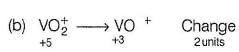All Exams > BMAT > Chemistry for BMAT (Section 2) > All Questions
All questions of Oxidation, Reduction and Redox for BMAT Exam
Which is chlorate (I) ion?
a) 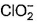
b)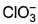
c)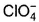
d)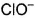
Correct answer i... mores option 'B'. Can you explain this answer?
| | Pooja Shah answered |
- ClO3- : A very reactive inorganic anion.
- The term chlorate can also be used to describe any compound containing the chlorate ion, normally chlorate salts.
- Example: Potassium chlorate, KClO3
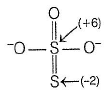
The difference in the oxidation number of the two types of sulphur atoms in Na2S4O6 is .........[HTJEE2011]
Correct answer is '7'. Can you explain this answer?
The difference in the oxidation number of the two types of sulphur atoms in Na2S4O6 is .........
[HTJEE2011]
| | Hrishikesh Sengupta answered |
Difference in oxidation number = 6 - (-1) = 7
Which of the following is not an example of redox reaction?- a)BaCl2 + H2SO4 ⎯→ BaSO4 + 2HCl
- b)Fe2O3 + 3CO ⎯→ 2Fe + 3CO2
- c)2K + F2 ⎯→ 2KF
- d)CuO + H2 ⎯→ Cu + H2O
Correct answer is option 'A'. Can you explain this answer?
Which of the following is not an example of redox reaction?
a)
BaCl2 + H2SO4 ⎯→ BaSO4 + 2HCl
b)
Fe2O3 + 3CO ⎯→ 2Fe + 3CO2
c)
2K + F2 ⎯→ 2KF
d)
CuO + H2 ⎯→ Cu + H2O
| | Raghav Bansal answered |
a) BaCl2 + H2SO4 → BaSO4 + 2HCl is not a redox reaction, as there is no change in the oxidation state of any element.
It is an example of double displacement reactions.
It is an example of double displacement reactions.
The oxidation half reaction for following reaction is
Fe2+(aq) + Cr2O72-(aq) → Fe3+ (aq) + Cr3+(aq)- a)Fe3+(aq) → Fe2+ (aq)
- b)Cr2O72-(aq) → Cr3+(aq)
- c)Cr3+(aq) → Cr2O72-(aq)
- d)Fe2+ (aq) → Fe3+(aq)
Correct answer is option 'D'. Can you explain this answer?
The oxidation half reaction for following reaction is
Fe2+(aq) + Cr2O72-(aq) → Fe3+ (aq) + Cr3+(aq)
Fe2+(aq) + Cr2O72-(aq) → Fe3+ (aq) + Cr3+(aq)
a)
Fe3+(aq) → Fe2+ (aq)
b)
Cr2O72-(aq) → Cr3+(aq)
c)
Cr3+(aq) → Cr2O72-(aq)
d)
Fe2+ (aq) → Fe3+(aq)
| | Geetika Shah answered |
Oxidation half reaction for a reaction is that reaction which gives us the reactant and product formed after the oxidation of the reactant. In this case Fe+2 oxidizes itself to Fe+3 and so the oxidation of Fe+2 is oxidation half reaction. Option d correct.
In the following reaction, 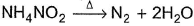 , oxidation number of
, oxidation number of- a)N changes from -2 to +2
- b)N changes from -2 to 0
- c)N in
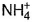 changes from -3 to 0 and that in
changes from -3 to 0 and that in 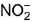 changes from +3 to 0
changes from +3 to 0 - d)N does not change
Correct answer is option 'C'. Can you explain this answer?
In the following reaction, 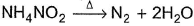 , oxidation number of
, oxidation number of
a)
N changes from -2 to +2
b)
N changes from -2 to 0
c)
N in 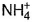 changes from -3 to 0 and that in
changes from -3 to 0 and that in 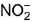 changes from +3 to 0
changes from +3 to 0
d)
N does not change
 | Learners Habitat answered |
In NH4NO2, the oxidation number of N in NH4+ is -3, and of N in NO2- is +3.
In which of the following reactions, equivalent mass of the underlined is equal to molar mass?- a)
- b)
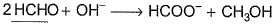
- c)
- d)
Correct answer is option 'A,B'. Can you explain this answer?
In which of the following reactions, equivalent mass of the underlined is equal to molar mass?
a)
b)
c)
d)
| | Vivek answered |
Equivalent mass = molecular mass/(valency in case of elements and charge of cationic part in case of compounds)
For Cl2, valency and for HCHO, charge of cationic part are 1 in magnitude. Hence,
equivalent mass = molecular mass for A and B.
For Cl2, valency and for HCHO, charge of cationic part are 1 in magnitude. Hence,
equivalent mass = molecular mass for A and B.
Oxidation numbers of P in PO4−3, of S in SO42− and that of Cr in Cr2O72− are respectively,- a) +5, +6 and +6
- b)+3, +6 and +5
- c)+5, +3 and +6
- d)-3, +6 and +6
Correct answer is option 'A'. Can you explain this answer?
Oxidation numbers of P in PO4−3, of S in SO42− and that of Cr in Cr2O72− are respectively,
a)
+5, +6 and +6
b)
+3, +6 and +5
c)
+5, +3 and +6
d)
-3, +6 and +6
| | Raghav Bansal answered |
The correct answer is option A
(I) xPO43− ⇒ x + 4 × (−2) = −3
⇒x = −3 + 8 = +5
⇒x = +5
Oxidation number of P = +5
(II) xSO42− ⇒ x + 4 × (−2) = −2
⇒x = −2 + 8
⇒x = +6
Oxidation number of S=+6
(III) xCr2O72− ⇒2x + 7 × (−2) = −2
⇒2x =−2+14
⇒2x=12
⇒x= 12/2 = +6
(I) xPO43− ⇒ x + 4 × (−2) = −3
⇒x = −3 + 8 = +5
⇒x = +5
Oxidation number of P = +5
(II) xSO42− ⇒ x + 4 × (−2) = −2
⇒x = −2 + 8
⇒x = +6
Oxidation number of S=+6
(III) xCr2O72− ⇒2x + 7 × (−2) = −2
⇒2x =−2+14
⇒2x=12
⇒x= 12/2 = +6
Direction (Q. Nos. 17) Choice the correct combination of elements and column I and coloumn II are given as option (a), (b), (c) and (d), out of which ONE option is correct.Match the compounds/ions having underlined atoms of different oxidation number (in Column I) with values (in Column II).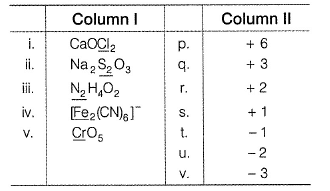


- a)a
- b)b
- c)c
- d)d
Correct answer is option 'B'. Can you explain this answer?
Direction (Q. Nos. 17) Choice the correct combination of elements and column I and coloumn II are given as option (a), (b), (c) and (d), out of which ONE option is correct.
Match the compounds/ions having underlined atoms of different oxidation number (in Column I) with values (in Column II).


a)
a
b)
b
c)
c
d)
d
| | Suresh Iyer answered |
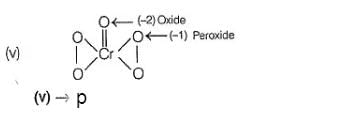
(i) CaOCI2 has
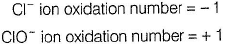
Oxidation number = + 6
Oxidation number = - 2
(ii) → (p,u)
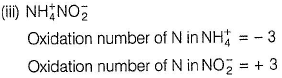
(iii) → (q.v)
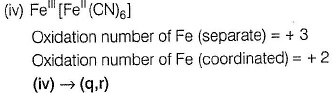
Oxidation number = + 6
Oxidation number = - 2
(ii) → (p,u)
(iii) → (q.v)

Oxidation number of 1/2 is assigned to oxygen atom in- a)superoxides
- b)when oxygen is bonded to fluorine
- c)when oxygen is bonded to metals
- d)peroxidesperoxides
Correct answer is option 'A'. Can you explain this answer?
Oxidation number of 1/2 is assigned to oxygen atom in
a)
superoxides
b)
when oxygen is bonded to fluorine
c)
when oxygen is bonded to metals
d)
peroxidesperoxides
| | Arka Desai answered |
The oxidation number is a concept used in chemistry to keep track of the distribution of electrons in a compound or molecule. It is a measure of the charge that an atom would have if all the shared electrons were assigned to the more electronegative atom in a bond.
In the case of oxygen, its most common oxidation number is -2. However, in certain compounds, such as superoxides and peroxides, the oxidation number of oxygen deviates from -2.
Oxidation number of 1/2 is assigned to the oxygen atom in superoxides. Superoxides are a class of compounds that contain the superoxide ion, O2-. In this ion, each oxygen atom has an oxidation number of -1/2. This is because the oxygen-oxygen bond in the superoxide ion is a single bond with a bond order of 1/2. Therefore, each oxygen atom is assigned an oxidation number of -1/2 to account for the distribution of electrons in the bond.
In the case of peroxides, such as hydrogen peroxide (H2O2), the oxidation number of oxygen is -1. In peroxides, the oxygen-oxygen bond is a single bond with a bond order of 1. Each oxygen atom is assigned an oxidation number of -1 to account for the distribution of electrons in the bond.
When oxygen is bonded to fluorine, the oxidation number of oxygen is -1. Fluorine is the most electronegative element, and therefore, it attracts the shared electrons in the bond more strongly than oxygen. As a result, oxygen is assigned an oxidation number of -1 to account for the unequal distribution of electrons in the bond.
When oxygen is bonded to metals, the oxidation number of oxygen is typically -2. However, there are some exceptions to this rule, such as in certain metal peroxides or superoxides, where the oxidation number of oxygen deviates from -2.
In summary, the oxidation number of 1/2 is assigned to the oxygen atom in superoxides, where the oxygen-oxygen bond is a single bond with a bond order of 1/2. In other compounds, such as peroxides, fluorides, and most metal oxides, the oxidation number of oxygen is typically -1 or -2.
In the case of oxygen, its most common oxidation number is -2. However, in certain compounds, such as superoxides and peroxides, the oxidation number of oxygen deviates from -2.
Oxidation number of 1/2 is assigned to the oxygen atom in superoxides. Superoxides are a class of compounds that contain the superoxide ion, O2-. In this ion, each oxygen atom has an oxidation number of -1/2. This is because the oxygen-oxygen bond in the superoxide ion is a single bond with a bond order of 1/2. Therefore, each oxygen atom is assigned an oxidation number of -1/2 to account for the distribution of electrons in the bond.
In the case of peroxides, such as hydrogen peroxide (H2O2), the oxidation number of oxygen is -1. In peroxides, the oxygen-oxygen bond is a single bond with a bond order of 1. Each oxygen atom is assigned an oxidation number of -1 to account for the distribution of electrons in the bond.
When oxygen is bonded to fluorine, the oxidation number of oxygen is -1. Fluorine is the most electronegative element, and therefore, it attracts the shared electrons in the bond more strongly than oxygen. As a result, oxygen is assigned an oxidation number of -1 to account for the unequal distribution of electrons in the bond.
When oxygen is bonded to metals, the oxidation number of oxygen is typically -2. However, there are some exceptions to this rule, such as in certain metal peroxides or superoxides, where the oxidation number of oxygen deviates from -2.
In summary, the oxidation number of 1/2 is assigned to the oxygen atom in superoxides, where the oxygen-oxygen bond is a single bond with a bond order of 1/2. In other compounds, such as peroxides, fluorides, and most metal oxides, the oxidation number of oxygen is typically -1 or -2.
I− reduces IO3- and I2 and itself oxidised to I2 in acidic medium. Thus, final reaction is- a)
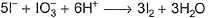
- b)
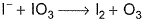
- c)
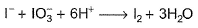
- d)None of the above is correct
Correct answer is option 'A'. Can you explain this answer?
I− reduces IO3- and I2 and itself oxidised to I2 in acidic medium. Thus, final reaction is
a)
b)
c)
d)
None of the above is correct
| | Pooja Shah answered |
To balance oxidation number, cross-multiply by change in oxidation number
The complex [Fe(H2O)5NO]2+ is formed in the ring-test for nitrate ion 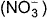 when freshly prepared FeSO4 solution is added to aqueous solution of
when freshly prepared FeSO4 solution is added to aqueous solution of 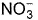 followed by the addition of conc. H2SO4. NO exists as NO+ (nitrosyl).Q. Magnetic moment
followed by the addition of conc. H2SO4. NO exists as NO+ (nitrosyl).Q. Magnetic moment 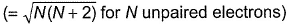 of Fe in the ring is
of Fe in the ring is - a)zero BM
- b)
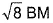
- c)
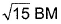
- d)
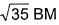
Correct answer is option 'C'. Can you explain this answer?
The complex [Fe(H2O)5NO]2+ is formed in the ring-test for nitrate ion 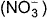 when freshly prepared FeSO4 solution is added to aqueous solution of
when freshly prepared FeSO4 solution is added to aqueous solution of 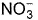 followed by the addition of conc. H2SO4. NO exists as NO+ (nitrosyl).
followed by the addition of conc. H2SO4. NO exists as NO+ (nitrosyl).
Q. Magnetic moment 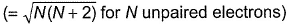 of Fe in the ring is
of Fe in the ring is
a)
zero BM
b)
c)
d)
| | Naina Sharma answered |
Fe2+ is added asFeSO4
Fe+ is formed by charge transfer from NO to Fe2+
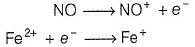

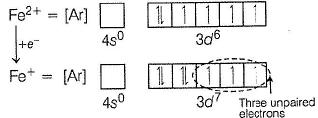
Fe+ has three unpaired electrons (N).
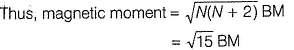
Fe+ is formed by charge transfer from NO to Fe2+
Fe+ has three unpaired electrons (N).
NaHC2O4 is neutralised by NaOH and can also be oxidised by KMnO4 (in acidic medium). Equivalent weight is related to m olecular w eight (M) of NaHC2O4 in these two reactions as- a)M,M
- b)M,M/2
- c)M/2, M/5
- d)M/5, M/2
Correct answer is option 'B'. Can you explain this answer?
NaHC2O4 is neutralised by NaOH and can also be oxidised by KMnO4 (in acidic medium). Equivalent weight is related to m olecular w eight (M) of NaHC2O4 in these two reactions as
a)
M,M
b)
M,M/2
c)
M/2, M/5
d)
M/5, M/2
| | Debanshi Datta answered |
Thus, equivalent weight when reacted with NaOH = M
Direction (Q. Nos. 13 and 14) This section contains 2 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THANT ONE is correct.Q. Which of the following species has/have oxidation number of the metal as + 6 ?- a)
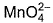
- b)
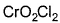
- c)
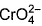
- d)

Correct answer is option 'A,B,C'. Can you explain this answer?
Direction (Q. Nos. 13 and 14) This section contains 2 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THANT ONE is correct.
Q. Which of the following species has/have oxidation number of the metal as + 6 ?
a)
b)
c)
d)
 | Knowledge Hub answered |
To balance the oxygen atom in the given reaction in acidic medium
Cr2O72- (aq) → Cr3+(aq) we- a)Add water (H2O) on right side
- b)Add water (H2O) on left side
- c)Add O on right side
- d)Add O on left side
Correct answer is option 'A'. Can you explain this answer?
To balance the oxygen atom in the given reaction in acidic medium
Cr2O72- (aq) → Cr3+(aq) we
Cr2O72- (aq) → Cr3+(aq) we
a)
Add water (H2O) on right side
b)
Add water (H2O) on left side
c)
Add O on right side
d)
Add O on left side
 | Supriya Senapati answered |
Yes because right side is deficient of oxygen.
Select the set of compounds with oxidation-reduction duality.- a)Cl2, H3PO4, HCHO, HNO2
- b)Cl2, H3PO3, C6H5CHO, H2O2
- c)Br2, H3PO2, CH3CHO, H3PO4
- d)CrO2Cl2, KMnO4, SO3, CO2
Correct answer is option 'B'. Can you explain this answer?
Select the set of compounds with oxidation-reduction duality.
a)
Cl2, H3PO4, HCHO, HNO2
b)
Cl2, H3PO3, C6H5CHO, H2O2
c)
Br2, H3PO2, CH3CHO, H3PO4
d)
CrO2Cl2, KMnO4, SO3, CO2
| | Neha Sharma answered |
Com pounds having oxidising and reducing nature in given reaction are said to have oxidation-reduction duality. Such compounds are said to undergo disproportionation reaction.
(This is called Cannizzaro reaction.)
Note Such compounds have O.N. of the affected atoms intermediate of oxidation part and reduction part
Assign oxidation number to P in NaH2PO4- a)+6
- b)+1
- c)+4
- d)5.0
Correct answer is option 'D'. Can you explain this answer?
Assign oxidation number to P in NaH2PO4
a)
+6
b)
+1
c)
+4
d)
5.0
 | Raksha Nambiar answered |
Oxidation state of P in NaH2PO4
Equivalent weight of H3PO2 in a reaction is found to be half of its molecular weight. It can be due to its- a)reaction of two H+ ion
- b)oxidation of H3PO3
- c)oxidation of H3PO4
- d)reduction of PH3
Correct answer is option 'B'. Can you explain this answer?
Equivalent weight of H3PO2 in a reaction is found to be half of its molecular weight. It can be due to its
a)
reaction of two H+ ion
b)
oxidation of H3PO3
c)
oxidation of H3PO4
d)
reduction of PH3
 | Anisha Chauhan answered |
An equivalent weight of H3PO2 when it disproportionates into PH3 and H3PO3 is
For disproportionation reactions, use a trick to find equivalent mass; E = E1 + E2; where E1 and E2 are equivalent masses of oxidation and reduction half-reactions of the same element.Now, find the n-factor(total change in oxidation number per molecule) and then equivalent mass = molar mass/n-factor.
So, when H3PO2 changes into PH3, the oxidation state of phosphorous changes from +1 to -3 so as n-factor is 4. Also when H3PO2 changes into H3PO3, the oxidation state of phosphorous changes from +1 to +5 so as n-factor is again 4.
Now equivalent mass of H3PO2, E =(M/4) + (M/4) = M/2
Hence, the correct option is b.
In this method, the two half equations are balanced separately and then added together to give balanced equation- a)Reluctant method
- b)Oxidizing agent method
- c)Reducing agent method
- d)Half reaction method
Correct answer is option 'D'. Can you explain this answer?
In this method, the two half equations are balanced separately and then added together to give balanced equation
a)
Reluctant method
b)
Oxidizing agent method
c)
Reducing agent method
d)
Half reaction method
| | Kavita Joshi answered |
In the ion-electron method (also called the half-reaction method), the redox equation is separated into two half-equations - one for oxidation and one for reduction. Each of these half-reactions is balanced separately and then combined to give the balanced redox equation.
In which of the following reactions oxidation number of chromium has been affected?- a)
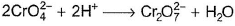
- b)
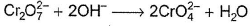
- c)
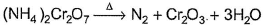
- d)

Correct answer is option 'C'. Can you explain this answer?
In which of the following reactions oxidation number of chromium has been affected?
a)
b)
c)
d)
 | Sankar Bose answered |
Oxidation number of Cr changes from +6 to +3.
Direction (Q. Nos. 16-18) This section contains 4 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONE or MORE THANT ONE is correct.Q. Sulphate (IV) ion reduces chromate (VI) to green coloured salt. Thus,- a) Cr (III) is formed
- b)Equivalent mass of chromate (VI) = M/3
- c) Equivalent mass of sulphate (IV) =M/2
- d) Oxidation product is sulphate (VI)
Correct answer is option 'A,B,C,D'. Can you explain this answer?
Direction (Q. Nos. 16-18) This section contains 4 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONE or MORE THANT ONE is correct.
choices (a), (b), (c) and (d), out of which ONE or MORE THANT ONE is correct.
Q. Sulphate (IV) ion reduces chromate (VI) to green coloured salt. Thus,
a)
Cr (III) is formed
b)
Equivalent mass of chromate (VI) = M/3
c)
Equivalent mass of sulphate (IV) =M/2
d)
Oxidation product is sulphate (VI)
| | Amrita Choudhary answered |
Thus, (a) is correct.
The number of moles of K2Cr2O7 that will be needed to react completely with one mole of ferric sulphite in acidic medium is- a)0.4
- b)0.6
- c)0.8
- d)1.0
Correct answer is option 'D'. Can you explain this answer?
The number of moles of K2Cr2O7 that will be needed to react completely with one mole of ferric sulphite in acidic medium is
a)
0.4
b)
0.6
c)
0.8
d)
1.0
| | Pranavi Banerjee answered |
To determine the number of moles of K2Cr2O7 needed to react completely with one mole of ferric sulphite in acidic medium, we need to first write and balance the chemical equation for the reaction.
The balanced equation for the reaction between K2Cr2O7 and Fe2(SO3)3 in acidic medium is as follows:
K2Cr2O7 + 14H+ + 6Fe2(SO3)3 → 2K+ + 2Cr3+ + 7H2O + 6Fe3+ + 6SO4^2-
From the balanced equation, we can see that the stoichiometric ratio between K2Cr2O7 and Fe2(SO3)3 is 1:6. This means that 1 mole of K2Cr2O7 reacts with 6 moles of Fe2(SO3)3.
Since we are given that we have 1 mole of Fe2(SO3)3, we can calculate the number of moles of K2Cr2O7 needed by using the stoichiometric ratio.
Number of moles of K2Cr2O7 = 1 mole of Fe2(SO3)3 × (1 mole of K2Cr2O7 / 6 moles of Fe2(SO3)3)
Simplifying the expression, we get:
Number of moles of K2Cr2O7 = 1/6 moles
Therefore, the correct answer is option 'D', 1.0 mole.
The balanced equation for the reaction between K2Cr2O7 and Fe2(SO3)3 in acidic medium is as follows:
K2Cr2O7 + 14H+ + 6Fe2(SO3)3 → 2K+ + 2Cr3+ + 7H2O + 6Fe3+ + 6SO4^2-
From the balanced equation, we can see that the stoichiometric ratio between K2Cr2O7 and Fe2(SO3)3 is 1:6. This means that 1 mole of K2Cr2O7 reacts with 6 moles of Fe2(SO3)3.
Since we are given that we have 1 mole of Fe2(SO3)3, we can calculate the number of moles of K2Cr2O7 needed by using the stoichiometric ratio.
Number of moles of K2Cr2O7 = 1 mole of Fe2(SO3)3 × (1 mole of K2Cr2O7 / 6 moles of Fe2(SO3)3)
Simplifying the expression, we get:
Number of moles of K2Cr2O7 = 1/6 moles
Therefore, the correct answer is option 'D', 1.0 mole.
Intensity of blue colour increases gradually when _________________- a)copper rod is dipped in silver nitrate solution
- b)silver rod is dipped in copper nitrate solution
- c)zinc rod is dipped in silver solution
- d)copper rod is dipped in zinc rod solution
Correct answer is option 'A'. Can you explain this answer?
Intensity of blue colour increases gradually when _________________
a)
copper rod is dipped in silver nitrate solution
b)
silver rod is dipped in copper nitrate solution
c)
zinc rod is dipped in silver solution
d)
copper rod is dipped in zinc rod solution
 | EduRev NEET answered |
When a copper rod is dipped in silver nitrate solution, a redox reaction occurs between Copper and an aqueous solution of silver nitrate.
- So the intensity of blue colour increases gradually as silver deposits on the rod.
The oxidation number of oxygen in most compounds is- a)-3
- b)1
- c)4
- d)-2.0
Correct answer is option 'D'. Can you explain this answer?
The oxidation number of oxygen in most compounds is
a)
-3
b)
1
c)
4
d)
-2.0
| | Niharika Nair answered |
Oxidation number of oxygen in most compounds is -2.
Explanation:
- Oxidation number is the number assigned to an atom to indicate its degree of oxidation or loss/gain of electrons.
- Oxygen is a highly electronegative element, meaning it has a strong tendency to attract electrons.
- In most compounds, oxygen has an oxidation number of -2 because it tends to gain electrons to achieve a stable octet configuration (8 valence electrons).
- For example, in water (H2O), each hydrogen atom has an oxidation number of +1 and the oxygen atom has an oxidation number of -2, which balances out the charge to zero.
- There are some exceptions to this rule, such as in peroxides where oxygen has an oxidation number of -1, and in compounds with more electronegative elements where oxygen may have a positive oxidation number.
- Overall, the oxidation number of oxygen in most compounds is -2.
Explanation:
- Oxidation number is the number assigned to an atom to indicate its degree of oxidation or loss/gain of electrons.
- Oxygen is a highly electronegative element, meaning it has a strong tendency to attract electrons.
- In most compounds, oxygen has an oxidation number of -2 because it tends to gain electrons to achieve a stable octet configuration (8 valence electrons).
- For example, in water (H2O), each hydrogen atom has an oxidation number of +1 and the oxygen atom has an oxidation number of -2, which balances out the charge to zero.
- There are some exceptions to this rule, such as in peroxides where oxygen has an oxidation number of -1, and in compounds with more electronegative elements where oxygen may have a positive oxidation number.
- Overall, the oxidation number of oxygen in most compounds is -2.
When a zinc rod is kept in a copper nitrate solution what happens?- a)zinc is deposited on copper
- b)copper is deposited in the beaker
- c)zinc is deposited in the beaker
- d)copper is deposited on zinc
Correct answer is option 'D'. Can you explain this answer?
When a zinc rod is kept in a copper nitrate solution what happens?
a)
zinc is deposited on copper
b)
copper is deposited in the beaker
c)
zinc is deposited in the beaker
d)
copper is deposited on zinc
 | EduRev NEET answered |
When zinc is placed in copper nitrate solution the intensity of the blue colour is produced and copper iron is deposited on zinc.
- This is a Redox reaction between zinc and an aqueous solution of copper nitrate occurring in a beaker.
Reduction is defined in terms ofI. electronation and hydrogenation
II. deelectronation and gain of oxygen
III. increase in oxidation number
IV. decrease in oxidation numberSelect the correct terms- a)I and IV
- b)I and III
- c)II and II
- d)I and II
Correct answer is option 'A'. Can you explain this answer?
Reduction is defined in terms of
I. electronation and hydrogenation
II. deelectronation and gain of oxygen
III. increase in oxidation number
IV. decrease in oxidation number
II. deelectronation and gain of oxygen
III. increase in oxidation number
IV. decrease in oxidation number
Select the correct terms
a)
I and IV
b)
I and III
c)
II and II
d)
I and II
 | Sharmila Sharma answered |
Gain of hydrogen , electron and decrease in oxidation number is reduction
The process in which the strength of an unknown solution is calculated using a known standard solution.- a)Titration
- b)Oxidation
- c)Reduction
- d)None of these
Correct answer is option 'A'. Can you explain this answer?
The process in which the strength of an unknown solution is calculated using a known standard solution.
a)
Titration
b)
Oxidation
c)
Reduction
d)
None of these
| | Nandini Iyer answered |
A titration is a technique used to work out the concentration of an unknown solution based on its chemical reaction with a solution of known concentration. The process usually involves adding the known solution (the titrant) to a known quantity of the unknown solution (the analyte) until the reaction is complete.
Consider the elements: Cs, Ne, I and F. Identify the element(s) that exhibits only negative oxidation state- a)F
- b)I
- c)s
- d)Cs and F
Correct answer is option 'A'. Can you explain this answer?
Consider the elements: Cs, Ne, I and F. Identify the element(s) that exhibits only negative oxidation state
a)
F
b)
I
c)
s
d)
Cs and F
 | Sinjini Datta answered |
F has negative oxidation state as it is very electro negative.
The correct order of N-compounds in its decreasing order of oxidation states is- a)HNO3, NO, N2, NH4Cl
- b)HNO3, NO, NH4Cl, N2
- c)HNO3, NH4Cl, NO, N2
- d)NH4Cl, N2, NO, HN03
Correct answer is option 'A'. Can you explain this answer?
The correct order of N-compounds in its decreasing order of oxidation states is
a)
HNO3, NO, N2, NH4Cl
b)
HNO3, NO, NH4Cl, N2
c)
HNO3, NH4Cl, NO, N2
d)
NH4Cl, N2, NO, HN03
 | Infinity Academy answered |
To determine the decreasing order of oxidation states of nitrogen in the given compounds, we need to find the oxidation state of nitrogen in each compound:
- HNO3 (Nitric acid): Oxidation state of nitrogen: +5
- NO (Nitric oxide): Oxidation state of nitrogen: +2
- N2 (Dinitrogen): Oxidation state of nitrogen: 0
- NH4Cl (Ammonium chloride): Oxidation state of nitrogen: -3
Now, let's arrange these compounds in decreasing order of oxidation states:
- HNO3: +5
- NO: +2
- N2: 0
- NH4Cl: -3
So, the correct order in decreasing oxidation state is:
HNO3, NO, N2, NH4Cl
Which of the following is true as per metal activity series?- a)Zn>Cu>Ag
- b)Zn<Cu<Ag
- c)Zn>Ag>Cu
- d)Zn<Ag<Cu
Correct answer is option 'A'. Can you explain this answer?
Which of the following is true as per metal activity series?
a)
Zn>Cu>Ag
b)
Zn<Cu<Ag
c)
Zn>Ag>Cu
d)
Zn<Ag<Cu
 | EduRev NEET answered |
Metal activity series or electrochemical series is a series in the decreasing order of metals which are active during a chemical reaction comparatively with each other.
- Here, Zinc’s activity is greater than Copper’s activity and Copper’s activity is greater than that of silver.
The more positive the value of E0, the greater is the tendency of the species to get reduced. Using the standard electrode potential of redox couples given below find out which of the following is the strongest oxidising agent. E0values : Fe3 + / Fe2+ = +0.77; I2(s)/l- = +0.54; cu2+/ Cu = +0.34; Ag+ / Ag = +0.80V- a)Ag+
- b)Fe3+
- c)I2 (s)
- d)Cu2+
Correct answer is option 'A'. Can you explain this answer?
The more positive the value of E0, the greater is the tendency of the species to get reduced. Using the standard electrode potential of redox couples given below find out which of the following is the strongest oxidising agent.
E0values : Fe3 + / Fe2+ = +0.77; I2(s)/l- = +0.54; cu2+/ Cu = +0.34; Ag+ / Ag = +0.80V
a)
Ag+
b)
Fe3+
c)
I2 (s)
d)
Cu2+
 | Mansi Mukherjee answered |
Oxidation number of H is not always +1 . It can be -1 , 0.
In the following reaction,
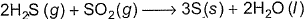
One equivalent of H2S (g)will reduce- a)1 Mole SO2
- b)0.5 Mole SO2
- c)0.25 Mole SO2
- d)2 Mole SO2
Correct answer is option 'C'. Can you explain this answer?
In the following reaction,
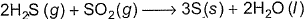
One equivalent of H2S (g)will reduce
One equivalent of H2S (g)will reduce
a)
1 Mole SO2
b)
0.5 Mole SO2
c)
0.25 Mole SO2
d)
2 Mole SO2
 | Rithika Khanna answered |
Change in oxidation number of S of H2S = 2 units
Change in oxidation number of S of SO2 = 4 units
Then one mole H2S = 2 equivalents H2S
One mole SO2 = 4 equivalent SO2
One equivalent H23 one equivalent SO2 = 0.25 mole SO2
The number of moles of KMnO4 that will be needed to react with one mole of ferrous sulphite in acidic solution is- a)0.4
- b)0.6
- c)0.8
- d)1.0
Correct answer is option 'B'. Can you explain this answer?
The number of moles of KMnO4 that will be needed to react with one mole of ferrous sulphite in acidic solution is
a)
0.4
b)
0.6
c)
0.8
d)
1.0
 | Ameya Pillai answered |
To determine the number of moles of KMnO4 needed to react with one mole of ferrous sulphite in acidic solution, we need to balance the chemical equation and use stoichiometry.
The balanced chemical equation for the reaction between KMnO4 and ferrous sulphite in acidic solution is as follows:
5FeSO3 + 2KMnO4 + 8H2SO4 → 5Fe2(SO4)3 + 2MnSO4 + K2SO4 + 8H2O
From the balanced equation, we can see that 2 moles of KMnO4 react with 5 moles of FeSO3. Therefore, the stoichiometric ratio is 2:5.
Now, let's calculate the number of moles of KMnO4 needed.
Given that we have 1 mole of FeSO3, we can set up the following proportion:
2 moles KMnO4 / 5 moles FeSO3 = x moles KMnO4 / 1 mole FeSO3
Cross-multiplying and solving for x, we get:
2 moles KMnO4 = 5 moles FeSO3 * x moles KMnO4
x moles KMnO4 = (2 moles KMnO4 * 1 mole FeSO3) / 5 moles FeSO3
x moles KMnO4 = 0.4 moles KMnO4
Therefore, the number of moles of KMnO4 that will be needed to react with one mole of ferrous sulphite in acidic solution is 0.4 moles, which corresponds to option B.
The balanced chemical equation for the reaction between KMnO4 and ferrous sulphite in acidic solution is as follows:
5FeSO3 + 2KMnO4 + 8H2SO4 → 5Fe2(SO4)3 + 2MnSO4 + K2SO4 + 8H2O
From the balanced equation, we can see that 2 moles of KMnO4 react with 5 moles of FeSO3. Therefore, the stoichiometric ratio is 2:5.
Now, let's calculate the number of moles of KMnO4 needed.
Given that we have 1 mole of FeSO3, we can set up the following proportion:
2 moles KMnO4 / 5 moles FeSO3 = x moles KMnO4 / 1 mole FeSO3
Cross-multiplying and solving for x, we get:
2 moles KMnO4 = 5 moles FeSO3 * x moles KMnO4
x moles KMnO4 = (2 moles KMnO4 * 1 mole FeSO3) / 5 moles FeSO3
x moles KMnO4 = 0.4 moles KMnO4
Therefore, the number of moles of KMnO4 that will be needed to react with one mole of ferrous sulphite in acidic solution is 0.4 moles, which corresponds to option B.
The oxidation number of an element in a compound is evaluated on the basis of certain rules. Which of the following rules is not correct in this respect?- a)The algebraic sum of all the oxidation numbers in a compound is zero.
- b)In all its compounds, the oxidation number of fluorine is – 1.
- c)An element in the free or the uncombined state bears oxidation number zero.
- d)The oxidation number of hydrogen is always +1.
Correct answer is option 'D'. Can you explain this answer?
The oxidation number of an element in a compound is evaluated on the basis of certain rules. Which of the following rules is not correct in this respect?
a)
The algebraic sum of all the oxidation numbers in a compound is zero.
b)
In all its compounds, the oxidation number of fluorine is – 1.
c)
An element in the free or the uncombined state bears oxidation number zero.
d)
The oxidation number of hydrogen is always +1.
 | Ishani Mehta answered |
as oxygen is more electronegative than Cl,Br and I. So they have positive oxidation state.
The highest value of oxidation number changes from 1 to 7- a)in the atoms of transition elements
- b)the first three groups
- c)In alkaline earth metals
- d)across the third period in the periodic table
Correct answer is option 'D'. Can you explain this answer?
The highest value of oxidation number changes from 1 to 7
a)
in the atoms of transition elements
b)
the first three groups
c)
In alkaline earth metals
d)
across the third period in the periodic table
 | Sai Mishra answered |
the highest value of oxidation number changes from 1 to 7.
The oxidation number of Cl in Cl2O7 is- a)+ 3
- b)+ 5
- c)+ 7
- d)– 7
Correct answer is option 'C'. Can you explain this answer?
The oxidation number of Cl in Cl2O7 is
a)
+ 3
b)
+ 5
c)
+ 7
d)
– 7
| | Rajeev Banerjee answered |
Understanding the Compound Cl2O7
To find the oxidation state of chlorine (Cl) in the compound Cl2O7, we need to analyze its structure and the general rules for oxidation states.
Oxidation State of Oxygen
- In most compounds, the oxidation state of oxygen is -2.
- Since there are 7 oxygen atoms in Cl2O7, the total contribution from oxygen is:
- 7 atoms × (-2) = -14
Calculating the Total Oxidation State
- Let the oxidation state of chlorine be x.
- Since there are 2 chlorine atoms, their total contribution to the oxidation state is 2x.
- The overall charge of the compound is neutral (0), leading to the equation:
- 2x + (-14) = 0
Solve for x
- Rearranging the equation gives:
- 2x = 14
- x = 14 / 2
- x = +7
Conclusion
- Therefore, the oxidation number of Cl in Cl2O7 is +7, which corresponds to option 'C'.
This analysis shows that chlorine exhibits a +7 oxidation state, a characteristic of its higher oxidation states in oxoacids and related compounds.
To find the oxidation state of chlorine (Cl) in the compound Cl2O7, we need to analyze its structure and the general rules for oxidation states.
Oxidation State of Oxygen
- In most compounds, the oxidation state of oxygen is -2.
- Since there are 7 oxygen atoms in Cl2O7, the total contribution from oxygen is:
- 7 atoms × (-2) = -14
Calculating the Total Oxidation State
- Let the oxidation state of chlorine be x.
- Since there are 2 chlorine atoms, their total contribution to the oxidation state is 2x.
- The overall charge of the compound is neutral (0), leading to the equation:
- 2x + (-14) = 0
Solve for x
- Rearranging the equation gives:
- 2x = 14
- x = 14 / 2
- x = +7
Conclusion
- Therefore, the oxidation number of Cl in Cl2O7 is +7, which corresponds to option 'C'.
This analysis shows that chlorine exhibits a +7 oxidation state, a characteristic of its higher oxidation states in oxoacids and related compounds.
In the reaction of metallic cobalt placed in nickel sulphate solution, therein is a competition for release of electrons At equilibrium, chemical tests reveal that both Ni+2 (aq) and Co+2 (aq) are present at moderate concentrations. The result is that:- a)Only one reactant and one product is greatly favoured.
- b)Only [Co(s) and Ni+2 (aq)] are favoured
- c)Only Co+2 (aq) and Ni (s)] are favoured
- d)neither the reactants nor the products [are greatly favoured.
Correct answer is option 'D'. Can you explain this answer?
In the reaction of metallic cobalt placed in nickel sulphate solution, therein is a competition for release of electrons At equilibrium, chemical tests reveal that both Ni+2 (aq) and Co+2 (aq) are present at moderate concentrations. The result is that:
a)
Only one reactant and one product is greatly favoured.
b)
Only [Co(s) and Ni+2 (aq)] are favoured
c)
Only Co+2 (aq) and Ni (s)] are favoured
d)
neither the reactants nor the products [are greatly favoured.
 | Arya Reddy answered |
The reaction of metallic cobalt in a nickel sulfate solution involves a competition for the release of electrons. This means that the cobalt metal can react with the nickel ions in the solution, or the nickel can deposit on the cobalt metal.
At equilibrium, the reaction has balanced out with no net change in the concentration of the reactants and products. The fact that both Ni+2 (aq) and Co+2 (aq) are present at moderate concentrations at equilibrium signifies that neither forward nor reverse reactions are greatly favoured.
- A: This option is incorrect because both reactants and products are present in moderate concentrations, indicating that neither is greatly favoured.
- B: This statement is not correct either. Even though Co (s) and Ni+2 (aq) are part of the reaction, the fact that Co+2 (aq) is also present at moderate concentrations shows that they are not the only favoured species.
- C: This option is also incorrect. Even though Co+2 (aq) and Ni (s) are part of the reaction, the fact that Ni+2 (aq) is also present at moderate concentrations shows that they are not the only favoured species.
- D: This is the correct answer. When a reaction is at equilibrium, it means that the rate of the forward reaction equals the rate of the reverse reaction. Therefore, neither the reactants nor the products are greatly favoured. In other words, the concentrations of the reactants and products remain constant over time
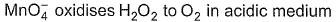
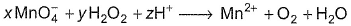 Coefficient x, y and z are respectively
Coefficient x, y and z are respectively

- a)a
- b)b
- c)c
- d)d
Correct answer is option 'C'. Can you explain this answer?
Coefficient x, y and z are respectively

a)
a
b)
b
c)
c
d)
d
| | Hansa Sharma answered |
Cross -multiply by change in oxidation number and balance H by H+ ions.
Thus, x = 2, y = 5 and z = 6
Consider the following experimental facts,I. When Cl2 gas is passed into Kl solution containing CHCI3, violet colour appears in CHCI3 layer.
II. When Cl2 gas is passed into KBr solution containing CHCI3, orange colour appears in CHCI3 layer.
III. When Cl2 gas is passed into a solution containing KBr, Kl and KCI, containing CHCI3, violet colour appears in CHCI3 layer.Select the correct experimental facts.- a)I and II
- b)I and III
- c)III Only
- d)II and III
Correct answer is option 'D'. Can you explain this answer?
Consider the following experimental facts,
I. When Cl2 gas is passed into Kl solution containing CHCI3, violet colour appears in CHCI3 layer.
II. When Cl2 gas is passed into KBr solution containing CHCI3, orange colour appears in CHCI3 layer.
III. When Cl2 gas is passed into a solution containing KBr, Kl and KCI, containing CHCI3, violet colour appears in CHCI3 layer.
II. When Cl2 gas is passed into KBr solution containing CHCI3, orange colour appears in CHCI3 layer.
III. When Cl2 gas is passed into a solution containing KBr, Kl and KCI, containing CHCI3, violet colour appears in CHCI3 layer.
Select the correct experimental facts.
a)
I and II
b)
I and III
c)
III Only
d)
II and III
| | Poulomi Singh answered |
Oxidising power of F2 > CI2 > Br2 > l2
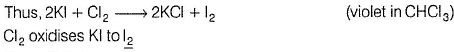
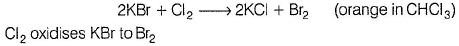
Thus, (II) is true.
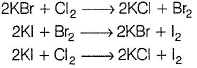
In experiment (III), orange colour of Br2 does not appear, as it also oxidises Kl to l2. Hence , only violet colour appears in CHCI3 layer. Thus, (III) is also true.
Thus, (II) is true.
In experiment (III), orange colour of Br2 does not appear, as it also oxidises Kl to l2. Hence , only violet colour appears in CHCI3 layer. Thus, (III) is also true.
Direction (Q. Nos. 1-18) This section contains 18 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.Q. Oxidation can be defined as the termsI. gain of electron and hydrogen
II. gain of oxygen and loss of electron
III. increase in oxidation number
IV. decrease in oxidation numberSelect the correct terms- a)I and II
- b)II and III
- c)I and III
- d)I and IV
Correct answer is option 'B'. Can you explain this answer?
Direction (Q. Nos. 1-18) This section contains 18 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Q. Oxidation can be defined as the terms
I. gain of electron and hydrogen
II. gain of oxygen and loss of electron
III. increase in oxidation number
IV. decrease in oxidation number
II. gain of oxygen and loss of electron
III. increase in oxidation number
IV. decrease in oxidation number
Select the correct terms
a)
I and II
b)
II and III
c)
I and III
d)
I and IV
 | Arpita Nambiar answered |
Oxidation number increases, oxygen gained and loss of electrons is oxidation.
the oxidation number of sulphur in S8,S2F2,H2S respectively are- a)0,+1 and -2
- b)+2, +1 and -2
- c)0, +1 and +2
- d)-2, +1 and -2
Correct answer is option 'A'. Can you explain this answer?
the oxidation number of sulphur in S8,S2F2,H2S respectively are
a)
0,+1 and -2
b)
+2, +1 and -2
c)
0, +1 and +2
d)
-2, +1 and -2
| | Sparsh Datta answered |
(i) Oxidation state of element in its free state is zero.
(ii) Sum of oxidation states of all atoms in compound is zero.
O.N of S in S8=0; O.N of S in S2F2=+1
O.N of S in H2S=-2
Which of the following atom has been assigned only single oxidation number?- a)H
- b)O
- c)N
- d)F
Correct answer is option 'D'. Can you explain this answer?
Which of the following atom has been assigned only single oxidation number?
a)
H
b)
O
c)
N
d)
F
 | Amrita Kumar answered |
Fluorine is the most electronegative element. It can gain one electron
F+ e- → F
-
Oxidation number = -1
Due to very high (IE), it cannot lose electron.
(a) HH- (-1), (H+ (+1)
(b) O -2 in oxide
-1 in peroxide
+2 in OF2
(c) N - 3 in N2O3, NH3
+3 in HNO2,
+5 in HNO3,
+ 1 in N2O
In balancing the half-reaction, CN- → CNO-The number of electrons that must be added is - a)0
- b)4 on ther ight
- c)4 on the left
- d)2 on the right
Correct answer is option 'D'. Can you explain this answer?
In balancing the half-reaction, CN- → CNO-
The number of electrons that must be added is
a)
0
b)
4 on ther ight
c)
4 on the left
d)
2 on the right
| | Rutuja Khanna answered |
Understanding the Half-Reaction
In this half-reaction, we are looking at the transformation from cyanide ion (CN-) to the cyanate ion (CNO-). To balance this reaction, we need to account for both charge and mass.
Analyzing the Charges
- Cyanide Ion (CN-): The charge is -1.
- Cyanate Ion (CNO-): The charge is -1.
These ions have the same charge, but we also need to consider the atoms involved.
Balancing the Atoms
- Atoms in CN-: 1 Carbon (C) and 1 Nitrogen (N).
- Atoms in CNO-: 1 Carbon (C), 1 Nitrogen (N), and 1 Oxygen (O).
In this case, we see that while the number of Nitrogen and Carbon atoms remains the same, we need to add an Oxygen atom to balance the transformation from CN- to CNO-.
Adding Electrons
To balance the change in oxidation state and ensure that both sides of the equation have the same charge:
- Oxygen Addition: When adding oxygen, we must also add electrons to maintain charge balance.
- Oxygen typically requires 2 electrons to form. Thus, we add 2 electrons to the right side of the half-reaction to balance the charges.
Final Balanced Half-Reaction
The balanced half-reaction can be expressed as follows:
CN- + 2 electrons → CNO-
This shows that 2 electrons must be added to the right side to balance the reaction.
Conclusion
The correct answer is indeed option 'D': 2 electrons on the right. This is necessary to account for the addition of the oxygen atom and to maintain charge balance in the half-reaction.
In this half-reaction, we are looking at the transformation from cyanide ion (CN-) to the cyanate ion (CNO-). To balance this reaction, we need to account for both charge and mass.
Analyzing the Charges
- Cyanide Ion (CN-): The charge is -1.
- Cyanate Ion (CNO-): The charge is -1.
These ions have the same charge, but we also need to consider the atoms involved.
Balancing the Atoms
- Atoms in CN-: 1 Carbon (C) and 1 Nitrogen (N).
- Atoms in CNO-: 1 Carbon (C), 1 Nitrogen (N), and 1 Oxygen (O).
In this case, we see that while the number of Nitrogen and Carbon atoms remains the same, we need to add an Oxygen atom to balance the transformation from CN- to CNO-.
Adding Electrons
To balance the change in oxidation state and ensure that both sides of the equation have the same charge:
- Oxygen Addition: When adding oxygen, we must also add electrons to maintain charge balance.
- Oxygen typically requires 2 electrons to form. Thus, we add 2 electrons to the right side of the half-reaction to balance the charges.
Final Balanced Half-Reaction
The balanced half-reaction can be expressed as follows:
CN- + 2 electrons → CNO-
This shows that 2 electrons must be added to the right side to balance the reaction.
Conclusion
The correct answer is indeed option 'D': 2 electrons on the right. This is necessary to account for the addition of the oxygen atom and to maintain charge balance in the half-reaction.
Chapter doubts & questions for Oxidation, Reduction and Redox - Chemistry for BMAT (Section 2) 2026 is part of BMAT exam preparation. The chapters have been prepared according to the BMAT exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for BMAT 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Oxidation, Reduction and Redox - Chemistry for BMAT (Section 2) in English & Hindi are available as part of BMAT exam. Download more important topics, notes, lectures and mock test series for BMAT Exam by signing up for free.
Chemistry for BMAT (Section 2)136 videos|126 docs|121 tests |
Top Courses BMAT
;
Signup on EduRev and stay on top of your study goals
10M+ students crushing their study goals daily