All Exams > MCAT > Organic Chemistry for MCAT > All Questions
All questions of Stereochemistry for MCAT Exam
Ethylene glycol is known to most Americans as antifreeze and to some for having a sweet syrupy taste. For scientists, it can be used as a protecting group for carbonyls. Which is the most stable conformer of the compound ethylene glycol depicted below?
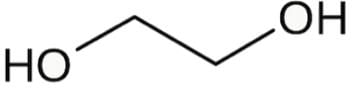
- a)Eclipsed conformer
- b)Gauche conformer
- c)Para conformer
- d)Anti conformer
Correct answer is option 'B'. Can you explain this answer?
Ethylene glycol is known to most Americans as antifreeze and to some for having a sweet syrupy taste. For scientists, it can be used as a protecting group for carbonyls. Which is the most stable conformer of the compound ethylene glycol depicted below?


a)
Eclipsed conformer
b)
Gauche conformer
c)
Para conformer
d)
Anti conformer
| | Ayesha Joshi answered |
- There is an inverse relationship between energy and stability. The more energy a conformer molecule possesses the more unstable it is, and the less energy the more stable. It is worth noting that there is enough energy to interconvert between the various conformers at room temperature.
- Eclipsed conformations tend to be less stable than the corresponding staggered conformations and to have a higher potential energy due to the interaction of electron clouds of adjacent atoms. Here are two examples of eclipsed conformers, fully eclipsed and eclipsed:
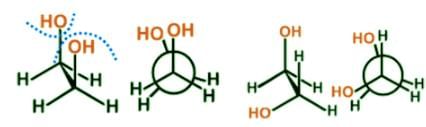
- For butane the gauche conformer is less stable than the anti conformer due to a crowding of the two methyl groups in the gauche structure, and that destabilizing factor is called steric strain or hindrance:
- There is no para conformer. Para is usually used to refer to the 1,4-isomer, where the substituents sit on carbon 1 and 4 of the aromatic ring structure.
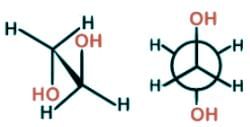
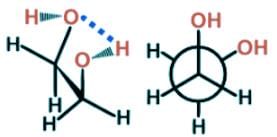
- In 1, 2-ethanediol, the gauche conformer presents an interesting configuration allowing for hydrogen bonding to occur between the two hydroxyl groups leading to a lower energy state and greater stability than the anti conformer.
Cavicularin is a natural phenolic secondary metabolite isolated from the liverwort Cavicularia densa, and the structure is depicted below. Your research group discovers that the sample rotates plane-polarized light 168.2 degrees. Based on the findings, which of the following statements can be deduced about the stereochemistry of this compound?
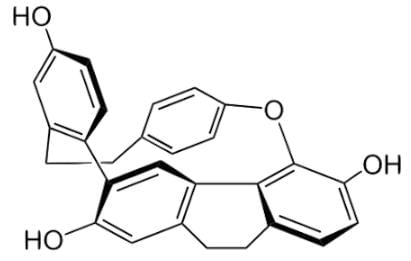
- a)This compound is not optically active because of its symmetry.
- b)This compound is not optically active since there are no stereogenic centers.
- c)This compound is optically active because it has stereogenic centers that create cis-trans isomers.
- d)This compound is optically active because the compound contains a center, plane, or axis of chirality.
Correct answer is option 'D'. Can you explain this answer?
Cavicularin is a natural phenolic secondary metabolite isolated from the liverwort Cavicularia densa, and the structure is depicted below. Your research group discovers that the sample rotates plane-polarized light 168.2 degrees. Based on the findings, which of the following statements can be deduced about the stereochemistry of this compound?


a)
This compound is not optically active because of its symmetry.
b)
This compound is not optically active since there are no stereogenic centers.
c)
This compound is optically active because it has stereogenic centers that create cis-trans isomers.
d)
This compound is optically active because the compound contains a center, plane, or axis of chirality.
 | Orion Classes answered |
- In identifying stereogenic centers, any quaternary or tertiary carbons would be a good place to look. There are no quaternary carbons, and all the tertiary carbons are part of a benzene ring structure.
- Any alkenyl groups would also be a good place to find stereogenic centers, but all the double bonded carbons in this molecules are in an aromatic ring structure.
- This molecule is definitely not symmetrical, so we cannot say that the compound is not optically active.
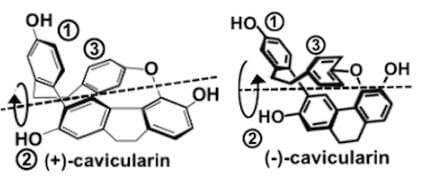
- Besides point chirality, there is planar and axial chirality. This molecule has an axis of chirality, so it is optically active:
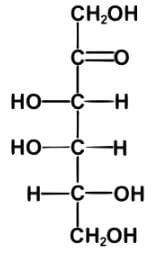
Tagalose is a functional sweetener and can be found in milk in small amounts. Commercially, Tagalose is produced from lactose, whereby galactose is isomerized under alkaline conditions to D-tagatose (shown below) by calcium hydroxide. Based on the Fischer projection, which of the following wedge-dash structures correctly represents D-tagatose?
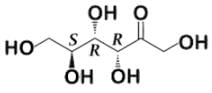
- a)
- b)
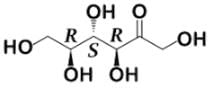
- c)
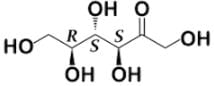
- d)
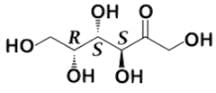
Correct answer is option 'D'. Can you explain this answer?
Tagalose is a functional sweetener and can be found in milk in small amounts. Commercially, Tagalose is produced from lactose, whereby galactose is isomerized under alkaline conditions to D-tagatose (shown below) by calcium hydroxide. Based on the Fischer projection, which of the following wedge-dash structures correctly represents D-tagatose?


a)

b)

c)

d)

| | Ayesha Joshi answered |
- There are two rules of thumb when dealing with Fischer projections of carbohydrates: when the hydroxyl group is on the right, the configuration is R, and when it is on the left, the configuration is S.
- For tagalose, the configurations are as follow: S, S, and R, starting from C-3.
- The second rule applies to all Fischer projections. For the sugar ribose, with all of its hydroxyl groups on the R in its Fischer projection, the hydroxyl groups alternate between dash and wedge:
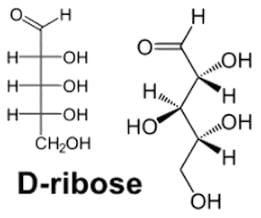
- Without having to determine the R/S configuration for any stereocenter. On adjacent stereocenters, when the configurations are the same, such as S, S or R, R, there is a switch from dash to wedge or wedge to dash. So if the configurations change, such as R, S or S, R, there are both dashes or wedges.
- Following that logic, this structure must be the correct answer:
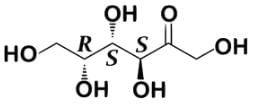
What is the total number of isomers with the formula C3H6O that are either cyclic or chiral?- a)3
- b)4
- c)6
- d)7
Correct answer is option 'C'. Can you explain this answer?
What is the total number of isomers with the formula C3H6O that are either cyclic or chiral?
a)
3
b)
4
c)
6
d)
7
| | Ayesha Joshi answered |
- Let’s consider possible cyclic compounds. The oxygen will be single bonded either as an ether and part of the ring structure or as an alcohol and outside any ring structure. Whether the nomenclature of the following structures is apparent or not is not important, but there are three possible structural isomers: cyclopropanol, oxetane, and methyl oxirane.
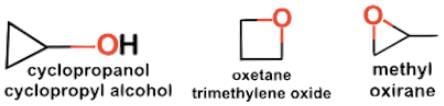
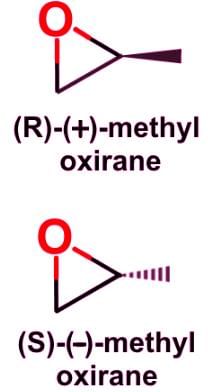
- Two isomers, propen-1-ol and methyl oxirane, have a stereogenic center, but from methyl oxirane there will be a set of enantiomers:
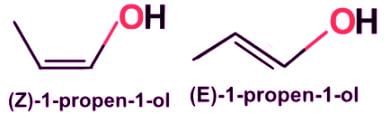
- Propen-1-ol’s geometric isomers are diastereomeric, and the count for stereoisomers is four:
- Since there is overlap of methyl oxirane in both categories, the total number of isomers is 6.
Which of the following cyclic hydrocarbon structures would be NOT categorized as an achiral compound?- a)
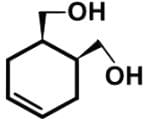
- b)
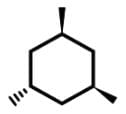
- c)
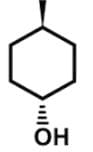
- d)
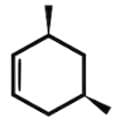
Correct answer is option 'D'. Can you explain this answer?
Which of the following cyclic hydrocarbon structures would be NOT categorized as an achiral compound?
a)

b)

c)

d)

| | Ayesha Joshi answered |
- There are two possibilities here for achirality. The first case is that there are no stereocenters on a particular molecule, and the second case is when a compound has a stereocenter, it can be achiral only when it is a meso compound.
- A meso compound is the non-optically active member of a set of stereoisomers, which necessarily have at least 2 stereocenters. It can be identified by an internal plane of symmetry, but sometimes a point or axis of symmetry, like in axial symmetry.
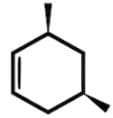
- For c,c,t-1, 3, 5-trimethylcyclohexane, this achiral compound has a plane of symmetry through C2 and C5:
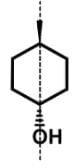
- For 4-methyl-1-cyclohexanol, this achiral compound has a plane of symmetry through C1 and C4:
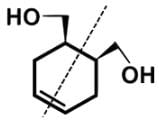
- For cyclohex-4-ene-1,2-diyldimethanol, there is a plane of symmetry bisecting the double bond between C-4 and C-5:
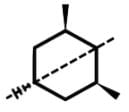
- The remaining compound, 3,5-dimethyl-cyclo-1-hexene, is not an achiral compound and will rotate plane-polarized light as a chiral compound due to a lack of a plane or axis of symmetry.
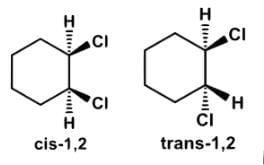
Which of the following statements most accurately describes the stereochemistry between the various cyclohexanes?- a)Cis-1,2-dichlorocyclohexane and trans-1,2-dichlorocyclohexane rotate plane-polarized light in opposite directions, and together in equal proportions form a racemic mixture.
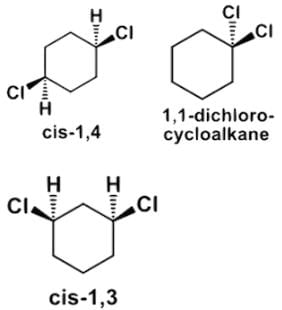
- b)Only cis-1,4-dichlorocyclohexane is achiral due to a plane of symmetry, and cis-1,4-dichlorocyclohexane is diastereomeric to trans-1,4-dichlorocyclohexane.
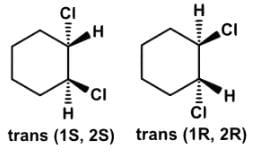
- c)The conformational isomers of trans-1,2-dichlorocyclohexane are enantiomers, which are not interconvertible, but resolvable.
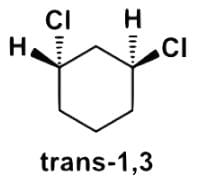
- d)The diaxial and diequatorial forms of trans-1,3-dichlorohexane can be separated by their differing physical properties.
Correct answer is option 'C'. Can you explain this answer?
Which of the following statements most accurately describes the stereochemistry between the various cyclohexanes?
a)
Cis-1,2-dichlorocyclohexane and trans-1,2-dichlorocyclohexane rotate plane-polarized light in opposite directions, and together in equal proportions form a racemic mixture.
b)
Only cis-1,4-dichlorocyclohexane is achiral due to a plane of symmetry, and cis-1,4-dichlorocyclohexane is diastereomeric to trans-1,4-dichlorocyclohexane.
c)
The conformational isomers of trans-1,2-dichlorocyclohexane are enantiomers, which are not interconvertible, but resolvable.
d)
The diaxial and diequatorial forms of trans-1,3-dichlorohexane can be separated by their differing physical properties.
| | Ayesha Joshi answered |
- Let’s look at trans-1,3-dichlorohexane. The chlorines cannot be positioned diaxial or diequatorial, but can only be axial and equatorial or vice versa. Additionally, if they were diastereomers or structural isomers, then they could be separated by physical means.
- Consider whether only cis-1,4-dichlorocylohexane is achiral. 1,1-dichlorocycloalkane and trans-1,4-dichlorocycloalkane are both achiral due to a plane of symmetry, for instance, but cis-1,4-dichlorocyclohexane is indeed diastereometric to trans-1,4-dichlorocyclohexane.
- If cis-1,2 and trans-1,2-dichlorocycloalkane rotate plane-polarized light in opposite directions and together they form a racemic mixture, that would make them enantiomers. In fact, they are diastereomers.
- Isomers are interconvertible only if there is a small energy barrier separating the two, such as the conformational isomers of butane. Two isomers are resolvable if no interconversion takes place.
- Since the two chlorines can be either be positioned diaxial or diequatorial, the enantiomer with diequatorial chlorines would be more stable, hence the isomers are not interconvertible and resolvable:
Plant extracts are widely used in many parts of Cameroon to treat infectious diseases or related symptoms including abdominal pains, itching, urinary and respiratory ailments, fever and coughing, diarrhea. Harunmadagascarin C as depicted below is an extract from the plant Harungana madagascariensis that has been studied for potential antimicrobial activity. Which statement best describes the stereochemistry of the compound?
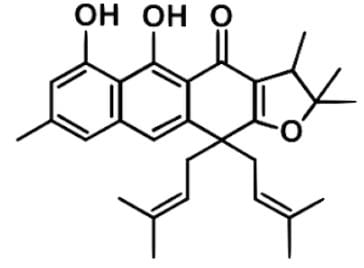
- a)The 2 stereogenic centers are currently in the E configuration, and the double bond in the ring structure is not stereogenic.
- b)There is one chiral center present in the molecule.
- c)There are four possible stereoisomers for Harunmadagascarin C since, as depicted, there are 2 stereocenters in the compound.
- d)Harunmadagascarin C is achiral and does not have any stereogenic or chiral centers.
Correct answer is option 'B'. Can you explain this answer?
Plant extracts are widely used in many parts of Cameroon to treat infectious diseases or related symptoms including abdominal pains, itching, urinary and respiratory ailments, fever and coughing, diarrhea. Harunmadagascarin C as depicted below is an extract from the plant Harungana madagascariensis that has been studied for potential antimicrobial activity. Which statement best describes the stereochemistry of the compound?


a)
The 2 stereogenic centers are currently in the E configuration, and the double bond in the ring structure is not stereogenic.
b)
There is one chiral center present in the molecule.
c)
There are four possible stereoisomers for Harunmadagascarin C since, as depicted, there are 2 stereocenters in the compound.
d)
Harunmadagascarin C is achiral and does not have any stereogenic or chiral centers.
 | Orion Classes answered |
- Look for the stereogenic centers, which can be either quaternary or tertiary carbons or double bonded carbons not within an aromatic ring structure.
- There is one quaternary carbon in the furan ring structure, but there are two methyl group attached, so it is achiral.
- There seems to be possible stereogenic centers in the alkenyl groups coming off the cyclohexanone ring structure. Since there are the same two methyl groups attached on to one end of the double bond for both, there is no chirality.
- There is a stereogenic center in the 5-carbon ring structure at the tertiary carbon. This is a chiral center since there are 4 different substituents attached to the central C atom.
Which of the following statements most accurately describes the characteristics of diastereomers, enantiomers, and meso compounds?- a)Diastereomers, which are stereoisomers not related by reflection or non-superimposable non-mirror images, are similar to other isomers in having distinct chemical and physical properties.
- b)The chemical properties of enantiomers, including both the qualitative reactions and the quantitative rates of reaction, are identical when reacting with other chiral chemical species.
- c)For a given set of stereoisomers, meso compounds must have at least 2 stereocenters and are diastereomeric to the other stereoisomers in the set.
- d)Enantiomers rotate plane polarized light and in opposite directions, while meso compounds are achiral and do not rotate plane polarized light.
Correct answer is option 'C'. Can you explain this answer?
Which of the following statements most accurately describes the characteristics of diastereomers, enantiomers, and meso compounds?
a)
Diastereomers, which are stereoisomers not related by reflection or non-superimposable non-mirror images, are similar to other isomers in having distinct chemical and physical properties.
b)
The chemical properties of enantiomers, including both the qualitative reactions and the quantitative rates of reaction, are identical when reacting with other chiral chemical species.
c)
For a given set of stereoisomers, meso compounds must have at least 2 stereocenters and are diastereomeric to the other stereoisomers in the set.
d)
Enantiomers rotate plane polarized light and in opposite directions, while meso compounds are achiral and do not rotate plane polarized light.
| | Ayesha Joshi answered |
- Enantiomers share the same physical properties except for their ability to rotate plane-polarized light. Enantiomers share the same chemical properties except when reacting with a chiral reagent.
- To elaborate, any chiral stereoisomers rotates plane-polarized light, but enantiomers rotate in a specific relationship, in opposite directions and in equal magnitude. If the S enantiomer rotates light +50°, then the R enantiomer rotates light -50°.
- Diastereomers are stereoisomers that are not related by reflection, but diastereomers share similar chemical properties and have distinct physical properties that allow them to be separated by physical means.
- The correct statement is that meso compounds must have at least 2 stereocenters and are diastereomeric to the other stereoisomers. Do not confuse that with meso compounds having an internal plane of symmetry.
While brucine is a naturally occurring alkaloid related to strychnine and hence poisonous, it can be used as a tool for stereospecific chemical syntheses. Which of the following statements best describes the result of this reaction series with a racemic mixture of an amino acid?
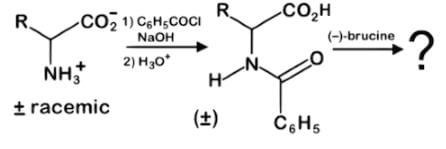
- a)The racemates react with the alkaline compound brucine in the same way to produce a racemic product.
- b)The products can be separated using thin layer chromatography techniques with a chiral stationary phase.
- c)The two products are separated by fractional distillation since as enantiomers they differ in their physical properties.
- d)The products formed are meso compounds which allow for physical separation of the two enantiomers originally in the racemic mixture.
Correct answer is option 'B'. Can you explain this answer?
While brucine is a naturally occurring alkaloid related to strychnine and hence poisonous, it can be used as a tool for stereospecific chemical syntheses. Which of the following statements best describes the result of this reaction series with a racemic mixture of an amino acid?


a)
The racemates react with the alkaline compound brucine in the same way to produce a racemic product.
b)
The products can be separated using thin layer chromatography techniques with a chiral stationary phase.
c)
The two products are separated by fractional distillation since as enantiomers they differ in their physical properties.
d)
The products formed are meso compounds which allow for physical separation of the two enantiomers originally in the racemic mixture.
 | Orion Classes answered |
- Brucine is considered a chiral resolving agent which acts by reacting with the racemic amino acid to form two diastereomeric salts.
- As such, the salts are now made separable by some physical means, i.e. solubility, boiling point, melting point, etc. While diastereomers are separable, enantiomers only differing in their optical activity are not.
- When the racemates react with brucine, diastereomeric salts NOT a racemic product nor a meso compound is produced. Once the salts are acidified, the separated amino acid enantiomers are obtained.
- Another technique for the separation of enantiomers is chromatography in association with a chiral stationary phase. The enantiomers react differently to the chiral stationary phase such that one enantiomer moves through the chromatographic column more slowly, in this case up the plate, than the other.
The compound hexachlorosulfolipid belongs to a class of compounds called chlorosulfolipids, which are integral components of algal membranes and are known to inhibit protein kinases. Some have been isolated from mussels and are linked to diarrhetic shellfish poisoning. Based on the wedge and dash structure below, what are the R and S designations from left to right for the stereocenters of this toxin?
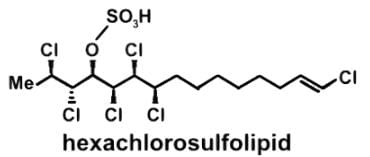
- a)R, S, R, S, S, R
- b)R, S, S, R, R, S
- c)S, R, S, S, S, S
- d)R, R, S, S, R, R
Correct answer is option 'A'. Can you explain this answer?
The compound hexachlorosulfolipid belongs to a class of compounds called chlorosulfolipids, which are integral components of algal membranes and are known to inhibit protein kinases. Some have been isolated from mussels and are linked to diarrhetic shellfish poisoning. Based on the wedge and dash structure below, what are the R and S designations from left to right for the stereocenters of this toxin?


a)
R, S, R, S, S, R
b)
R, S, S, R, R, S
c)
S, R, S, S, S, S
d)
R, R, S, S, R, R
| | Ayesha Joshi answered |
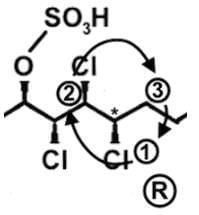
- Let’s look at the first stereocenter on the very left. In determining the priority, use the Cahn-Ingold-Prelog rules, which essentially tells us to arrange in terms of atomic number. The first stereocenter has a Cl, C, C attached, and since Cl has the highest atomic number, it has the highest priority. Then we have a C attached to H’s or attached to a Cl, and for the same reason, C with the Cl takes priority:
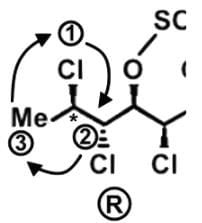
- Draw a circle connecting them in this order 1 → 2 → 3 → 1. Remember that clockwise is R and counterclockwise is S. If the H is sticking into the plane of the page, however, the configuration must be switched. For So the first stereocenter is R
- Following a similar procedure for the second stereocenter to determine that it has a S configuration. Here since Cl is pointed back into the page and H is pointed forward out of the page, the configuration is switched from R to S:
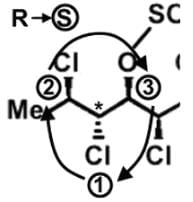
- Following a similar procedure for the second stereocenter to determine that it has an S configuration:
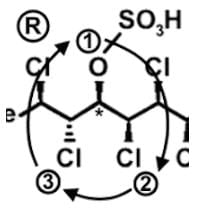
- Following a similar procedure for the third stereocenter to determine that it has an R configuration:
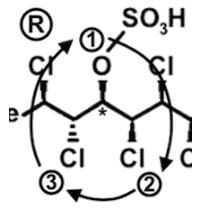
- Following a similar procedure for the fourth stereocenter to determine that it has an S configuration:
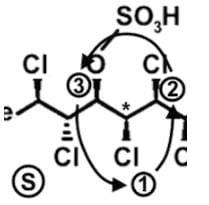
- Following a similar procedure for the fifth stereocenter to determine that it has an S configuration:
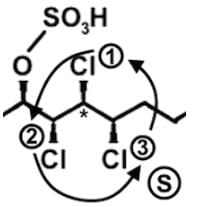
- Following a similar procedure for the sixth stereocenter to determine that it has a R configuration. So the correct order is R, S, R, S, S, and R:
Chapter doubts & questions for Stereochemistry - Organic Chemistry for MCAT 2026 is part of MCAT exam preparation. The chapters have been prepared according to the MCAT exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for MCAT 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Stereochemistry - Organic Chemistry for MCAT in English & Hindi are available as part of MCAT exam. Download more important topics, notes, lectures and mock test series for MCAT Exam by signing up for free.
Organic Chemistry for MCAT140 videos|5 docs|15 tests |
Top Courses MCAT
;
Signup to see your scores go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup

