All Exams > SSS 3 > Chemistry for SSS 3 > All Questions
All questions of Organic Chemistry for SSS 3 Exam
Which among the following defines Meso forms of isomers?- a)Meso form is optically inactive due to external compensation.
- b)The molecules of the meso isomers are chiral.
- c)It can be separated into optically active enantiomeric pairs.
- d)It is a single compound.
Correct answer is option 'D'. Can you explain this answer?
Which among the following defines Meso forms of isomers?
a)
Meso form is optically inactive due to external compensation.
b)
The molecules of the meso isomers are chiral.
c)
It can be separated into optically active enantiomeric pairs.
d)
It is a single compound.
| | Rohan Singh answered |
Meso forms of isomers are single compounds and their molecules are achiral hence they cannot be separated into pairs.
Additional Information: A meso compound or meso isomer is a non-optically active member of a set of stereoisomers, at least two of which are optically active. This means that despite containing two or more stereogenic centers, the molecule is not chiral.

- a)3
- b)4
- c)5
- d)6
Correct answer is option 'D'. Can you explain this answer?
a)
3
b)
4
c)
5
d)
6
 | Knowledge Hub answered |
General formula of ketone = CnH2nO
12x+2x+16 = 100
x = 6
So, the formula of compound is C6H12O. We have to make ketone only. These are as follow
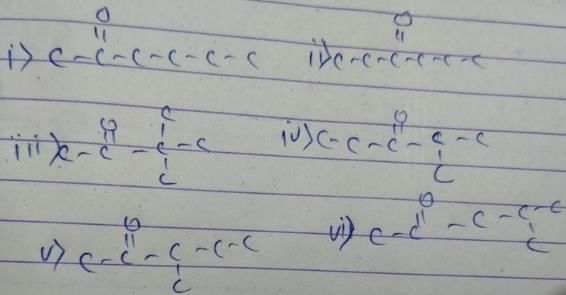
12x+2x+16 = 100
x = 6
So, the formula of compound is C6H12O. We have to make ketone only. These are as follow
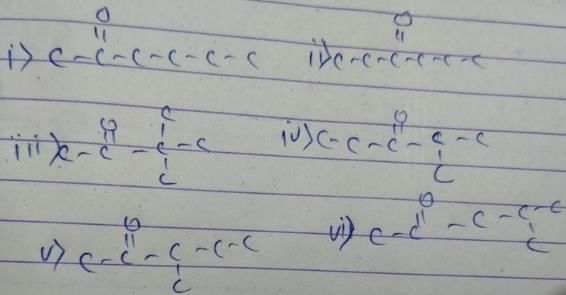
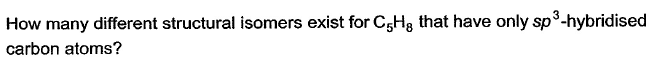
- a)3
- b)4
- c)5
- d)7
Correct answer is option 'A'. Can you explain this answer?
a)
3
b)
4
c)
5
d)
7
| | Riya Banerjee answered |
The compounds with each doubly bonded carbon attached to two different groups (like Cab=Cab, Cab=Ccd) exhibit geometrical isomerism i.e., cis and trans forms. The geometrical isomerism arises due to restricted rotation of double bond.
However, even though there is restricted rotation for triple bond, alkynes do not exhibit geometrical isomerism, since the triply bonded carbons are attached to one group each only.
The observed order of the stability of the cabocation is:- a)(CH3)2CH+ < (CH3)3C+ 3+ < CH3CH2+
- b)CH3+ < CH3CH2+ <(CH3)2CH+ < (CH3)3C+
- c)CH3CH2+ <(CH3)2CH+ < (CH3)3C+ < CH3+
- d)CH3+ < CH3CH2+ < (CH3)3C+ <(CH3)2CH+
Correct answer is option 'B'. Can you explain this answer?
The observed order of the stability of the cabocation is:
a)
(CH3)2CH+ < (CH3)3C+ 3+ < CH3CH2+
b)
CH3+ < CH3CH2+ <(CH3)2CH+ < (CH3)3C+
c)
CH3CH2+ <(CH3)2CH+ < (CH3)3C+ < CH3+
d)
CH3+ < CH3CH2+ < (CH3)3C+ <(CH3)2CH+
| | Rajesh Gupta answered |
Alkyl groups directly attached to the +vely charged carbon stabilize the carbocations due to inductive and hyperconjugation effects.
Inductive effect:
→ Stability of carbocation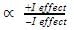
→ More number of +I group more stable carbocation.
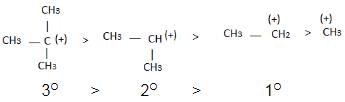
Hyperconjugation:
Inductive effect:
→ Stability of carbocation
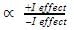
→ More number of +I group more stable carbocation.
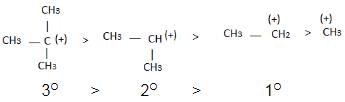
Hyperconjugation:
Stability
∝Number of canonical structures
∝Number of H (alpha hydrogen)
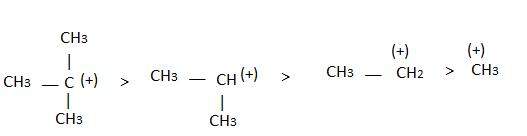
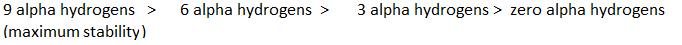
∝Number of canonical structures
∝Number of H (alpha hydrogen)
Which is true about conformers of 1,1,2,2-tetrachloroethane?Which is true about conformers of 1,1,2,2-tetrachloroethane?
a) The most stable conformer has dihedral angle of 60° between all adjacent chlorine atoms
b) In the least stable conformer, two Cl-atoms are eclipsing one another while other two Cl-atoms are eclipsed to hydrogen atoms
c) In the most stable conformer, dihedral angle between hydrogen atoms is 60°
d) The most stable conformer is non-polar - a)The most stable conformer has dihedral angle of 60° between all adjacent chlorine atoms
- b)In the least stable conformer, two Cl-atoms are eclipsing one another while other two Cl-atoms are eclipsed to hydrogen atoms
- c)In the most stable conformer, dihedral angle between hydrogen atoms is 60°
- d)The most stable conformer is non-polar
Correct answer is option 'D'. Can you explain this answer?
Which is true about conformers of 1,1,2,2-tetrachloroethane?
Which is true about conformers of 1,1,2,2-tetrachloroethane?
a) The most stable conformer has dihedral angle of 60° between all adjacent chlorine atoms
b) In the least stable conformer, two Cl-atoms are eclipsing one another while other two Cl-atoms are eclipsed to hydrogen atoms
c) In the most stable conformer, dihedral angle between hydrogen atoms is 60°
d) The most stable conformer is non-polar
a) The most stable conformer has dihedral angle of 60° between all adjacent chlorine atoms
b) In the least stable conformer, two Cl-atoms are eclipsing one another while other two Cl-atoms are eclipsed to hydrogen atoms
c) In the most stable conformer, dihedral angle between hydrogen atoms is 60°
d) The most stable conformer is non-polar
a)
The most stable conformer has dihedral angle of 60° between all adjacent chlorine atoms
b)
In the least stable conformer, two Cl-atoms are eclipsing one another while other two Cl-atoms are eclipsed to hydrogen atoms
c)
In the most stable conformer, dihedral angle between hydrogen atoms is 60°
d)
The most stable conformer is non-polar
| | Mira Joshi answered |
The correct answer is option D
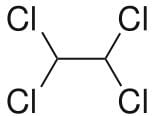
This molecule is non-polar two Cl atoms in one carbon atom cancel the polarity other two Cl atoms on the next carbon.
Also all the Cl atoms are at max distance from each other so max. Stability due to less repulsion between Cl atoms. Dihedral angle between H and Cl is 60°

This molecule is non-polar two Cl atoms in one carbon atom cancel the polarity other two Cl atoms on the next carbon.
Also all the Cl atoms are at max distance from each other so max. Stability due to less repulsion between Cl atoms. Dihedral angle between H and Cl is 60°

- a)3
- b)5
- c)7
- d)8
Correct answer is option 'C'. Can you explain this answer?
a)
3
b)
5
c)
7
d)
8
| | Neha Joshi answered |
The correct answer is Option B.
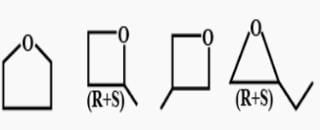
5 isomers are possible. C5H10(CnH2n). Molecules having the CnH2n formulas are most likely to be cyclic alkanes. The five isomers are:
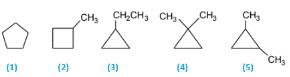
(1) Cyclopentane
(2) 1-Methylcyclobutane
(3) 1-Ethylcyclopropane
(4) 1,1-Dimethylcyclopropane
(5) 1,2-Dimethylcyclopropane
5 isomers are possible. C5H10(CnH2n). Molecules having the CnH2n formulas are most likely to be cyclic alkanes. The five isomers are:

(1) Cyclopentane
(2) 1-Methylcyclobutane
(3) 1-Ethylcyclopropane
(4) 1,1-Dimethylcyclopropane
(5) 1,2-Dimethylcyclopropane
- a)3
- b)4
- c)5
- d)6
Correct answer is option 'C'. Can you explain this answer?
a)
3
b)
4
c)
5
d)
6
| | Neha Joshi answered |
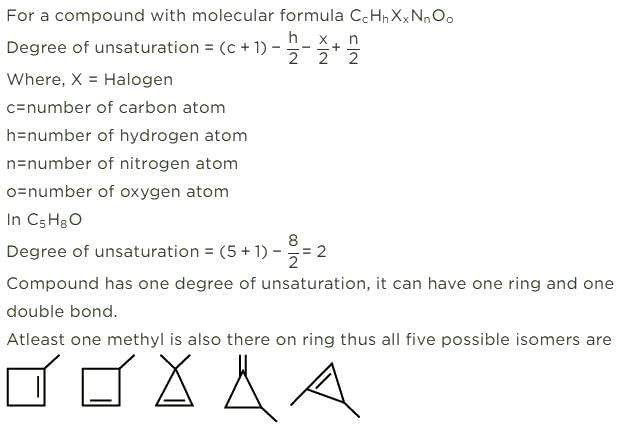
First, we will check the Double bond equivalent. In C7H8O, DBE = 4,this means that we will have 3 double bonds and a ring and no additional double bond outside the ring.
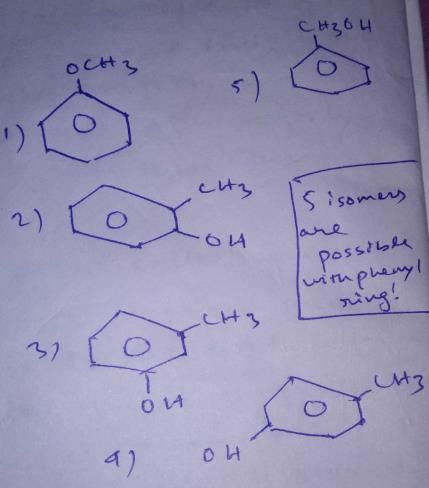
These are the structural isomers that exists for C7H8O

These are the structural isomers that exists for C7H8O
The isomer of ethanol is:- a)Dimethyl ether
- b)Diethyl ether
- c)Methanol
- d)Acetone
Correct answer is option 'A'. Can you explain this answer?
The isomer of ethanol is:
a)
Dimethyl ether
b)
Diethyl ether
c)
Methanol
d)
Acetone
| | Pranavi Kulkarni answered |
The isomer of ethanol(CH3-CH2-OH) is dimethyl ether(CH3-O-CH3). This is a functional isomer of ethanol.
Explanation:
- Functional isomers are the ones with the same molecular formula but different functional groups.
- The molecular formula for dimethyl ether is C2H6O similar to ethanol.
- Dimethyl ether has the structural formula CH3OCH3 and ethanol has the structural formula CH3CH2OH.
- Therefore, dimethyl ether is the functional isomer of ethanol as both have the same molecular formula but one is ether and the other is alcohol.
What is the minimum number of carbon atoms of an alkane must have to form an isomer?- a)4
- b)3
- c)2
- d)1
Correct answer is option 'A'. Can you explain this answer?
What is the minimum number of carbon atoms of an alkane must have to form an isomer?
a)
4
b)
3
c)
2
d)
1
| | Ananya Das answered |
An alkane with minimum four carbon atoms can show isomerism.
 [JEE Main 2014 Online Exam]
[JEE Main 2014 Online Exam]- a)a
- b)b
- c)c
- d)d
Correct answer is option 'B'. Can you explain this answer?
[JEE Main 2014 Online Exam]
a)
a
b)
b
c)
c
d)
d
| | Siddharth Mehra answered |
The correct answer is option 'B' - sp2 and sp.
Explanation:
In allene (C3H4), there are two carbon atoms, each with three sigma bonds. To determine the hybridization of the carbon atoms, we need to look at their bonding and lone pairs of electrons.
1. Hybridization of the first carbon atom:
- The first carbon atom is bonded to two other carbon atoms and one hydrogen atom.
- It has three sigma bonds, which means it will have three hybrid orbitals.
- The carbon atom also has one lone pair of electrons.
- The presence of three sigma bonds and one lone pair indicates sp2 hybridization.
- Therefore, the first carbon atom is sp2 hybridized.
2. Hybridization of the second carbon atom:
- The second carbon atom is also bonded to two other carbon atoms and one hydrogen atom.
- It also has three sigma bonds, which means it will have three hybrid orbitals.
- The carbon atom does not have any lone pairs of electrons.
- The presence of only three sigma bonds indicates sp hybridization.
- Therefore, the second carbon atom is sp hybridized.
In summary, in allene (C3H4), the type of hybridization of the carbon atoms is sp2 and sp.
Explanation:
In allene (C3H4), there are two carbon atoms, each with three sigma bonds. To determine the hybridization of the carbon atoms, we need to look at their bonding and lone pairs of electrons.
1. Hybridization of the first carbon atom:
- The first carbon atom is bonded to two other carbon atoms and one hydrogen atom.
- It has three sigma bonds, which means it will have three hybrid orbitals.
- The carbon atom also has one lone pair of electrons.
- The presence of three sigma bonds and one lone pair indicates sp2 hybridization.
- Therefore, the first carbon atom is sp2 hybridized.
2. Hybridization of the second carbon atom:
- The second carbon atom is also bonded to two other carbon atoms and one hydrogen atom.
- It also has three sigma bonds, which means it will have three hybrid orbitals.
- The carbon atom does not have any lone pairs of electrons.
- The presence of only three sigma bonds indicates sp hybridization.
- Therefore, the second carbon atom is sp hybridized.
In summary, in allene (C3H4), the type of hybridization of the carbon atoms is sp2 and sp.
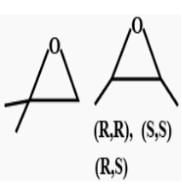
Consider all possible isomeric ketones, including stereoisomers of Molecular weight 100. All these isomers are independently reacted with NaBH4
(Note stereoisomers are also reacted separately). The total number of ketones that give a racemic product(s) is/are
[JEE Advanced 2014] - a)3
- b)4
- c)5
- d)6
Correct answer is option 'C'. Can you explain this answer?
Consider all possible isomeric ketones, including stereoisomers of Molecular weight 100. All these isomers are independently reacted with NaBH4
(Note stereoisomers are also reacted separately). The total number of ketones that give a racemic product(s) is/are
(Note stereoisomers are also reacted separately). The total number of ketones that give a racemic product(s) is/are
[JEE Advanced 2014]
a)
3
b)
4
c)
5
d)
6
 | Infinity Academy answered |
The general formula of isomeric ketone having molecular mass 100 is C6H12O [6×12+12×1+16].
The possible structure will be :
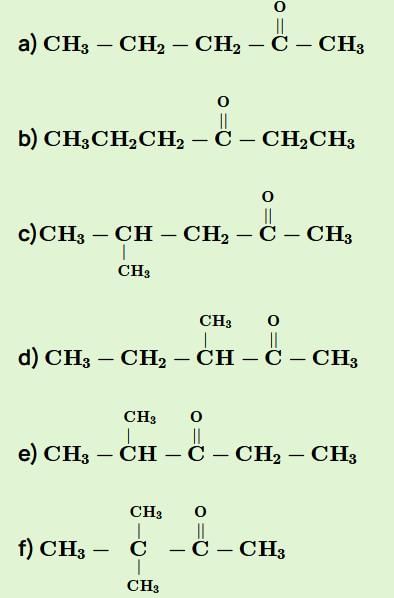
The number of ketones that gives racemic mixture with NaBH4 is 5 as the ketone with chiral center will give diastereoisomer with NaBH4
How many structural isomers are possible with molecular formula C4H10O ?- a)3
- b)5
- c)7
- d)8
Correct answer is option 'C'. Can you explain this answer?
How many structural isomers are possible with molecular formula C4H10O ?
a)
3
b)
5
c)
7
d)
8
| | Preeti Iyer answered |
The formula that these isomers contain no rings or double bonds. The isomers must be alcohols and ethers.
Let's start by drawing the alcohols.
Start with four carbons in a row and put an OH group in every possible position.
This gives us
1. CH3CH2CH2CH2OH, butan 1-ol
and
2. CH3CH2CH(OH)CH3, butan-2-ol
Now use a 3-carbon chain with a CH3 on the middle carbon.
This gives
(CH3)2CHCH2OH, 2-methylpropan-1-ol
and
(CH3)3COH, 2-methylpropan-2-ol.
Now for the ethers.
Let's start with five atoms in a row.
This gives
CH3CH2CH2OCH3, 1-methoxypropane
and
CH3CH2OCH2CH3 , ethoxyethane
Finally, the only choice for a branched-chain ether is
(CH3)2CHOCH3 , 2-methoxpropane.
And there are your seven isomers of C4H10O.
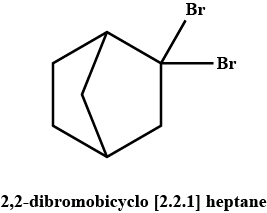
A pair of enantiomers is possible for _______ isomer of 2,2-dibromobicyclo [2.2.1] heptane.- a)cis
- b)trans
- c)both cis and trans
- d)none of the above
Correct answer is option 'A'. Can you explain this answer?
A pair of enantiomers is possible for _______ isomer of 2,2-dibromobicyclo [2.2.1] heptane.
a)
cis
b)
trans
c)
both cis and trans
d)
none of the above
| | Tejas Verma answered |
Only one pair of enantiomers is possible for cis-2,2-dibromobicyclo [2.2.1] heptane. The trans arrangement of one carbon bridge is structurally impossible. Such a molecule would have too much strain.
Direction (Q. Nos. 1-8) This section contains 8 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Q.how many structural isomers (aldehyde + ketone) are possible for C5H10O ?- a)3
- b)4
- c)6
- d)7
Correct answer is option 'D'. Can you explain this answer?
Direction (Q. Nos. 1-8) This section contains 8 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Q.
how many structural isomers (aldehyde + ketone) are possible for C5H10O ?
a)
3
b)
4
c)
6
d)
7
| | Hansa Sharma answered |
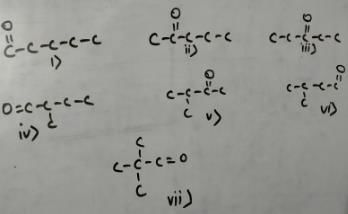
Direction (Q. Nos. 1 - 9) This section contains 9 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Q.
- a)3
- b)5
- c)7
- d)9
Correct answer is option 'C'. Can you explain this answer?
Direction (Q. Nos. 1 - 9) This section contains 9 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Q.
a)
3
b)
5
c)
7
d)
9
 | Kamna Science Academy answered |
The number of optically active isomers = 2n−1−2n−1/2 =23−1−23−1/2 =2.
The number of optical isomers possible for 2, 3-pentanediol is:- a)2
- b)3
- c)4
- d)5
Correct answer is option 'C'. Can you explain this answer?
The number of optical isomers possible for 2, 3-pentanediol is:
a)
2
b)
3
c)
4
d)
5
| | Preeti Iyer answered |
2 optical centers.
Total optically active isomers =22
=4
Total optically active isomers =22
=4
C3H6O represents an aldehyde and a ketone, is a type of:- a)Chain isomerism
- b)Functional group isomerism
- c)Metamerism
- d)Position isomerism
Correct answer is option 'B'. Can you explain this answer?
C3H6O represents an aldehyde and a ketone, is a type of:
a)
Chain isomerism
b)
Functional group isomerism
c)
Metamerism
d)
Position isomerism
| | Preeti Iyer answered |
The aldehyde and ketone that can be represented by molecular formula C3H6O are propanal and propanone respectively.
Aldehyde - Propanal - CH3CH2CHO
Ketone - Propanone - CH3COCH3
These two carbon compounds are called functional isomers. They do not have the same physical properties since their functional group is different.
When hydrogen is present on one side of C=O group and other side is R, (RCHO), then it is an aldehyde group. When R is present on both sides of C=O group, then it is a ketone.
Heterolytic cleavage is a way to cleave the:- a)Non-ionic bonds
- b)Ionic bonds
- c)Covalent bonds
- d)Polar bonds
Correct answer is option 'C'. Can you explain this answer?
Heterolytic cleavage is a way to cleave the:
a)
Non-ionic bonds
b)
Ionic bonds
c)
Covalent bonds
d)
Polar bonds
| | Rahul Bansal answered |
In heterolytic cleavage, a covalent bond breaks in such a way that one fragment gets both of the shared electrons. In homolytic cleavage, a covalent bond breaks in such a way that each fragment gets one of the shared electrons. The word heterolytic comes from the Greek heteros, "different", and lysis, "loosening".
Which of the following statements are correct?I. A pair of positional isomers differs in the position of the same functional group.
II. A pair of. structural isomers have the same relative molar mass.
Ill. A pair of functional group isomers belongs to different homologous series. - a)I, II and Ill
- b)I and Ill
- c)II and III
- d)I and II
Correct answer is option 'A'. Can you explain this answer?
Which of the following statements are correct?
I. A pair of positional isomers differs in the position of the same functional group.
II. A pair of. structural isomers have the same relative molar mass.
Ill. A pair of functional group isomers belongs to different homologous series.
II. A pair of. structural isomers have the same relative molar mass.
Ill. A pair of functional group isomers belongs to different homologous series.
a)
I, II and Ill
b)
I and Ill
c)
II and III
d)
I and II
| | Preeti Iyer answered |
According to me all statements are correct. For statement I, positional isomers have the same functional group. They just differ in the position of that group.
Statenmen II- Structural isomers have the same relative molar mass. Actually relative mass means the mass of one isomer is samee relative to another. Structural isomers differ in the structure of molecules. However they have the same molecular formula and hence; same molar mass.
Statement III- According to me, this statement is correct. As homologous series must contain the same functional group with the same physical & chemical properties. Also functional group isomers differ in the functional group attached with them. So, functional group isomers belong to different homologous series.
Statenmen II- Structural isomers have the same relative molar mass. Actually relative mass means the mass of one isomer is samee relative to another. Structural isomers differ in the structure of molecules. However they have the same molecular formula and hence; same molar mass.
Statement III- According to me, this statement is correct. As homologous series must contain the same functional group with the same physical & chemical properties. Also functional group isomers differ in the functional group attached with them. So, functional group isomers belong to different homologous series.
Which is not true regarding conformers of ethane?- a)Theoretically infinite conformers exist
- b)Staggered conformer has lower torsional strain than eclipsed one
- c)Increasing temperature increases the percentage of eclipsed conformer
- d)By precise experimental setup, staggered conformer can be separated out of system
Correct answer is option 'D'. Can you explain this answer?
Which is not true regarding conformers of ethane?
a)
Theoretically infinite conformers exist
b)
Staggered conformer has lower torsional strain than eclipsed one
c)
Increasing temperature increases the percentage of eclipsed conformer
d)
By precise experimental setup, staggered conformer can be separated out of system
| | Neha Joshi answered |
The correct answer is Option D.
Although conformers differ in potential energy and stability, the difference is so small that it does not allow their practical separation.
Although conformers differ in potential energy and stability, the difference is so small that it does not allow their practical separation.
An organic acid containing carbon, oxygen and hydrogen only has molar mass of 132 g. 6.6 g of this acid require 80 mL 1.25 M NaOH for complete neutralisation. How many carboxylic groups are present in one molecule of this acid? - a)1
- b)2
- c)3
- d)4
Correct answer is option 'B'. Can you explain this answer?
An organic acid containing carbon, oxygen and hydrogen only has molar mass of 132 g. 6.6 g of this acid require 80 mL 1.25 M NaOH for complete neutralisation. How many carboxylic groups are present in one molecule of this acid?
a)
1
b)
2
c)
3
d)
4
| | Krishna Iyer answered |
Let us consider organic acid as ‘A’.
Moles of A = mass of A/Molar mass = 6.6/132 = 0.05
Molarity of NaOH = 1.25M
And volume of NaOH = 80 mL
Therefore, moles of NaOH = molarity × Volume
= 1.25×80×10-3 = 0.1 moles.
SO, 0.05 moles of A requires 0.1 moles of NaOH
1 mole of acid requires x moles of NaOH
x = 0.1/0.05×1 = 2
So for neutralisation of 1 mole of A, we need 2 moles of NaOH. Therefore given acid is dibasic and contains 2 -COOH groups.
Moles of A = mass of A/Molar mass = 6.6/132 = 0.05
Molarity of NaOH = 1.25M
And volume of NaOH = 80 mL
Therefore, moles of NaOH = molarity × Volume
= 1.25×80×10-3 = 0.1 moles.
SO, 0.05 moles of A requires 0.1 moles of NaOH
1 mole of acid requires x moles of NaOH
x = 0.1/0.05×1 = 2
So for neutralisation of 1 mole of A, we need 2 moles of NaOH. Therefore given acid is dibasic and contains 2 -COOH groups.
Which of the following are not functional isomers of each other?- a)CH3CH2NO2 and CH3CH2ON=O
- b)C2H5CHO and CH3COCH3
- c)CH3CH2NH2 and CH3NHCH3
- d)C3H7NH2 and (CH3)2CHNH2
Correct answer is option 'D'. Can you explain this answer?
Which of the following are not functional isomers of each other?
a)
CH3CH2NO2 and CH3CH2ON=O
b)
C2H5CHO and CH3COCH3
c)
CH3CH2NH2 and CH3NHCH3
d)
C3H7NH2 and (CH3)2CHNH2
| | Anjali Sharma answered |
Functional isomers are structural isomers that have the same molecular formula (that is, the same number of atoms of the same elements), but the atoms are connected in different ways so that the groupings are dissimilar or different functional groups.
10 amine to different 10 amines are not functional isomers.
Hence, option ′d′ is the answer.

The number of isomers of C6H14 is:- a)4
- b)5
- c)6
- d)7
Correct answer is option 'B'. Can you explain this answer?
The number of isomers of C6H14 is:
a)
4
b)
5
c)
6
d)
7
| | Anu Mukherjee answered |
**Explanation:**
To determine the number of isomers of C6H14, we need to consider the different ways in which carbon atoms can be arranged and the different ways in which hydrogen atoms can be distributed among these carbon atoms.
**1. Straight-chain isomers:**
The simplest isomer is the straight chain, where the carbon atoms are arranged in a linear sequence. For C6H14, there is only one straight-chain isomer.
**2. Branched-chain isomers:**
In branched-chain isomers, the carbon atoms are arranged in a branched manner. To determine the number of possible branched-chain isomers, we need to consider the different ways in which the branches can be arranged and the different positions at which the branches can be attached to the main chain.
In this case, one of the carbons in the straight chain can have three branches attached to it. We can have three different arrangements of these branches, which are:
- One branch attached to the first carbon and two branches attached to the second carbon.
- Two branches attached to the first carbon and one branch attached to the second carbon.
- Three branches attached to the first carbon and no branches attached to the second carbon.
For each of these arrangements, there are different possibilities for the positions of the branches along the main chain. Therefore, for the branched-chain isomers, there are a total of three possibilities.
**3. Cycloalkane isomers:**
Cycloalkanes are ring structures where the carbon atoms are arranged in a closed loop. For C6H14, we can have cycloalkane isomers with different ring sizes.
- Cyclohexane: In this case, all six carbon atoms form a single ring. There is only one possibility.
- Cyclopentane: In this case, five carbon atoms form a ring and the remaining carbon atom is attached to one of the carbons in the ring. There is only one possibility.
Therefore, the total number of isomers of C6H14 is the sum of the straight-chain isomer, branched-chain isomers, and cycloalkane isomers, which is 1 + 3 + 2 = 6.
Therefore, the correct answer is option **c) 6**.
To determine the number of isomers of C6H14, we need to consider the different ways in which carbon atoms can be arranged and the different ways in which hydrogen atoms can be distributed among these carbon atoms.
**1. Straight-chain isomers:**
The simplest isomer is the straight chain, where the carbon atoms are arranged in a linear sequence. For C6H14, there is only one straight-chain isomer.
**2. Branched-chain isomers:**
In branched-chain isomers, the carbon atoms are arranged in a branched manner. To determine the number of possible branched-chain isomers, we need to consider the different ways in which the branches can be arranged and the different positions at which the branches can be attached to the main chain.
In this case, one of the carbons in the straight chain can have three branches attached to it. We can have three different arrangements of these branches, which are:
- One branch attached to the first carbon and two branches attached to the second carbon.
- Two branches attached to the first carbon and one branch attached to the second carbon.
- Three branches attached to the first carbon and no branches attached to the second carbon.
For each of these arrangements, there are different possibilities for the positions of the branches along the main chain. Therefore, for the branched-chain isomers, there are a total of three possibilities.
**3. Cycloalkane isomers:**
Cycloalkanes are ring structures where the carbon atoms are arranged in a closed loop. For C6H14, we can have cycloalkane isomers with different ring sizes.
- Cyclohexane: In this case, all six carbon atoms form a single ring. There is only one possibility.
- Cyclopentane: In this case, five carbon atoms form a ring and the remaining carbon atom is attached to one of the carbons in the ring. There is only one possibility.
Therefore, the total number of isomers of C6H14 is the sum of the straight-chain isomer, branched-chain isomers, and cycloalkane isomers, which is 1 + 3 + 2 = 6.
Therefore, the correct answer is option **c) 6**.
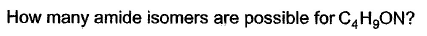
- a)4
- b)5
- c)6
- d)8
Correct answer is option 'D'. Can you explain this answer?
a)
4
b)
5
c)
6
d)
8
 | Knowledge Hub answered |
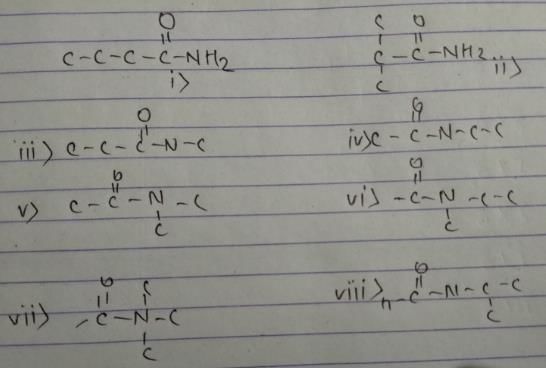
These are 8 isomers of C4H9ON.
 [JEE Main 2013 Online Exam]
[JEE Main 2013 Online Exam]- a)a
- b)b
- c)c
- d)d
Correct answer is option 'C'. Can you explain this answer?
[JEE Main 2013 Online Exam]
a)
a
b)
b
c)
c
d)
d
| | Om Desai answered |
In ethers, alcohols and amines, there is no double bond. However, esters have one double bond as same as monocarboxylic acid. So monocarboxylic acid is a functional isomer of ester (only for no. of carbon greater than 2. As we need to have at least 3 carbon atoms to form ester.)

- a)a
- b)b
- c)c
- d)d
Correct answer is option 'B'. Can you explain this answer?
a)
a
b)
b
c)
c
d)
d
| | Om Desai answered |
Double bond equivalent of C5H10O is 1. So, it will have either a double bond or a ring.So, it may be an aldehyde, ketone, alcohol( carbon chain having double bond) or epoxide.
Propyl ethanoate has 2 oxygen atoms. So, it will never be its isomer. HOwever option a and c will be its isomer as they are aldehyde.
Propyl ethanoate has 2 oxygen atoms. So, it will never be its isomer. HOwever option a and c will be its isomer as they are aldehyde.
Direction (Q. Nos. 1-18) This section contains 18 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.Q.
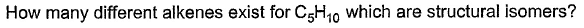
- a)3
- b)5
- c)6
- d)7
Correct answer is option 'B'. Can you explain this answer?
Direction (Q. Nos. 1-18) This section contains 18 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Q.
a)
3
b)
5
c)
6
d)
7
| | Naina Sharma answered |
C5 H10
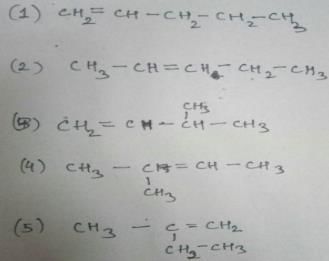
Two times double bond can’t appear as it has asked alkene and not alkadiene. So, these are the only types of alkenes that can be formed which are structural isomers.
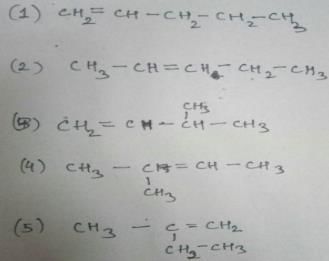
Two times double bond can’t appear as it has asked alkene and not alkadiene. So, these are the only types of alkenes that can be formed which are structural isomers.
Which,of the following correctly lists the conformations of cyclohexane in order of increasing potential energies?- a)Chair < Boat < Twist boat < Half-chair
- b)Half-chair < Boat < Twist boat < Chair
- c)Chair < Twist boat < Half-chair < Boat
- d)Chair < Twist boat < Boat < Half-chair
Correct answer is option 'D'. Can you explain this answer?
Which,of the following correctly lists the conformations of cyclohexane in order of increasing potential energies?
a)
Chair < Boat < Twist boat < Half-chair
b)
Half-chair < Boat < Twist boat < Chair
c)
Chair < Twist boat < Half-chair < Boat
d)
Chair < Twist boat < Boat < Half-chair
| | Riya Banerjee answered |
Correct answer is option D
Chair >Twist boat > Boat > Half-chair
above is the stability order of
the conformed.
Stability is inversely proportional to potential energy.
the conformed.
Stability is inversely proportional to potential energy.
Which of the following correctly ranks the cycloalkanes in order of increasing ring strain per methylene group? - a)Cyclopropane < Cyclobutane < Cyclopentane < Cyclohexane
- b)Cyclohexane < Cyclopentane < Cyclobutane < Cyclopropane
- c)Cyclohexane < Cyclobutane < Cyclopentane < Cyclopropane
- d)Cyclopropane < Cyclopentane < Cyclobutane < Cyclohexane
Correct answer is option 'B'. Can you explain this answer?
Which of the following correctly ranks the cycloalkanes in order of increasing ring strain per methylene group?
a)
Cyclopropane < Cyclobutane < Cyclopentane < Cyclohexane
b)
Cyclohexane < Cyclopentane < Cyclobutane < Cyclopropane
c)
Cyclohexane < Cyclobutane < Cyclopentane < Cyclopropane
d)
Cyclopropane < Cyclopentane < Cyclobutane < Cyclohexane
| | Rajesh Gupta answered |
The correct answer is Option B.
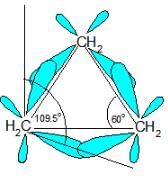
The C-C-C bond angles in cyclopropane (60o) and cyclobutane (90o) are much different than the ideal bond angle of 109.5o.This bond angle causes cyclopropane and cyclobutane to have a high ring strain. However, molecules, such as cyclohexane and cyclopentane, would have a much lower ring strain because the bond angle between the carbons is much closer to 109.5o.

The C-C-C bond angles in cyclopropane (60o) and cyclobutane (90o) are much different than the ideal bond angle of 109.5o.This bond angle causes cyclopropane and cyclobutane to have a high ring strain. However, molecules, such as cyclohexane and cyclopentane, would have a much lower ring strain because the bond angle between the carbons is much closer to 109.5o.
The organic reaction which proceeds through heterolytic bond cleavage are known as:- a)Covalent reactions
- b)Ionic reactions
- c)Free radical reaction
- d)Polar reactions
Correct answer is option 'B'. Can you explain this answer?
The organic reaction which proceeds through heterolytic bond cleavage are known as:
a)
Covalent reactions
b)
Ionic reactions
c)
Free radical reaction
d)
Polar reactions
| | Hansa Sharma answered |
In heterolytic bond cleavage the bond breaks unevenly and the shared pair of electrons is accommodated by one of the products, which produces one or more ions.As heterolytic bond cleavage gives ions. So the reaction which proceeds through heterolytic bond cleavage is an ionic reaction.
Hence B is the correct answer.
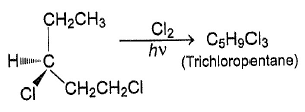
How many positional isomers exist for C3H5Cl3 ?
Correct answer is '5'. Can you explain this answer?
How many positional isomers exist for C3H5Cl3 ?
| | Nandini Iyer answered |
Explanation:
1,1,1-trichloropropane;
1,2,3-trichloropropane;
1,2,2-trichloropropane;
1,1,2-trichloropropane; this has 2 stereoisomers, with the chiral centre at C2;
1,1,3-trichloropropane;
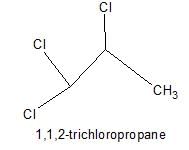

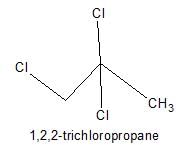
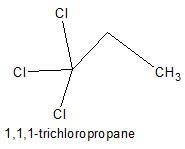
What is wrong about enantiomers of 2-chloropropanoic acid?- a)Have same solubility in water
- b)Have same pKa value
- c)Have same refractive indices
- d)Have same rate of reactions with (+)-2-butanol
Correct answer is option 'D'. Can you explain this answer?
What is wrong about enantiomers of 2-chloropropanoic acid?
a)
Have same solubility in water
b)
Have same pKa value
c)
Have same refractive indices
d)
Have same rate of reactions with (+)-2-butanol
| | Preeti Iyer answered |
The correct answer is option D.
Pair of enantiomers react differently with pure enantiomers of other compounds.
Pair of enantiomers react differently with pure enantiomers of other compounds.
A sequential account of each step, describing details of electron movement, energetics during bond cleavage and bond formation, and the rates of transformation of reactants into products (kinetics) is referred to as:- a)Reaction mechanism
- b)Reaction kinetics
- c)Thermodynamics
- d)Equation
Correct answer is option 'A'. Can you explain this answer?
A sequential account of each step, describing details of electron movement, energetics during bond cleavage and bond formation, and the rates of transformation of reactants into products (kinetics) is referred to as:
a)
Reaction mechanism
b)
Reaction kinetics
c)
Thermodynamics
d)
Equation
 | Rajesh Nirfarake answered |
Reaction mechanism is a stepwise description of each and every step involved in reaction so the correct option is A
Which of the following compounds will exhibit cis-trans isomerism? - a)2-butene
- b)2-butyne
- c)2-butanol
- d)butanal
Correct answer is option 'A'. Can you explain this answer?
Which of the following compounds will exhibit cis-trans isomerism?
a)
2-butene
b)
2-butyne
c)
2-butanol
d)
butanal
| | Gaurav Kumar answered |
The correct answer is Option A.
2-butene can show geometrical isomerism, due to different atoms/group of atoms attached on both sides of double bonded carbons as H and CH3 − groups.

2-butene can show geometrical isomerism, due to different atoms/group of atoms attached on both sides of double bonded carbons as H and CH3 − groups.


- a)a
- b)b
- c)c
- d)d
Correct answer is option 'A,B,C'. Can you explain this answer?
a)
a
b)
b
c)
c
d)
d
| | Hansa Sharma answered |
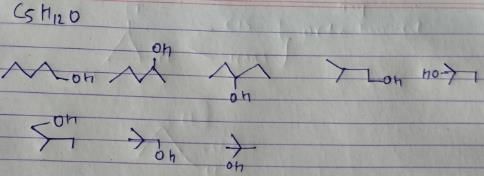
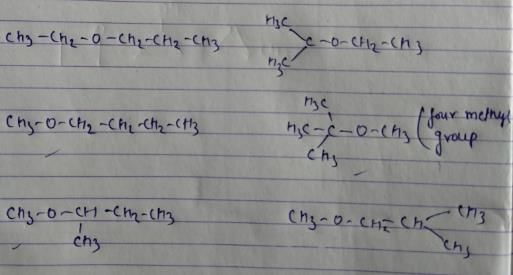
All the 8 isomers of alcohol are intermolecular H-bonding.
There are 6 ethers which have H-bond acceptor(i.e O atm) but not H-bond donor(i.e H atom)
Also, the isomer with 4 methyl groups is mentioned above.
According to me, CH3CH2COCH2CH3 is metamer of CH3COCH2CH2CH3 OR CH3COCH(CH3)2
So all options are correct.
Which among the following is a very unstable and reactive species:- a)Carbaanion
- b)Polar ions
- c)Carbocation
- d)Free radical
Correct answer is option 'C'. Can you explain this answer?
Which among the following is a very unstable and reactive species:
a)
Carbaanion
b)
Polar ions
c)
Carbocation
d)
Free radical
 | Geethika Reddy answered |
Actually carbanions are filled with octet, so they are stable and less reactive, in case of polar ions already they are stable, and free radicle is heptet i.e, near to octet whereas carbocation is sextet in nature, so in order to gain to octet it is more reactive and less stable
A hydrocarbon X is optically. X upon hydrogenation gives an optically inactive alkane Y. Which of the following pair of compounds can be X and Y respectively?- a)4-methyl-2-hexene and 3-methyl hexane
- b)3-methyl-1-pentene and 3-methyl pentane
- c) 4-methyl cyclopentene and methyl cyclopentane
- d)2-methyl-1-butene and 2-methyl butane
Correct answer is option 'B'. Can you explain this answer?
A hydrocarbon X is optically. X upon hydrogenation gives an optically inactive alkane Y. Which of the following pair of compounds can be X and Y respectively?
a)
4-methyl-2-hexene and 3-methyl hexane
b)
3-methyl-1-pentene and 3-methyl pentane
c)
4-methyl cyclopentene and methyl cyclopentane
d)
2-methyl-1-butene and 2-methyl butane
| | Pooja Shah answered |
The correct answer is Option B.
The optically active hydrocarbon X is 3-methyl-1-pentene CH2=CH−CH(CH3)CH2CH3. On catalytic hydrogenation, it forms 3-methyl pentane CH3CH2CH(CH3)CH2CH3, which is optically inactive.
The optically active hydrocarbon X is 3-methyl-1-pentene CH2=CH−CH(CH3)CH2CH3. On catalytic hydrogenation, it forms 3-methyl pentane CH3CH2CH(CH3)CH2CH3, which is optically inactive.
The minimum number of C atoms required for a hydrocarbon to exhibit optical isomerism:- a)4
- b)5
- c)6
- d)7
Correct answer is option 'D'. Can you explain this answer?
The minimum number of C atoms required for a hydrocarbon to exhibit optical isomerism:
a)
4
b)
5
c)
6
d)
7
| | Varun Kapoor answered |
The minimum number of C atoms required for a hydrocarbon to exhibit optical isomerism = 7

How many different stereoisomers exist for 1-chloro-2-(3-chlorocyclobutane) ethene?a)2b)4c)5d)7Correct answer is option 'B'. Can you explain this answer?
| | Om Desai answered |
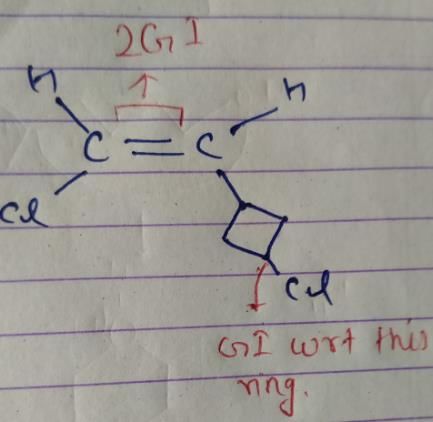
So, there are 4 isomers of given compound.
Which among the following does not exhibit geometric isomerism- a)1-hexene
- b)2-hexene
- c)3-hexene
- d)4-hexene
Correct answer is option 'A'. Can you explain this answer?
Which among the following does not exhibit geometric isomerism
a)
1-hexene
b)
2-hexene
c)
3-hexene
d)
4-hexene
 | Abhiram Choudhary answered |
Alkenes like 1-hexene when flipped from top to bottom they have identical structures and also they have C=CH2 unit which does not exist as cis- trans isomers.
Direction (Q. Nos. 15-18) This section contains 4 questions. when worked out will result in an integer from 0 to 9 (both inclusive)Q.
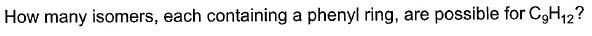
Correct answer is '8'. Can you explain this answer?
Direction (Q. Nos. 15-18) This section contains 4 questions. when worked out will result in an integer from 0 to 9 (both inclusive)
Q.
| | Geetika Shah answered |
Double bond equivalent = 20-12/4 = 4
For phenyl ring, Double bond equivalent = 4(3 double bond and 1 ring)
So, we won’t have any double bond outside the ring.
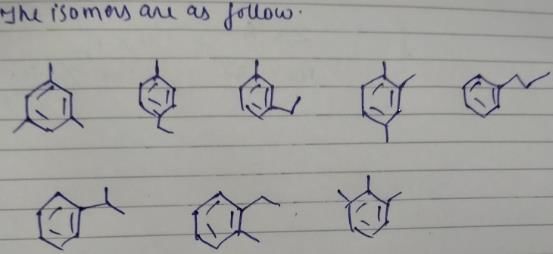
For phenyl ring, Double bond equivalent = 4(3 double bond and 1 ring)
So, we won’t have any double bond outside the ring.

What is/are correct deduction regarding compounds with their composition C3H60?- a)It can be a ketone
- b)It can be an ether without any pi-bond
- c)It can be an alcohol without any pi-bond
- d)It can be an unsaturated ether
Correct answer is option 'A,B,C,D'. Can you explain this answer?
What is/are correct deduction regarding compounds with their composition C3H60?
a)
It can be a ketone
b)
It can be an ether without any pi-bond
c)
It can be an alcohol without any pi-bond
d)
It can be an unsaturated ether
| | Upasana Roy answered |
Compounds with the composition C3H6O can exhibit different functional groups such as ketones, ethers, and alcohols. Let's explore each option in detail:
a) It can be a ketone:
A ketone is a type of organic compound that contains a carbonyl group (C=O) bonded to two carbon atoms. In the given composition C3H6O, if one of the carbons is double-bonded to an oxygen atom, it forms a ketone. The remaining three carbons and six hydrogens can be arranged in various ways to form different ketones.
b) It can be an ether without any pi-bond:
An ether is an organic compound that has an oxygen atom bonded to two alkyl or aryl groups. In the given composition C3H6O, if the oxygen atom is bonded to two carbon atoms without any double bond, it forms an ether. The remaining three carbons and six hydrogens can be arranged in various ways to form different ethers.
c) It can be an alcohol without any pi-bond:
An alcohol is a type of organic compound that contains a hydroxyl group (-OH) bonded to a carbon atom. In the given composition C3H6O, if one of the carbons is bonded to an oxygen atom through a single bond and also has a hydroxyl group, it forms an alcohol. The remaining three carbons and five hydrogens can be arranged in various ways to form different alcohols.
d) It can be an unsaturated ether:
An unsaturated ether is an organic compound that contains a double bond between one of the carbon atoms and the oxygen atom. In the given composition C3H6O, if one of the carbons is double-bonded to an oxygen atom and the remaining two carbons are single-bonded, it forms an unsaturated ether. The remaining three carbons and five hydrogens can be arranged in various ways to form different unsaturated ethers.
In summary, the given composition C3H6O can form compounds with different functional groups such as ketones, ethers, alcohols, and unsaturated ethers. The specific compound formed depends on the arrangement of the carbon and hydrogen atoms within the molecule.
a) It can be a ketone:
A ketone is a type of organic compound that contains a carbonyl group (C=O) bonded to two carbon atoms. In the given composition C3H6O, if one of the carbons is double-bonded to an oxygen atom, it forms a ketone. The remaining three carbons and six hydrogens can be arranged in various ways to form different ketones.
b) It can be an ether without any pi-bond:
An ether is an organic compound that has an oxygen atom bonded to two alkyl or aryl groups. In the given composition C3H6O, if the oxygen atom is bonded to two carbon atoms without any double bond, it forms an ether. The remaining three carbons and six hydrogens can be arranged in various ways to form different ethers.
c) It can be an alcohol without any pi-bond:
An alcohol is a type of organic compound that contains a hydroxyl group (-OH) bonded to a carbon atom. In the given composition C3H6O, if one of the carbons is bonded to an oxygen atom through a single bond and also has a hydroxyl group, it forms an alcohol. The remaining three carbons and five hydrogens can be arranged in various ways to form different alcohols.
d) It can be an unsaturated ether:
An unsaturated ether is an organic compound that contains a double bond between one of the carbon atoms and the oxygen atom. In the given composition C3H6O, if one of the carbons is double-bonded to an oxygen atom and the remaining two carbons are single-bonded, it forms an unsaturated ether. The remaining three carbons and five hydrogens can be arranged in various ways to form different unsaturated ethers.
In summary, the given composition C3H6O can form compounds with different functional groups such as ketones, ethers, alcohols, and unsaturated ethers. The specific compound formed depends on the arrangement of the carbon and hydrogen atoms within the molecule.
In a free radical reaction, free radicals are formed at- a)initiation step
- b)propagation step
- c)termination step
- d)both A and B
Correct answer is option 'D'. Can you explain this answer?
In a free radical reaction, free radicals are formed at
a)
initiation step
b)
propagation step
c)
termination step
d)
both A and B
 | Nitin Nair answered |
Once a reactive free radical is generated, it can react with stable molecules to form new free radicals. These new free radicals go on to generate yet more free radicals, and so on. Propagation steps often involve hydrogen abstraction or addition of the radical to double bonds.
Direction (Q. Nos. 10-14) This section contains 5 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONE or MORE THANT ONE is correct.
Q.Consider the followring molecule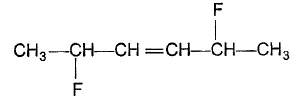 The correct statement concerning above molecule is/are
The correct statement concerning above molecule is/are - a)It has a total of six stereoisomers
- b)It's meso form upon ozonolysis followed by Zn-hydrolysis gives racemic mixture
- c)It's optically active isomers, each upon ozonolysis followed by Zn-hydrolysis gives a single enantiomer
- d)It has only two optically active isomers
Correct answer is option 'A,B,C'. Can you explain this answer?
Direction (Q. Nos. 10-14) This section contains 5 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONE or MORE THANT ONE is correct.
choices (a), (b), (c) and (d), out of which ONE or MORE THANT ONE is correct.
Q.
Consider the followring molecule
The correct statement concerning above molecule is/are
a)
It has a total of six stereoisomers
b)
It's meso form upon ozonolysis followed by Zn-hydrolysis gives racemic mixture
c)
It's optically active isomers, each upon ozonolysis followed by Zn-hydrolysis gives a single enantiomer
d)
It has only two optically active isomers
| | Tanuja Kapoor answered |
- The given compound has a total of six stereoisomers
- It's meso form upon ozonolysis followed by Zn-hydrolysis gives racemic mixture
- It's optically active isomers, each upon ozonolysis followed by Zn-hydrolysis gives a single enantiomer.
A stereoisomer of cyclobutane-1,2-diol has lower solubility in water than its other stereoisomer. Which is this isomer and why?- a)Trans, lower dipole moment
- b)Cis, higher dipole moment
- c)Cis, intramolecular H-bonding
- d)Trans, intramolecular H-bonding
Correct answer is option 'C'. Can you explain this answer?
A stereoisomer of cyclobutane-1,2-diol has lower solubility in water than its other stereoisomer. Which is this isomer and why?
a)
Trans, lower dipole moment
b)
Cis, higher dipole moment
c)
Cis, intramolecular H-bonding
d)
Trans, intramolecular H-bonding
 | Ciel Knowledge answered |
The correct answer is option c
The cis isomer is more likely to exhibit intramolecular hydrogen bonding between the two hydroxyl groups. This can make the molecule less soluble in water because hydrogen bonding with water molecules becomes less favorable when intramolecular hydrogen bonding is present.Considering the factors influencing solubility in water, the correct answer is option c. The cis stereoisomer has a higher dipole moment and is more likely to exhibit intramolecular hydrogen bonding, making it less soluble in water compared to the trans isomer.
Chapter doubts & questions for Organic Chemistry - Chemistry for SSS 3 2026 is part of SSS 3 exam preparation. The chapters have been prepared according to the SSS 3 exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for SSS 3 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Organic Chemistry - Chemistry for SSS 3 in English & Hindi are available as part of SSS 3 exam. Download more important topics, notes, lectures and mock test series for SSS 3 Exam by signing up for free.
Chemistry for SSS 338 videos|86 docs|29 tests |
Top Courses SSS 3
;
Signup to see your scores go up
within 7 days!
within 7 days!
Takes less than 10 seconds to signup