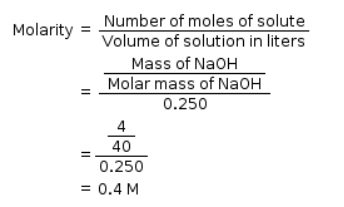All Exams > NEET > Chemistry Class 11 > All Questions
All questions of Some Basic Concepts Of Chemistry for NEET Exam
Can you explain the answer of this question below:Molecular mass of glucose molecule (C6H12O6) is:
- A:
180 g
- B:
172 g
- C:
182 g
- D:
192 g
The answer is a.
Molecular mass of glucose molecule (C6H12O6) is:
180 g
172 g
182 g
192 g
 | Samridhi Kaur answered |
Molecular mass : The sum of the atomic masses of all the atoms in a molecule of a substance is called the molecular mass of the molecule.
Generally we use relative atomic masses of atoms for calculating the molecular mass of 1 mole of any molecular or ionic substances.
Molecular formula of glucose is C6H12O6
Atomic mass of H = 1
Atomic mass pf C = 12
Atomic mass of O = 16
Molecular mass of C6H12O6 = 12(Atomic mass of Hydrogen) + 6(Atomic mass of carbon) + 6(Atomic mass of oxygen)
= 12 x 1 + 6X12 + 6X16
= 12 + 72 + 96 = 180 u.
The scientific notation for 0.00016 is:- a)1.6 Χ 10-4
- b)16 Χ 10-4
- c)1.6 Χ 10-2
- d)1.6 Χ 104
Correct answer is option 'A'. Can you explain this answer?
The scientific notation for 0.00016 is:
a)
1.6 Χ 10-4
b)
16 Χ 10-4
c)
1.6 Χ 10-2
d)
1.6 Χ 104
| | Arya Dasgupta answered |
Here, the decimal has to be moved four places to the right side and hence -4 is the exponent in the scientific notation.
Which law is also known as Law of constant composition?- a)Law of multiple proportions
- b)Avogadro’s law
- c)Law of conservation of mass
- d)Law of definite proportions
Correct answer is option 'D'. Can you explain this answer?
Which law is also known as Law of constant composition?
a)
Law of multiple proportions
b)
Avogadro’s law
c)
Law of conservation of mass
d)
Law of definite proportions
| | Pooja Shah answered |
The law of definite proportions, also known as the law of constant composition states that all pure samples of the same chemical compound contain the same elements combined in the same proportions by mass.
What this law emphasizes is that, if pure samples of the same chemical substance, wherever they may be found, are analyzed, it will be found that they all consist of the same elements, as well as having these elements combine in the same proportions by mass.
For examples, pure sample of copper(II) oxide is composed of copper and oxygen, in the proportion of 1:1 by mole, or 64 g of copper to 16 g of oxygen or 1 g of copper to 0.25 g of oxygen.
What this law emphasizes is that, if pure samples of the same chemical substance, wherever they may be found, are analyzed, it will be found that they all consist of the same elements, as well as having these elements combine in the same proportions by mass.
For examples, pure sample of copper(II) oxide is composed of copper and oxygen, in the proportion of 1:1 by mole, or 64 g of copper to 16 g of oxygen or 1 g of copper to 0.25 g of oxygen.
The molar mass of ZnSO4 is- a)161.47 g
- b)136.4g
- c)166.4g
- d)156.4g
Correct answer is option 'A'. Can you explain this answer?
The molar mass of ZnSO4 is
a)
161.47 g
b)
136.4g
c)
166.4g
d)
156.4g
| | Lavanya Menon answered |
Chemical Formula: ZnSO4
Molar Mass: 161.47 g/mol (anhydrous)
Molar Mass: 161.47 g/mol (anhydrous)
Can you explain the answer of this question below:100mL of gaseous hydrogen combines with 50mL of gaseous oxygen to give 100mL of water vapours. This can be explained on the basis of:- A:Law of definite proportions
- B:Gay Lussac’s law
- C:Law of multiple proportions
- D:Avogadro law
The answer is b.
100mL of gaseous hydrogen combines with 50mL of gaseous oxygen to give 100mL of water vapours. This can be explained on the basis of:
A:
Law of definite proportions
B:
Gay Lussac’s law
C:
Law of multiple proportions
D:
Avogadro law
| | Nandini Patel answered |
For the reaction 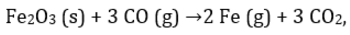 224 g of CO is available to react with 400 g Fe2O3, the yield of iron and CO2, are:
224 g of CO is available to react with 400 g Fe2O3, the yield of iron and CO2, are:- a)225 and 279
- b)280 and 330 g
- c)210 and 290
- d)210 and 279 g
Correct answer is 'B'. Can you explain this answer?
For the reaction
224 g of CO is available to react with 400 g Fe2O3, the yield of iron and CO2, are:
a)
225 and 279
b)
280 and 330 g
c)
210 and 290
d)
210 and 279 g
| | Neha Sharma answered |
Moles of CO =8 moles Moles of Fe2O3= 2.5 moles.
3 moles of CO is needed for 1 mole of Fe2O3 so 8 moles of CO will require 2.66 mole of Fe2O3 so Fe2O3 is limiting reagent.
1 mole of Fe2O3 produce 2 mole of Fe so 2.5 mole of Fe2O3will produce 5 mole of Fe = 280g of Fe.
Also 1 mole of Fe2O3 also produce 3 mole of CO2 so 2.5 mole of Fe2O3 will produce 7.5 mole of CO2=330g.
The number of significant figures in 3256 is:- a)4
- b)3
- c)5
- d)2
Correct answer is option 'A'. Can you explain this answer?
The number of significant figures in 3256 is:
a)
4
b)
3
c)
5
d)
2
 | Lohit Matani answered |
- The significant figures of numbers are digits to convey the meaning that contributes to its measurement resolution.
- Non-zero digits are always significant.
- Here, 3256, this number has four non-zero numbers.
- That's why it has four significant figures.
Law of conservation of mass was given by:- a)Avogadro
- b)Antoine Lavoisier
- c)John Dalton
- d)Joseph Proust
Correct answer is option 'B'. Can you explain this answer?
Law of conservation of mass was given by:
a)
Avogadro
b)
Antoine Lavoisier
c)
John Dalton
d)
Joseph Proust
| | Lavanya Menon answered |
The Law of Conservation of Mass (or Matter) in a chemical reaction can be stated thus:
In a chemical reaction, matter is neither created nor destroyed.
It was discovered by Antoine Laurent Lavoisier (1743-94) about 1785. However, philosophical speculation and even some quantitative experimentation preceeded him. In addition, he was certainly not the first to accept this law as true or to teach it, but he is credited as its discoverer.
In a chemical reaction, matter is neither created nor destroyed.
It was discovered by Antoine Laurent Lavoisier (1743-94) about 1785. However, philosophical speculation and even some quantitative experimentation preceeded him. In addition, he was certainly not the first to accept this law as true or to teach it, but he is credited as its discoverer.
Suppose the elements X and Y combine to form two compounds XY2 and X3Y2. When 0.1 mole of XY2 weighs 10 g and 0.05 mole of X3Y2 weighs 9 g, the atomic weights of X and Y are- a)40, 30
- b)60, 40
- c)20, 30
- d)30, 20
Correct answer is option 'A'. Can you explain this answer?
Suppose the elements X and Y combine to form two compounds XY2 and X3Y2. When 0.1 mole of XY2 weighs 10 g and 0.05 mole of X3Y2 weighs 9 g, the atomic weights of X and Y are
a)
40, 30
b)
60, 40
c)
20, 30
d)
30, 20
| | Pranavi Chopra answered |
Let atomic weight of element X is x and that of element Y is y

What will be the molality of the solution containing 18.25 g of HCl gas in 500 g of water?- a)0.1 m
- b)1 M
- c)0.5 m
- d)1 m
Correct answer is option 'D'. Can you explain this answer?
What will be the molality of the solution containing 18.25 g of HCl gas in 500 g of water?
a)
0.1 m
b)
1 M
c)
0.5 m
d)
1 m
 | Saikat Sharma answered |
Given:
Mass of HCl = 18.25 gm
Molar mass of HCl = 36.5 g/mol
Mass of water = 500 gm
= 0.5 kg
No. of moles of HCl;

Molality is,

Molality of solution is 1 m.
The distinction between atoms and molecules was made by:- a)Dalton’s atomic theory
- b)Avogadro law
- c)Laws of chemical combination
- d)Gay lussacs law
Correct answer is 'B'. Can you explain this answer?
The distinction between atoms and molecules was made by:
a)
Dalton’s atomic theory
b)
Avogadro law
c)
Laws of chemical combination
d)
Gay lussacs law
| | Preeti Iyer answered |
Avogadro made the distinction between atoms and molecules, which today seems clear. However, Dalton rejected Avogadro's hypothesis because Dalton believed that atoms of the same kind could not combine. Since it was believed that atoms were held together by an electrical force, only unlike atoms would be attracted together, and like atoms should repel. Therefore it seemed impossible for a molecule of oxygen, O2, to exist. Avogadro's work, even if it was read appears not to have been understood, and was pushed into the dark recesses of chemistry libraries and ignored. Avogadro continued to teach at the university of Turin, when it was not closed because of the political upheavals going on in Italy at the time, and died in 1854, an unknown figure.
A statement which is not a part of Dalton’s atomic theory is:- a)Matter is made of atoms.
- b)All atoms of a given element are identical.
- c)Atoms are composed of sub-atomic particles called electron, proton and neutron.
- d)Atoms of different elements have different mass and different properties.
Correct answer is option 'C'. Can you explain this answer?
A statement which is not a part of Dalton’s atomic theory is:
a)
Matter is made of atoms.
b)
All atoms of a given element are identical.
c)
Atoms are composed of sub-atomic particles called electron, proton and neutron.
d)
Atoms of different elements have different mass and different properties.
| | Shreya Gupta answered |
According to the Dalton’s atomic theory, atoms are indivisible i.e. they are not composed of any sub-atomic particles
Laws of chemical combinations can be explained on the basis of:- a)Avogadro law
- b)Gay lussacs law
- c)Dalton’s atomic theory
- d)Mole concept
Correct answer is option 'C'. Can you explain this answer?
Laws of chemical combinations can be explained on the basis of:
a)
Avogadro law
b)
Gay lussacs law
c)
Dalton’s atomic theory
d)
Mole concept
| | Gaurav Kumar answered |
The old ideas were put on a scientific scale by John Dalton in the form of a theory, known as Dalton’s atomic theory,
Main postulates of which are as follows :
• All matters are made of atoms. Atoms are indivisible and indestructible.
• All atoms of a given element are identical in mass and properties.
• Atoms of different elements differ in properties and have different masses and sizes.
• Compounds are formed by a combination of two or more different kinds of atoms.
• A chemical reaction is a rearrangement of atoms.These are neither created nor destroyed in a chemical reaction
Main postulates of which are as follows :
• All matters are made of atoms. Atoms are indivisible and indestructible.
• All atoms of a given element are identical in mass and properties.
• Atoms of different elements differ in properties and have different masses and sizes.
• Compounds are formed by a combination of two or more different kinds of atoms.
• A chemical reaction is a rearrangement of atoms.These are neither created nor destroyed in a chemical reaction
What is incorrect about the Law of conservation of mass?- a)A given compound always conatains exactly the same proportion of elements by weight
- b)Mass of reactants is equal to the mass of products
- c)Matter can neither be created nor destroyed
- d)It was given by Antoine Lavoiser
Correct answer is option 'A'. Can you explain this answer?
What is incorrect about the Law of conservation of mass?
a)
A given compound always conatains exactly the same proportion of elements by weight
b)
Mass of reactants is equal to the mass of products
c)
Matter can neither be created nor destroyed
d)
It was given by Antoine Lavoiser
| | Jyoti Kumar answered |
The law of definite proportions, also known law of definite composition, states that regardless of the amount, a pure compound always contains the same elements in the same proportions by mass. Law of multiple proportions, also known as Dalton s Law, states that when one element combines with another to form more than one compound, the mass rations of the elements in the compounds are simple whole numbers of each other.
The correct relationship between picometer and nanometer is- a)1nm = 1000pm
- b)1nm = 10pm
- c)1pm = 10nm
- d)1pm = 100nm
Correct answer is option 'A'. Can you explain this answer?
The correct relationship between picometer and nanometer is
a)
1nm = 1000pm
b)
1nm = 10pm
c)
1pm = 10nm
d)
1pm = 100nm
| | Neha Joshi answered |
1 picometer = 10-12 m
1 nanometer=10-9 m
So, 1 picometer = 0.001 nanometer .
From this we get 1 nanometer= 1000 picometer.
1 nanometer=10-9 m
So, 1 picometer = 0.001 nanometer .
From this we get 1 nanometer= 1000 picometer.
Hence, the correct answer is Option A.
How many atoms of hydrogen are in 67.2 L of H2 at STP?- a)5.612 × 1024
- b)2.612 × 1024
- c)4.612 × 1024
- d)3.6132 × 1024
Correct answer is option 'D'. Can you explain this answer?
How many atoms of hydrogen are in 67.2 L of H2 at STP?
a)
5.612 × 1024
b)
2.612 × 1024
c)
4.612 × 1024
d)
3.6132 × 1024
| | Anjana Sharma answered |
Standard temperature and pressure (STP) is defined as 0 degrees Celsius and 1 atmosphere of pressure. At STP, 1 mole of any gas occupies 22.4 liters.
First, calculate the number of moles of hydrogen gas (H2) in 67.2 liters:
67.2 L / 22.4 L/mole = 3 moles of H2
Each molecule of H2 contains 2 atoms of hydrogen. Therefore, 3 moles of H2 contains:
3 moles * (6.022 x 10^23 molecules/mole) * 2 atoms/molecule = 3.6132 x 10^24 atoms of hydrogen.
First, calculate the number of moles of hydrogen gas (H2) in 67.2 liters:
67.2 L / 22.4 L/mole = 3 moles of H2
Each molecule of H2 contains 2 atoms of hydrogen. Therefore, 3 moles of H2 contains:
3 moles * (6.022 x 10^23 molecules/mole) * 2 atoms/molecule = 3.6132 x 10^24 atoms of hydrogen.
A measured temperature is 100 0F on Fahrenheit scale, then what is this reading be on Celsius scale :- a)11.2 0C
- b)78 0C
- c)102.7 0C
- d)37.8 0C
Correct answer is option 'D'. Can you explain this answer?
A measured temperature is 100 0F on Fahrenheit scale, then what is this reading be on Celsius scale :
a)
11.2 0C
b)
78 0C
c)
102.7 0C
d)
37.8 0C
| | Krishna Iyer answered |
C-0/100-0 = F-32/180.
C/5= F-32/9.
C/5= 100-32/9.
C/5= 68/9.
C= 68×5/9.
C= 340/9.
C= 37.77.
C= 37.8
C/5= F-32/9.
C/5= 100-32/9.
C/5= 68/9.
C= 68×5/9.
C= 340/9.
C= 37.77.
C= 37.8
For a reaction A + 2B → C, the amount of C formed by starting the reaction with 5 moles of A and 8 moles of B is- a)5 moles
- b)8 moles
- c)16 moles
- d)4 moles
Correct answer is option 'D'. Can you explain this answer?
For a reaction A + 2B → C, the amount of C formed by starting the reaction with 5 moles of A and 8 moles of B is
a)
5 moles
b)
8 moles
c)
16 moles
d)
4 moles
| | Neha Joshi answered |
A +2B → C
t = 0 5 8 0
t= ? 5x 8-2x x
nx = 9a
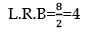
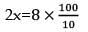
x = 4
mole of C = 4 mole
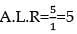
nx = 9a
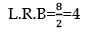
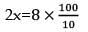
x = 4
mole of C = 4 mole
The number of atoms in 0.1 mol of a triatomic gas is :[2010](NA = 6.02 ×1023 mol–1)- a)6.026 × 1022
- b)1.806 × 1023
- c)3.600 × 1023
- d)1.800 × 1022
Correct answer is option 'B'. Can you explain this answer?
The number of atoms in 0.1 mol of a triatomic gas is :[2010](NA = 6.02 ×1023 mol–1)
a)
6.026 × 1022
b)
1.806 × 1023
c)
3.600 × 1023
d)
1.800 × 1022
 | Dipika Das answered |
The number of atoms in 0.1 mole of a triatomic gas = 0.1 × 3 × 6.023 × 1023.
= 1.806 × 1023
= 1.806 × 1023
Can you explain the answer of this question below:How many atoms of Oxygen are there in 18g of water? (Hint: Avagadro’s Number = 6.02 x 1023 atoms/mol)- a:8.02 x 1023
- b:6.02 x 1023
- c:5 .02 x 1023
- d:7.02 x 1023
Correct answer is 'b'.
How many atoms of Oxygen are there in 18g of water? (Hint: Avagadro’s Number = 6.02 x 1023 atoms/mol)
a:
8.02 x 1023
b:
6.02 x 1023
c:
5 .02 x 1023
d:
7.02 x 1023
| | Gaurav Kumar answered |
18g H2O = 1mol water = 6.02 x 1023 molecules of water = 6.02 x 1023 atoms of oxygen.
The molar mass of Al2O3 is- a)112
- b)82
- c)102
- d)92
Correct answer is option 'C'. Can you explain this answer?
The molar mass of Al2O3 is
a)
112
b)
82
c)
102
d)
92
| | Pooja Shah answered |
Molar mass = 2(27) +3 (16) = 102g.
Which of the following statements is/are correct?- a)Two or more atoms combine to give molecules
- b)The atoms of different elements are present in a compound in a fixed and definite ratio
- c)The constituents of a compound can be separated by physical and chemical methods
- d)Both (a) and (b)
Correct answer is option 'D'. Can you explain this answer?
Which of the following statements is/are correct?
a)
Two or more atoms combine to give molecules
b)
The atoms of different elements are present in a compound in a fixed and definite ratio
c)
The constituents of a compound can be separated by physical and chemical methods
d)
Both (a) and (b)
| | Preeti Khanna answered |
- Two or more atoms combine to give molecules.
- The atoms of different elements are present in a compound in a fixed and definite ratio.
- These two are correct by concepts of chemistry.
Which one of the following is not a mixture?- a)Distilled water
- b)Sugar dissolved in water
- c)Liquefied Petroleum Gas (LPG)
- d)Gasoline
Correct answer is option 'A'. Can you explain this answer?
Which one of the following is not a mixture?
a)
Distilled water
b)
Sugar dissolved in water
c)
Liquefied Petroleum Gas (LPG)
d)
Gasoline
| | Gaurav Kumar answered |
Distilled water is the pure form of water with no impurities present in it; others are the mixture of two or more components.
Can you explain the answer of this question below:Choose the most appropriate answer amongst the options given below for the statement. A solution of a desired concentration is prepared by diluting- a:solution of known higher concentration
- b:solution of known lower concentration
- c:from a serially diluted solution
- d:stock solution.
Correct answer is 'd'.
Choose the most appropriate answer amongst the options given below for the statement. A solution of a desired concentration is prepared by diluting
a:
solution of known higher concentration
b:
solution of known lower concentration
c:
from a serially diluted solution
d:
stock solution.
| | Geetika Shah answered |
Stock solution is diluted to prepare the solution of desired concentration.
The molar mass of AgNO3 is- a)169.87 g
- b)189.9
- c)179.9
- d)159.9
Correct answer is option 'A'. Can you explain this answer?
The molar mass of AgNO3 is
a)
169.87 g
b)
189.9
c)
179.9
d)
159.9
| | Geetika Shah answered |
Molar mass of AgNO3 = mass of Ag + N + O3 = 107.87 + 14 + 3* 16 = 107. 87 + 14 + 48 = 169.87 g
Number of atoms is 560 g of Fe (atomic mass = 56 g mol-1)- a)Is twice that of 70 g N (Nitrogen)
- b)Is half that of 20 g H(Hydrogen)
- c)Both are correct
- d)none is correct
Correct answer is option 'C'. Can you explain this answer?
Number of atoms is 560 g of Fe (atomic mass = 56 g mol-1)
a)
Is twice that of 70 g N (Nitrogen)
b)
Is half that of 20 g H(Hydrogen)
c)
Both are correct
d)
none is correct
 | Simran Mishra answered |
Fe atom= 560/56 =10 mole
= 10 x NA atom
option(A) = 5 mol
5 x NA
option (B) = 20/1 = 20
20 x NA
= 10 x NA atom
option(A) = 5 mol
5 x NA
option (B) = 20/1 = 20
20 x NA
How many moles of magnesium phosphate, Mg3 (PO4)2 will contain 0.25 mole of oxygen atoms?- a)3.125 × 10-2
- b)1.25 × 10-2
- c)2.5 × 10-2
- d)0.02
Correct answer is option 'A'. Can you explain this answer?
How many moles of magnesium phosphate, Mg3 (PO4)2 will contain 0.25 mole of oxygen atoms?
a)
3.125 × 10-2
b)
1.25 × 10-2
c)
2.5 × 10-2
d)
0.02
 | Arjun Saini answered |
Mg3(P04)2 → 3Mg
→2 P
→ 8 mole oxygen
8 mole of oxygen - 1 mole Mg3 (P0)2
0.25 - ? 0.25/8 = 3 x 10-2
→2 P
→ 8 mole oxygen
8 mole of oxygen - 1 mole Mg3 (P0)2
0.25 - ? 0.25/8 = 3 x 10-2
Which of the following is an element?- a)Sugar solution
- b)Brass
- c)HCl
- d)Copper metal
Correct answer is option 'D'. Can you explain this answer?
Which of the following is an element?
a)
Sugar solution
b)
Brass
c)
HCl
d)
Copper metal
| | Krishna Iyer answered |
Copper is a chemical element with symbol Cu and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity.
According to Dalton’s atomic theory chemical reactions involve:- a)destruction of atoms
- b)reorganisation of nuclei
- c)reorganisation of atoms
- d)construction of atoms
Correct answer is 'C'. Can you explain this answer?
According to Dalton’s atomic theory chemical reactions involve:
a)
destruction of atoms
b)
reorganisation of nuclei
c)
reorganisation of atoms
d)
construction of atoms
| | Shreya Gupta answered |
Chemical reactions involve merely the combination, separation, or rearrangement of atoms and that during these processes atoms are not subdivided, created, or destroyed.
The following equations are balanced atomwise and chargewise.(i) Cr2O72- + 8H+ + 2H202 → 2Cr3+ + 7H20 + 202(ii) Cr2072- + 8H+ + 5H202→ 2Cr3+ + 9H20 + 402(iii) Cr2072- + 8H+ + 7H202→ 2Cr+ + 11H20 + 502The precise equationlequations representing the oxidation of H202 is/are- a)(i) only
- b)(ii) only
- c)(iii) only
- d)all the three
Correct answer is option 'A'. Can you explain this answer?
The following equations are balanced atomwise and chargewise.
(i) Cr2O72- + 8H+ + 2H202 → 2Cr3+ + 7H20 + 202
(ii) Cr2072- + 8H+ + 5H202→ 2Cr3+ + 9H20 + 402
(iii) Cr2072- + 8H+ + 7H202→ 2Cr+ + 11H20 + 502
The precise equationlequations representing the oxidation of H202 is/are
a)
(i) only
b)
(ii) only
c)
(iii) only
d)
all the three
| | Krishna Iyer answered |
The correct answer is option A
Cr2O72- converts into Cr3+ in acidic medium I.e. in H+ medium.
First balance the Cr atom on both sides and then Oxygen atom. H+ is in excess due to acidic medium.
Add H+ as +ve charge to balance the charge on both sides.
Cr2O72- converts into Cr3+ in acidic medium I.e. in H+ medium.
First balance the Cr atom on both sides and then Oxygen atom. H+ is in excess due to acidic medium.
Add H+ as +ve charge to balance the charge on both sides.
Law of definite proportions is given by:- a)Antoine Lavoisier
- b)Joseph Proust
- c)Avogadro
- d)John Dalton
Correct answer is option 'B'. Can you explain this answer?
Law of definite proportions is given by:
a)
Antoine Lavoisier
b)
Joseph Proust
c)
Avogadro
d)
John Dalton
| | Naina Bansal answered |
In chemistry, the law of definite proportion, sometimes called Proust's law or the law of definite composition, or law of constant composition states that a given chemical compound always contains its component elements in fixed ratio (by mass) and does not depend on its source and method of preparation.
KIO3 reacts with KI to liberate iodine and liberated Iodine is titrated with standard hypo solution, The reactions are1. I03– + I– →I2 (valency factor = 5/3)2. I2 + S2032– → S4062– + I– (valency factor = 2)meq of hypo = meq of I2 = meq of I03– + meq of I–I03– react with I– ⇒ meq of I03– = meq of I–Statement-1 : meq of hypo = 2 x meq of I03–Statement-2 : valency factor of I2 in both the equation are different therefore we cannot equate milliequivalents in sequence- a)Statement-1 is True, Statement-2 is True; Statement-2 is a correct explanation for Statement-2.
- b)Statement-1 is True, Statement-2 is True; Statement-2 is NOT a correct explanation for Statement-2.
- c)Statement-2 is True, Statement-2 is False.
- d)Statement-1 is False, Statement-2 is True.
Correct answer is option 'A'. Can you explain this answer?
KIO3 reacts with KI to liberate iodine and liberated Iodine is titrated with standard hypo solution, The reactions are
1. I03– + I– →I2 (valency factor = 5/3)
2. I2 + S2032– → S4062– + I– (valency factor = 2)
meq of hypo = meq of I2 = meq of I03– + meq of I–
I03– react with I– ⇒ meq of I03– = meq of I–
Statement-1 : meq of hypo = 2 x meq of I03–
Statement-2 : valency factor of I2 in both the equation are different therefore we cannot equate milliequivalents in sequence
a)
Statement-1 is True, Statement-2 is True; Statement-2 is a correct explanation for Statement-2.
b)
Statement-1 is True, Statement-2 is True; Statement-2 is NOT a correct explanation for Statement-2.
c)
Statement-2 is True, Statement-2 is False.
d)
Statement-1 is False, Statement-2 is True.
| | Krishna Iyer answered |
The correct answer is option A
Milliequivalent of IO3− is equal to milliequivalent of I−.
Milliequivalent of hypo is equal to milliequivalent of IO3− plus milliequivalent of I−.
The milliequivalent of hypo is twice the milliequivalent of IO3−. It is also twice the milliequivalent of I−.
Milliequivalent of IO3− is equal to milliequivalent of I−.
Milliequivalent of hypo is equal to milliequivalent of IO3− plus milliequivalent of I−.
The milliequivalent of hypo is twice the milliequivalent of IO3−. It is also twice the milliequivalent of I−.
If the true value for a result is 3.00 m and a student records two readings as 3.01 m and 2.99 m, then we can conclude that:- a)the values are neither accurate nor precise
- b)the values are both accurate and precise
- c)the values are precise
- d)the values are accurate
Correct answer is 'B'. Can you explain this answer?
If the true value for a result is 3.00 m and a student records two readings as 3.01 m and 2.99 m, then we can conclude that:
a)
the values are neither accurate nor precise
b)
the values are both accurate and precise
c)
the values are precise
d)
the values are accurate
 | Anjana Desai answered |
Precision indicates how closely repeated measurements match each other.
Accuracy indicates how closely a measurement matches the correct or expected value.
Accuracy indicates how closely a measurement matches the correct or expected value.
Can you explain the answer of this question below:Law of constant composition does not hold good for:- A:Endothermic compounds
- B:Non-stoichiometric compounds
- C:Stoichiometric compounds
- D:Exothermic compounds
The answer is b.
Law of constant composition does not hold good for:
A:
Endothermic compounds
B:
Non-stoichiometric compounds
C:
Stoichiometric compounds
D:
Exothermic compounds
| | Naina Bansal answered |
Law of definite proportion is not true for all types of compounds. The law is not valid for non-stoichiometric compounds.These are compounds whose compositions vary from sample to sample.Mainly inorganic compounds come into this class.An example of these types of compound is wustite, which has the formula FeO, but in actual the stoichiometry in this compound is Fe0.95O. This is so because some of the Fe^+2 gets easily oxidised to Fe^+3 and takes place of Fe^+2 in the lattice. To balance the charge 3 Fe^+2 atoms are being replaced by 2 Fe^+3 atoms. Elements having mixture of stable isotopes also form non-stoichiometric compound. Some of the element position in the crystal structure gets replaced by their isotopes and causes difference in actual composition of the crystal. For example , natural carbon, silicon, and germanium show this type of defect.
One sample of air is found to have 0.03% carbon dioxide and another sample 0.02%. This illustrates that:- a)Air is a compound
- b)Air is an element
- c)Air does not follow the law of constant proportions
- d)Air is a mixture
Correct answer is option 'D'. Can you explain this answer?
One sample of air is found to have 0.03% carbon dioxide and another sample 0.02%. This illustrates that:
a)
Air is a compound
b)
Air is an element
c)
Air does not follow the law of constant proportions
d)
Air is a mixture
| | Raghav Bansal answered |
Air is a mixture.
When we combine more than one type of element, known as a mixture.
The different percentages are showing that these other elements are present.
When we combine more than one type of element, known as a mixture.
The different percentages are showing that these other elements are present.
Match the prefixes present in column I with their multiples in column II and mark the appropriate choice.
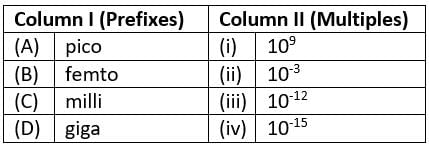
- a)(A)→(i), (B)→(ii), (C)→(iii), (D)→(iv)
- b)(A)→(ii), (B)→(i), (C)→(iv), (D)→(iii)
- c)(A)→(iv), (B)→(iii), (C)→(i), (D)→(ii)
- d)(A)→(iii), (B)→(iv), (C)→(ii), (D)→(i)
Correct answer is option 'D'. Can you explain this answer?
Match the prefixes present in column I with their multiples in column II and mark the appropriate choice.


a)
(A)→(i), (B)→(ii), (C)→(iii), (D)→(iv)
b)
(A)→(ii), (B)→(i), (C)→(iv), (D)→(iii)
c)
(A)→(iv), (B)→(iii), (C)→(i), (D)→(ii)
d)
(A)→(iii), (B)→(iv), (C)→(ii), (D)→(i)
 | Whatsapp Status answered |
D
SI unit of density is- a)kg−1m3
- b)kg m−2
- c)kg m3
- d)kg m−3
Correct answer is option 'D'. Can you explain this answer?
SI unit of density is
a)
kg−1m3
b)
kg m−2
c)
kg m3
d)
kg m−3
 | Kunal Patel answered |
density = mass/volume. SI unit of mass is kg and that of volume is m3
In an experiment it showed that 10 mL of 0.05 M solution of chloride required 10 mL of 0.1 M solution of AgNO3, which of the following will be the formula of the chloride (X stands for the symbol of the element other than chlorine): [NEET Kar. 2013]- a)X2Cl
- b)X2Cl2
- c)XCl2
- d)XCl4
Correct answer is option 'C'. Can you explain this answer?
In an experiment it showed that 10 mL of 0.05 M solution of chloride required 10 mL of 0.1 M solution of AgNO3, which of the following will be the formula of the chloride (X stands for the symbol of the element other than chlorine): [NEET Kar. 2013]
a)
X2Cl
b)
X2Cl2
c)
XCl2
d)
XCl4
 | Pallabi Reddy answered |
Millimoles of solution of chloride = 0.05 × 10 = 0.5
Millimoles of AgNO3 solution = 10 × 0.1 = 1
So, the millimoles of AgNO3 are double than the chloride solution
Millimoles of AgNO3 solution = 10 × 0.1 = 1
So, the millimoles of AgNO3 are double than the chloride solution
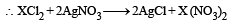
Molecular mass of glucose molecule (C6H12O6) is:- a) 180 g
- b)172 g
- c)182 g
- d)192 g
Correct answer is option 'A'. Can you explain this answer?
Molecular mass of glucose molecule (C6H12O6) is:
a)
180 g
b)
172 g
c)
182 g
d)
192 g
 | Gowri Kulkarni answered |
Molecular mass : The sum of the atomic masses of all the atoms in a molecule of a substance is called the molecular mass of the molecule.
Generally we use relative atomic masses of atoms for calculating the molecular mass of 1 mole of any molecular or ionic substances.
Molecular formula of glucose is C6H12O6
Atomic mass of H = 1
Atomic mass pf C = 12
Atomic mass of O = 16
Molecular mass of C6H12O6 = 12(Atomic mass of Hydrogen) + 6(Atomic mass of carbon) + 6(Atomic mass of oxygen)
= 12 x 1 + 6X12 + 6X16
= 12 + 72 + 96 = 180 u.
A pure substance can only be:- a)A compound
- b)An element
- c)An element or a compound
- d)A heterogeneous mixture
Correct answer is option 'C'. Can you explain this answer?
A pure substance can only be:
a)
A compound
b)
An element
c)
An element or a compound
d)
A heterogeneous mixture
| | Pooja Shah answered |
A pure substance is a substance in which only one type of atom or molecule is present.
Example: Water, Sulphur, Sucrose(pure), Helium, etc.
Thus, a pure substance can only be an element or a compound.
Example: Water, Sulphur, Sucrose(pure), Helium, etc.
Thus, a pure substance can only be an element or a compound.
25.3 g of sodium carbonate, Na2CO3 is dissolved in enough water to make 250 mL of solution. If sodium carbonate dissociates completely, molar concentration of sodium ions, Na+ and carbonate ions, 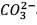 are respectively. (At. mass of carbon = 40)
are respectively. (At. mass of carbon = 40) - a)0.477 M and 0.477 M
- b)0.955 M and 1.910 M
- c)1.910 M and 0.955 M
- d)1.90 M and 1.910 M
Correct answer is option 'C'. Can you explain this answer?
25.3 g of sodium carbonate, Na2CO3 is dissolved in enough water to make 250 mL of solution. If sodium carbonate dissociates completely, molar concentration of sodium ions, Na+ and carbonate ions, 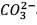 are respectively. (At. mass of carbon = 40)
are respectively. (At. mass of carbon = 40)
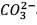 are respectively. (At. mass of carbon = 40)
are respectively. (At. mass of carbon = 40) a)
0.477 M and 0.477 M
b)
0.955 M and 1.910 M
c)
1.910 M and 0.955 M
d)
1.90 M and 1.910 M
 | Knowledge Hub answered |
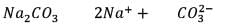
= 2.53/106 2.59 × 10-2 5.9 × 10-2
0.238 × 0.25 = 1.18 × 10-3
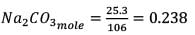
250 ml - 0.238
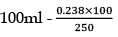
= 0.955
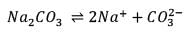
0.955
 0.955
0.955 105 ml of pure water at 4°C saturated with NH3 gas yielded a solution of density 0.9 g/ml and containing 30% NH by mass. Find the valume of resulting NH3 solution. - a)66.67 ml
- b)166.67 ml
- c)133.33 ml
- d)266.67 ml
Correct answer is option 'B'. Can you explain this answer?
105 ml of pure water at 4°C saturated with NH3 gas yielded a solution of density 0.9 g/ml and containing 30% NH by mass. Find the valume of resulting NH3 solution.
a)
66.67 ml
b)
166.67 ml
c)
133.33 ml
d)
266.67 ml
 | EduRev JEE answered |
Given -
volume of water = 105 mL
density of solution = 0.9 g/mL
Calculation -
Since ammonia dissolved in water is 30% by weight, then using mass % formula, we can calculate the amount of ammonia in water
volume of water = 105 mL
density of solution = 0.9 g/mL
Calculation -
Since ammonia dissolved in water is 30% by weight, then using mass % formula, we can calculate the amount of ammonia in water
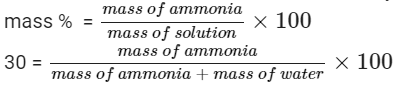
At 4 degree C, density of water = 0.999 g/mL
since density = mass /volume
0.999 = mass/ 105
mass of water = 104.895 g
now, let mass of ammonia be x.
since density = mass /volume
0.999 = mass/ 105
mass of water = 104.895 g
now, let mass of ammonia be x.
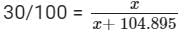
30x + 3146.85 = 100x
70x = 3146.85
x = 44.955 g
Thus, ammonia dissolved in 105 mL water is 44.955 g
Therefore, total mass of solution = mass of ammonia + mass of water
Mass of solution = 44.955 + 104.895
Mass of solution = 149.85 g
So,density of solution = mass of solution / volume of solution
0.9 g/mL = 149.85 g / Volume of solution
Volume of solution = 149.85/ 0.9
Volume of solution = 166.5 mL
70x = 3146.85
x = 44.955 g
Thus, ammonia dissolved in 105 mL water is 44.955 g
Therefore, total mass of solution = mass of ammonia + mass of water
Mass of solution = 44.955 + 104.895
Mass of solution = 149.85 g
So,density of solution = mass of solution / volume of solution
0.9 g/mL = 149.85 g / Volume of solution
Volume of solution = 149.85/ 0.9
Volume of solution = 166.5 mL
An organic compound contains 80% (by wt.) carbon and the remaining percentage of hydrogen. The right option for the empirical formula of this compound is: [Atomic wt. of C is 12, H is 1] (2021)- a)CH3
- b)CH4
- c)CH
- d)CH2
Correct answer is option 'A'. Can you explain this answer?
An organic compound contains 80% (by wt.) carbon and the remaining percentage of hydrogen. The right option for the empirical formula of this compound is: [Atomic wt. of C is 12, H is 1] (2021)
a)
CH3
b)
CH4
c)
CH
d)
CH2
| | Ajay Yadav answered |
An empirical formula represents the simplest whole number ratio of various atoms present in a compound, whereas, the molecular formula shows the exact number of different types of atoms present in a molecule of a compound.
How many moles of lead (II) chloride will be formed from a reaction between 6.5 g of Pbo and 3.2 g of HCl?- a)0.044
- b)0.333
- c)0.011
- d)0.029
Correct answer is option 'D'. Can you explain this answer?
How many moles of lead (II) chloride will be formed from a reaction between 6.5 g of Pbo and 3.2 g of HCl?
a)
0.044
b)
0.333
c)
0.011
d)
0.029
 | Bhavana Chavan answered |
Pbo + 2Hcl
6.5\223
= 2.9 x 10-2
= 2.9x10-2 - x
nx = 9a
PbCl2 + H20
3.2/36.5
= 8.76 x 10-2
8.76 x 10-2 - x
x = 2.9 x 10-2
6.5\223
= 2.9 x 10-2
= 2.9x10-2 - x
nx = 9a
PbCl2 + H20
3.2/36.5
= 8.76 x 10-2
8.76 x 10-2 - x
x = 2.9 x 10-2
Chapter doubts & questions for Some Basic Concepts Of Chemistry - Chemistry Class 11 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Some Basic Concepts Of Chemistry - Chemistry Class 11 in English & Hindi are available as part of NEET exam. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
;
Signup to see your scores go up
within 7 days!
within 7 days!
Takes less than 10 seconds to signup