOH) attached to a central carbon atom.
- There are 20 different types of amino acids that can be found in proteins, each with a unique side chain or R-group.
- The order and sequence of amino acids in a polypeptide chain determine the structure and function of the protein.
- Polypeptides can vary in length, ranging from just a few amino acids to thousands of amino acids in length.
- Polypeptides can fold into specific three-dimensional structures, such as alpha helices and beta sheets, which are stabilized by various types of interactions between amino acid side chains.
- These folded structures are critical for the proper functioning of proteins and can determine their enzymatic activity, binding specificity, and overall stability.
- Polypeptides can also undergo post-translational modifications, such as phosphorylation or glycosylation, which can further modify their structure and function.
- Polypeptides can interact with other molecules, such as other polypeptides or small molecules, to form larger protein complexes or perform specific biological functions.
- Polypeptides are an essential component of all living organisms and play critical roles in processes such as metabolism, signaling, and immune response.
In conclusion, when more than ten amino acids react, the product formed is called a polypeptide, which is a chain of amino acids joined together by peptide bonds. Polypeptides are the building blocks of proteins and are essential for the structure and function of biological systems.
Explanation:
When more than ten amino acids react, the product formed is called a polypeptide. A polypeptide is a chain of amino acids joined together by peptide bonds. Peptide bonds are formed through a condensation reaction between the carboxyl group of one amino acid and the amino group of another amino acid, resulting in the release of a water molecule.
Key points:
- Polypeptides are formed by the polymerization of amino acids.
- Amino acids are organic compounds that contain an amino group (-NH2) and a carboxyl group (-COOH) attached to a central carbon atom.
- There are 20 different types of amino acids that can be found in proteins, each with a unique side chain or R-group.
- The order and sequence of amino acids in a polypeptide chain determine the structure and function of the protein.
- Polypeptides can vary in length, ranging from just a few amino acids to thousands of amino acids in length.
- Polypeptides can fold into specific three-dimensional structures, such as alpha helices and beta sheets, which are stabilized by various types of interactions between amino acid side chains.
- These folded structures are critical for the proper functioning of proteins and can determine their enzymatic activity, binding specificity, and overall stability.
- Polypeptides can also undergo post-translational modifications, such as phosphorylation or glycosylation, which can further modify their structure and function.
- Polypeptides can interact with other molecules, such as other polypeptides or small molecules, to form larger protein complexes or perform specific biological functions.
- Polypeptides are an essential component of all living organisms and play critical roles in processes such as metabolism, signaling, and immune response.
In conclusion, when more than ten amino acids react, the product formed is called a polypeptide, which is a chain of amino acids joined together by peptide bonds. Polypeptides are the building blocks of proteins and are essential for the structure and function of biological systems.
Which of the following B group vitamins can be stored in our body?- a)Vitamin B1
- b)Vitamin B2
- c)Vitamin B6
- d)Vitamin B12
Correct answer is option 'D'. Can you explain this answer?
Which of the following B group vitamins can be stored in our body?
a)
Vitamin B1
b)
Vitamin B2
c)
Vitamin B6
d)
Vitamin B12
| | Om Desai answered |
The correct answer is option D
Vitamin B12
The body has a limited capacity to store most of the B-group vitamins except B12 and folate, which are stored in the liver.
Vitamin B12
The body has a limited capacity to store most of the B-group vitamins except B12 and folate, which are stored in the liver.
What does the following reaction of glucose with HI elucidates about the structure of glucose?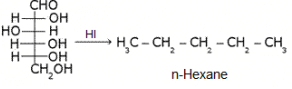
- a)Shows the presence of -CHO group
- b)Shows the presence of C=O group
- c)Shows the presence of six carbons linked linearly
- d)Shows the presence of ring structure of glucose
Correct answer is option 'C'. Can you explain this answer?
What does the following reaction of glucose with HI elucidates about the structure of glucose?
a)
Shows the presence of -CHO group
b)
Shows the presence of C=O group
c)
Shows the presence of six carbons linked linearly
d)
Shows the presence of ring structure of glucose
| | Shreya Singh answered |
It shows that glucose exists as n-hexane...i.e. it has straight linear chain...
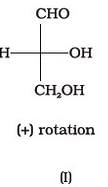
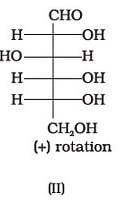
Optical rotations of some compounds along with their structures are given below which of them have D configuration.
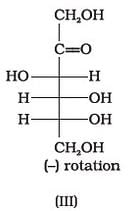
- a)I, II, III
- b)II, III
- c)I, II
- d)III
Correct answer is option 'A'. Can you explain this answer?
Optical rotations of some compounds along with their structures are given below which of them have D configuration.






a)
I, II, III
b)
II, III
c)
I, II
d)
III
 | Divey Sethi answered |
The correct answer is Option A.
I, II and III structures have D configuration with -OH group on the lowest asymmetric carbon is on the right side which is comparable to (+) glyceraldehyde.
I, II and III structures have D configuration with -OH group on the lowest asymmetric carbon is on the right side which is comparable to (+) glyceraldehyde.
Directions: This question consist of two statements, each printed as Assertion and Reason. While answering this question, you are required to choose any one of the following four responses.
Assertion : Vitamin D cannot be stored in our body
Reason : Vitamin D is fat soluble vitamin and is excreted from the body in urine- a)If both Assertion and Reason are correct and the Reason is a correct explanation of the Assertion.
- b)If both Assertion and Reason are correct but Reason is not a correct explanation of the Assertion.
- c)If the Assertion is correct but Reason is incorrect.
- d)If both the Assertion and Reason are incorrect.
Correct answer is option 'D'. Can you explain this answer?
Directions: This question consist of two statements, each printed as Assertion and Reason. While answering this question, you are required to choose any one of the following four responses.
Assertion : Vitamin D cannot be stored in our body
Reason : Vitamin D is fat soluble vitamin and is excreted from the body in urine
Assertion : Vitamin D cannot be stored in our body
Reason : Vitamin D is fat soluble vitamin and is excreted from the body in urine
a)
If both Assertion and Reason are correct and the Reason is a correct explanation of the Assertion.
b)
If both Assertion and Reason are correct but Reason is not a correct explanation of the Assertion.
c)
If the Assertion is correct but Reason is incorrect.
d)
If both the Assertion and Reason are incorrect.
| | Gaurav Kumar answered |
Vitamin D is a fat soluble vitamin and can be stored in the body since it is not excreted out of the body.
Chapter doubts & questions for Biomolecules - NCERT Based Tests for NEET 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Biomolecules - NCERT Based Tests for NEET in English & Hindi are available as part of NEET exam. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
Top Courses NEET
Related NEET Content
;
Signup to see your scores go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup