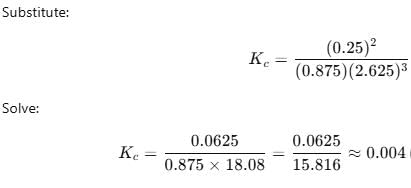All Exams > NEET > 4 Months Preparation for NEET > All Questions
All questions of Equilibrium - Chemistry for NEET Exam
Ca(HCO3)2 is strongly heated and after equilibrium is attained, temperature changed to 25° C.
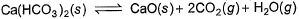
Kp = 36 (pressure taken in atm)
Thus, pressure set up due to CO2 is- a)36 atm
- b)18 atm
- c)12 atm
- d)6 atm
Correct answer is 'D'. Can you explain this answer?
Ca(HCO3)2 is strongly heated and after equilibrium is attained, temperature changed to 25° C.
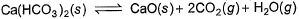
Kp = 36 (pressure taken in atm)
Thus, pressure set up due to CO2 is
Kp = 36 (pressure taken in atm)
Thus, pressure set up due to CO2 is
a)
36 atm
b)
18 atm
c)
12 atm
d)
6 atm
| | Mira Joshi answered |
The reaction is as follow:-
Ca(HCO3)2(s)⇌CaO(s) + 2CO2 (g) + H2O(g)
At 25° C H2O goes in liquid state
Kp = (PCaO)1×(PCO2)2
(PCa(HCO3)2)
Since, Ca(HCO3)2, CaO and H2O are not in gaseous state, so their partial pressure is taken 1.
Putting all values, we have
36 = (PCO2)2
Or PCO2 = 6 atm
Ca(HCO3)2(s)⇌CaO(s) + 2CO2 (g) + H2O(g)
At 25° C H2O goes in liquid state
Kp = (PCaO)1×(PCO2)2
(PCa(HCO3)2)
Since, Ca(HCO3)2, CaO and H2O are not in gaseous state, so their partial pressure is taken 1.
Putting all values, we have
36 = (PCO2)2
Or PCO2 = 6 atm
Following equilibrium is set up at 298 K in a 1 L flask.
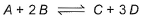
If one starts with 2 moles of A and 1 mole of B, it is found that moles of B and D are equal.Thus Kc is - a)9.0
- b)15.0
- c)3.0
- d)0.0667
Correct answer is option 'B'. Can you explain this answer?
Following equilibrium is set up at 298 K in a 1 L flask.
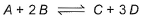
If one starts with 2 moles of A and 1 mole of B, it is found that moles of B and D are equal.Thus Kc is
If one starts with 2 moles of A and 1 mole of B, it is found that moles of B and D are equal.Thus Kc is
a)
9.0
b)
15.0
c)
3.0
d)
0.0667
 | Sushil Kumar answered |
For the equilibrium reaction:
A+2B ⇌ 2C+D
volume of flask = 1L
Initial moles of A = 2 mol
initial concentration of A=[A]i = 2 M
initial mole of B = 1 mol
[B]i = 1 M
[A]eq = 2-x, [B]eq = 1-2x, [C]eq = x, [D]eq = 3x
Given [D]eq = 1 * 1L
= 1 M
Thus x = 1M
[A]eq = 1, [B]eq = -1, [C]eq = 1, [D] = 3
Kc = {([D]eq)3 * ([C]eq)}/{[A]eq * ([B]eq)2}
= Kc = {(3)3*1}/{1*(-1)2}
= 27/1
= 27
A+2B ⇌ 2C+D
volume of flask = 1L
Initial moles of A = 2 mol
initial concentration of A=[A]i = 2 M
initial mole of B = 1 mol
[B]i = 1 M
[A]eq = 2-x, [B]eq = 1-2x, [C]eq = x, [D]eq = 3x
Given [D]eq = 1 * 1L
= 1 M
Thus x = 1M
[A]eq = 1, [B]eq = -1, [C]eq = 1, [D] = 3
Kc = {([D]eq)3 * ([C]eq)}/{[A]eq * ([B]eq)2}
= Kc = {(3)3*1}/{1*(-1)2}
= 27/1
= 27
Graphite is added to a vessel that contains CO2(g) at a pressure of 0.830 atm at a certain high temperature. The pressure rises due to a reaction that produces CO (g). The total pressure reaches an equilibrium value of 1.366 atm. Calculate the equilibrium constant of the following reaction.
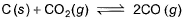
- a)2.909 atm
- b)6.909 atm
- c)4.909 atm
- d)3.909 atm
Correct answer is option 'D'. Can you explain this answer?
Graphite is added to a vessel that contains CO2(g) at a pressure of 0.830 atm at a certain high temperature. The pressure rises due to a reaction that produces CO (g). The total pressure reaches an equilibrium value of 1.366 atm. Calculate the equilibrium constant of the following reaction.
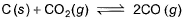
a)
2.909 atm
b)
6.909 atm
c)
4.909 atm
d)
3.909 atm
 | Top Rankers answered |

Direction (Q. Nos. 1-10) This section contains 10 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.Q. Vapour pressure of NH4HS (s)is 20 mm at 25°C, for
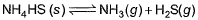
Total pressure when NH4HS (s) dissociates at 25°Cin a vessel which already contains H2S (g)at a pressure of 15 mm, is- a)25 mm
- b)50 mm
- c)5 mm
- d)10 mm
Correct answer is option 'A'. Can you explain this answer?
Direction (Q. Nos. 1-10) This section contains 10 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Q. Vapour pressure of NH4HS (s)is 20 mm at 25°C, for
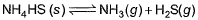
Total pressure when NH4HS (s) dissociates at 25°Cin a vessel which already contains H2S (g)at a pressure of 15 mm, is
Total pressure when NH4HS (s) dissociates at 25°Cin a vessel which already contains H2S (g)at a pressure of 15 mm, is
a)
25 mm
b)
50 mm
c)
5 mm
d)
10 mm
| | Hansa Sharma answered |
The correct answer is Option A.
NH4HS -------> NH3 + H2S
Let P be the pressure at eq. of NH3 and H2S.
Therefore, Kp = P2
= (20 / 2)2
= 100 mm
= 100
Also, Kp = (15 + P) (P)
100 = 15 P + P2
P2 + 15 P – 100 = 0
P = 5
Total pressure = 15 + 2(P)
= 15 + 2(5)
= 25 mm
NH4HS -------> NH3 + H2S
Let P be the pressure at eq. of NH3 and H2S.
Therefore, Kp = P2
= (20 / 2)2
= 100 mm
= 100
Also, Kp = (15 + P) (P)
100 = 15 P + P2
P2 + 15 P – 100 = 0
P = 5
Total pressure = 15 + 2(P)
= 15 + 2(5)
= 25 mm
Following equilibrium is set up at 1000 K and 1 bar in a 5 L flask,
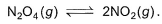
At equilibrium, NO2 is 50% o f the total volume. Thus, equilibrium constant Kc is - a)0.133
- b)0.266
- c)0.200
- d)0.400
Correct answer is option 'A'. Can you explain this answer?
Following equilibrium is set up at 1000 K and 1 bar in a 5 L flask,
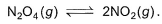
At equilibrium, NO2 is 50% o f the total volume. Thus, equilibrium constant Kc is
At equilibrium, NO2 is 50% o f the total volume. Thus, equilibrium constant Kc is
a)
0.133
b)
0.266
c)
0.200
d)
0.400
 | Manish Aggarwal answered |
The correct answer is Option A.
N2O4 ⇌ 2NO2
Initial 1 0
Equilibrium 1−x 2x
N2O4 ⇌ 2NO2
Initial 1 0
Equilibrium 1−x 2x
Total moles = 1 - x + 2x
NO2 is 50% of the total volume when equilibrium is set up.
Thus, the volume fraction (at equilibrium) of NO2 = 50/100 = 0.5 = ½
So, 2x / (1+x) = ½
=> x = ⅓
Thus, the volume fraction (at equilibrium) of NO2 = 50/100 = 0.5 = ½
So, 2x / (1+x) = ½
=> x = ⅓
For 1 litre;
Kc = [NO2] / [N2O4]
= [4*(1/9)] / [⅔]
= 0.66;
Kc = [NO2] / [N2O4]
= [4*(1/9)] / [⅔]
= 0.66;
For 5 litres;
Kc = 0.66 / 5
= 0.133
Thus, option A is correct.
Kc = 0.66 / 5
= 0.133
Thus, option A is correct.
Assume following equilibria when total pressure set up in each are equal to 1 atm, and equilibrium constant (Kp) as K1; K2 and K3
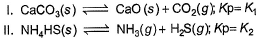
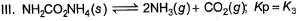
Thus,- a) K1 = K2 = K3
- b)K1 < K2 < K3
- c)K3 < K2 < K1
- d)None of these
Correct answer is option 'C'. Can you explain this answer?
Assume following equilibria when total pressure set up in each are equal to 1 atm, and equilibrium constant (Kp) as K1; K2 and K3
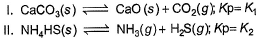
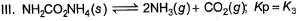
Thus,
Thus,
a)
K1 = K2 = K3
b)
K1 < K2 < K3
c)
K3 < K2 < K1
d)
None of these
 | Pioneer Academy answered |
The correct answer is option C
CaCO3 → CaO + CO2
Kp = k1 = Pco2
total pressure of container P
k1 = p
NH4HS → NH3 + H2S
PNH3 = PH2S = P0
P0 + P0 = p (total pressure)
P0 = p/2
k2 = kp = [PNH3][PH2s] p24
NH2CoNH2 → 2NH3 + CO2
PNH3 = 2P0 PCO2 = P0
2P0 + P0 = P
CaCO3 → CaO + CO2
Kp = k1 = Pco2
total pressure of container P
k1 = p
NH4HS → NH3 + H2S
PNH3 = PH2S = P0
P0 + P0 = p (total pressure)
P0 = p/2
k2 = kp = [PNH3][PH2s] p24
NH2CoNH2 → 2NH3 + CO2
PNH3 = 2P0 PCO2 = P0
2P0 + P0 = P
At 1000 K, pressure of CO2 in equilibrium with CaCO3 and CaO is equal to 2.105 atm. The equilibrium constant for the reaction,
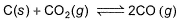
is 1.9 at the same temperature when pressure are in atm. Solid C, CaO, and CaCO3 are mixed and allowed to come to equilibrium at 1000 K in a closed vessel.Q. What is the pressure of CO (g)at equilibrium (in atm)?
Correct answer is '2'. Can you explain this answer?
At 1000 K, pressure of CO2 in equilibrium with CaCO3 and CaO is equal to 2.105 atm. The equilibrium constant for the reaction,
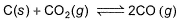
is 1.9 at the same temperature when pressure are in atm. Solid C, CaO, and CaCO3 are mixed and allowed to come to equilibrium at 1000 K in a closed vessel.
is 1.9 at the same temperature when pressure are in atm. Solid C, CaO, and CaCO3 are mixed and allowed to come to equilibrium at 1000 K in a closed vessel.
Q. What is the pressure of CO (g)at equilibrium (in atm)?
 | Learners Habitat answered |
K= (partial pressure of co2/(partial pressure of co2)
since k =1.9
So 1.9 = (partial pressure of co)2/2.105
(partial pressure of co)2 =2.105×1.9
= 3.99 = 4
(partial pressure of co) =2
since k =1.9
So 1.9 = (partial pressure of co)2/2.105
(partial pressure of co)2 =2.105×1.9
= 3.99 = 4
(partial pressure of co) =2
Ca(HCO3)2 decomposes as,Ca (HCO3)2(s) ⇌ CaCO3(s) + H2O(g) + CO2(g)
Equilibrium pressure is found to be 0.12 bar. What is pco2 if the reaction mixture also contains H2O(g)at 0.20 bar?- a) 0.20 bar
- b) 0.017 ba
- c)0.040 bar
- d)0.10 bar
Correct answer is option 'B'. Can you explain this answer?
Ca(HCO3)2 decomposes as,
Ca (HCO3)2(s) ⇌ CaCO3(s) + H2O(g) + CO2(g)
Equilibrium pressure is found to be 0.12 bar. What is pco2 if the reaction mixture also contains H2O(g)at 0.20 bar?
Equilibrium pressure is found to be 0.12 bar. What is pco2 if the reaction mixture also contains H2O(g)at 0.20 bar?
a)
0.20 bar
b)
0.017 ba
c)
0.040 bar
d)
0.10 bar
 | Top Rankers answered |

The equilibrium constant for the following reaction, is 1.6 x 105 at 1024 K.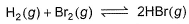
HBr (g)at 10.0 bar is introduced into a sealed container at 1024 K. Thus, partial pressure of H2(g)and Br2(g), together is- a)10 bar
- b)0.05 bar
- c)0.025 bar
- d)0.10 bar
Correct answer is option 'A'. Can you explain this answer?
The equilibrium constant for the following reaction, is 1.6 x 105 at 1024 K.
HBr (g)at 10.0 bar is introduced into a sealed container at 1024 K. Thus, partial pressure of H2(g)and Br2(g), together is
a)
10 bar
b)
0.05 bar
c)
0.025 bar
d)
0.10 bar
| | Rahul Bansal answered |

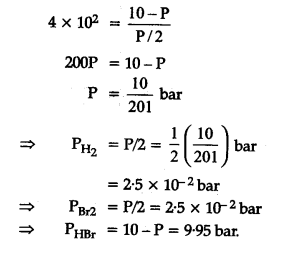
=> 10 bar approximately
For the reversible reaction, 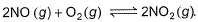
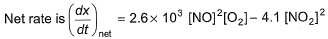
In a reaction vessel, [NO]= [O2]= 0.01 mol L-1 and [NO2]= 0.1 mol L-1 then above reaction is - a)shifted in forward direction
- b)shifted in backward direction
- c)in equilibrium
- d)not predictable in the absence of required data
Correct answer is option 'A'. Can you explain this answer?
For the reversible reaction, 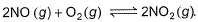
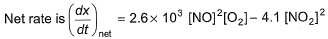
In a reaction vessel, [NO]= [O2]= 0.01 mol L-1 and [NO2]= 0.1 mol L-1 then above reaction is
In a reaction vessel, [NO]= [O2]= 0.01 mol L-1 and [NO2]= 0.1 mol L-1 then above reaction is
a)
shifted in forward direction
b)
shifted in backward direction
c)
in equilibrium
d)
not predictable in the absence of required data
| | Lavanya Menon answered |
On substituting the values of conc. of NO, O2 and NO2 in given rate equation, we get a +ve (positive) value indicating that the reaction takes place in forward direction.
A sample of N2O4(g)with a pressure of 1.00 atm is placed in a flask. When equilibrium is reached, 20% of N2O4(g)has been converted to NO2(g)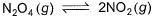 If the original pressure is made 10% of the earlier pressure, then per cent dissociation will be
If the original pressure is made 10% of the earlier pressure, then per cent dissociation will be- a)20%
- b)42%
- c)54%
- d)62%
Correct answer is option 'C'. Can you explain this answer?
A sample of N2O4(g)with a pressure of 1.00 atm is placed in a flask. When equilibrium is reached, 20% of N2O4(g)has been converted to NO2(g)
If the original pressure is made 10% of the earlier pressure, then per cent dissociation will be
a)
20%
b)
42%
c)
54%
d)
62%
 | Lohit Matani answered |
Correct answer is A.

Kc forthe decomposition of NH4HS(s) is 1.8x 10-4 at 25°C.
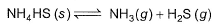
If the system already contains [NH3] = 0.020 M, then when equilibrium is reached, molar concentration are
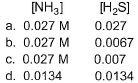
- a)a
- b)b
- c)c
- d)d
Correct answer is option 'B'. Can you explain this answer?
Kc forthe decomposition of NH4HS(s) is 1.8x 10-4 at 25°C.
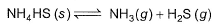
If the system already contains [NH3] = 0.020 M, then when equilibrium is reached, molar concentration are

If the system already contains [NH3] = 0.020 M, then when equilibrium is reached, molar concentration are
a)
a
b)
b
c)
c
d)
d
 | Sushil Kumar answered |
NH4HS (s) ⇋ NH3 (g) + H2S (g)
Initial 1 - -
At eqm 1-x x+0.02 x
Kc = [NH3][H2S] (Since NH4HS is solid, we ignore it.)
1.8×10-4 = (x+0.02)(x)
x2+0.02x-1.8×10-4 = 0
Applying quadratic formula; x = -0.02+√{(0.02)2-4×1.8×10-4}
= 0.033-0.020/2 = 0.0065
Therefore, concn of NH3 at equilibrium = x+0.020 = 0.0265
concn of H2S at equilibrium = x = 0.0065
So, option b is correct
Initial 1 - -
At eqm 1-x x+0.02 x
Kc = [NH3][H2S] (Since NH4HS is solid, we ignore it.)
1.8×10-4 = (x+0.02)(x)
x2+0.02x-1.8×10-4 = 0
Applying quadratic formula; x = -0.02+√{(0.02)2-4×1.8×10-4}
= 0.033-0.020/2 = 0.0065
Therefore, concn of NH3 at equilibrium = x+0.020 = 0.0265
concn of H2S at equilibrium = x = 0.0065
So, option b is correct
Equilibrium constant for the reaction PCL5⇋PCL3+CL2 is 0.0205 at 230°C and 1 atmospheric pressure if at equilibrium concentration of PCL5 is 0.235 moles liter−1liter-1and that of CL2= 0.028 moleslit−1lit-1 then conc. of PCL3 at equilibrium is- a)0.0174
- b)0.174
- c)0.0348
- d)1.74
Correct answer is option 'A'. Can you explain this answer?
Equilibrium constant for the reaction PCL5⇋PCL3+CL2 is 0.0205 at 230°C and 1 atmospheric pressure if at equilibrium concentration of PCL5 is 0.235 moles liter−1liter-1and that of CL2= 0.028 moleslit−1lit-1 then conc. of PCL3 at equilibrium is
a)
0.0174
b)
0.174
c)
0.0348
d)
1.74
| | Mira Sharma answered |

Passage ISolid ammonium chloride is in equilibrium with ammonia and hydrogen chloride gases
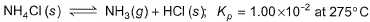
0.980 g of solid NH4CI is taken in a closed vessel of 1 L capacity and heated to 275° C.Q. Percentage decomposition of the original sample is- a)24.81%
- b)6.24%
- c)3.12%
- d)12.13%
Correct answer is option 'D'. Can you explain this answer?
Passage I
Solid ammonium chloride is in equilibrium with ammonia and hydrogen chloride gases
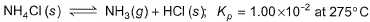
0.980 g of solid NH4CI is taken in a closed vessel of 1 L capacity and heated to 275° C.
0.980 g of solid NH4CI is taken in a closed vessel of 1 L capacity and heated to 275° C.
Q. Percentage decomposition of the original sample is
a)
24.81%
b)
6.24%
c)
3.12%
d)
12.13%
 | Knowledge Hub answered |
The state of HCl is given wrong. It will be in gaseous state.
So, the reaction be like;-
NH4Cl(s) ⇌ NH3(g) + HCl(g) kp = 1.00×10-2 at 275° C
Kp = kc(RT)2
1.00×10-2 = kc(0.0821×548)2
Or kc = 4.94×10-6
NH4Cl(s) ⇌ NH3(g) + HCl(g)
Initial 1 - -
At eqm 1-x x x
Kc = x2
x = √(4.94×10-6)
= 2.22×10-3
Therefore, NH4Cl dissociated at eqm = 2.22×10-3 × 53.5 = 0.118
%age decomposition = 0.118/0.980×100 = 12.13%
So, the reaction be like;-
NH4Cl(s) ⇌ NH3(g) + HCl(g) kp = 1.00×10-2 at 275° C
Kp = kc(RT)2
1.00×10-2 = kc(0.0821×548)2
Or kc = 4.94×10-6
NH4Cl(s) ⇌ NH3(g) + HCl(g)
Initial 1 - -
At eqm 1-x x x
Kc = x2
x = √(4.94×10-6)
= 2.22×10-3
Therefore, NH4Cl dissociated at eqm = 2.22×10-3 × 53.5 = 0.118
%age decomposition = 0.118/0.980×100 = 12.13%
Direction (Q. Nos. 21) This section contains 2 questions. when worked out will result in an integer from 0 to 9 (both inclusive)Q. For the equilibrium in gaseous phase in 2 L flask we start with 2 moles of SO2 and 1 mole of O2 at 3 atm, 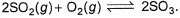
When equilibrium is attained, pressure changes to 2.5 atm. Hence, equilibrium constant Kc is
Correct answer is '4'. Can you explain this answer?
Direction (Q. Nos. 21) This section contains 2 questions. when worked out will result in an integer from 0 to 9 (both inclusive)
Q. For the equilibrium in gaseous phase in 2 L flask we start with 2 moles of SO2 and 1 mole of O2 at 3 atm, 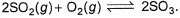
When equilibrium is attained, pressure changes to 2.5 atm. Hence, equilibrium constant Kc is
When equilibrium is attained, pressure changes to 2.5 atm. Hence, equilibrium constant Kc is
| | Om Desai answered |
The correct answer is 4
2SO2(g) + O2(g) ⇋ 2SO3
Initial moles 2 1
At equilibrium 2 - 2x 1 - x 2x
Net moles at equilibrium = 2 - 2x + 1 - x + 2x
=(3 - x)moles
Initial:
moles = 3,
Pressure = 3 atm,
Volume = 2L,
PV = nRT
3 x 2 = 3RT -------- 1
At equilibrium
Moles = 3 - x,
Pressure = 2.5 atm
Volume = 2L
P‘V = n’RT ---------- 2
Divide eqn 2 by 1
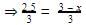
⇒2.5 = 3 - x
⇒x = 0.5
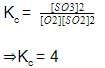
2SO2(g) + O2(g) ⇋ 2SO3
Initial moles 2 1
At equilibrium 2 - 2x 1 - x 2x
Net moles at equilibrium = 2 - 2x + 1 - x + 2x
=(3 - x)moles
Initial:
moles = 3,
Pressure = 3 atm,
Volume = 2L,
PV = nRT
3 x 2 = 3RT -------- 1
At equilibrium
Moles = 3 - x,
Pressure = 2.5 atm
Volume = 2L
P‘V = n’RT ---------- 2
Divide eqn 2 by 1
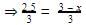
⇒2.5 = 3 - x
⇒x = 0.5
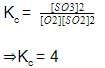
For the equilibrium,
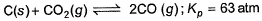
at 1000 K. If at equilibrium pCO = 10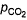 then total pressure at equilibrium is
then total pressure at equilibrium is - a)6.30 atm
- b)0.63 atm
- c)6.93 atm
- d)69.3 atm
Correct answer is option 'C'. Can you explain this answer?
For the equilibrium,
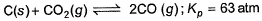
at 1000 K. If at equilibrium pCO = 10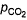 then total pressure at equilibrium is
then total pressure at equilibrium is
at 1000 K. If at equilibrium pCO = 10
a)
6.30 atm
b)
0.63 atm
c)
6.93 atm
d)
69.3 atm
| | Lavanya Menon answered |
C(s) + CO2(g) <=========> 2CO(g)
Kp = pCO2/pCO2
GIven Kp = 63 and pCO = 10pCO2
Putting the value of pCO in above equation,
63 = 100(pCO2)2/pCO2
Or pCO2 = 0.63
pCO = 6.3
Therefore, total pressure = 6.3+0.63 = 6.93 atm
Kp = pCO2/pCO2
GIven Kp = 63 and pCO = 10pCO2
Putting the value of pCO in above equation,
63 = 100(pCO2)2/pCO2
Or pCO2 = 0.63
pCO = 6.3
Therefore, total pressure = 6.3+0.63 = 6.93 atm
At 700 K and 350 bar, a 1 : 3 mixture of N2(g) and H2(g) reacts to form an equilibrium mixture containing X (NH3)= 0.50. Assuming ideal behaviour Kp for the equilibrium reaction, 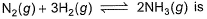
- a)2.03x 10-4
- b)3.55x 10-3
- c)1.02 x 10-4
- d)3.1 x 10-4
Correct answer is option 'D'. Can you explain this answer?
At 700 K and 350 bar, a 1 : 3 mixture of N2(g) and H2(g) reacts to form an equilibrium mixture containing X (NH3)= 0.50. Assuming ideal behaviour Kp for the equilibrium reaction, 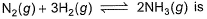
a)
2.03x 10-4
b)
3.55x 10-3
c)
1.02 x 10-4
d)
3.1 x 10-4
 | Knowledge Hub answered |
The correct answer is option A
2.03x 10-4
The given equation is :-
N2(g)+3H2(g) ⇌ 2NH3(g)
Initial moles : 1 3 0
At eqm ; (1−x) (3−3x) (2x)
(let)
Total moles of equation
=1 − x + 3 − 3x + 2x = (4−2x)
Now, X(NH3) =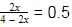
⇒ 2x = 2 − x
⇒ 3x = 2 ⇒ x = 0.66 =
32
Now, at equation, moles of N2= 1/3, moles of NH3 = 4/3
moles of H2 =3 − 2 = 1
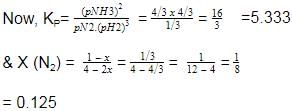
2.03x 10-4
The given equation is :-
N2(g)+3H2(g) ⇌ 2NH3(g)
Initial moles : 1 3 0
At eqm ; (1−x) (3−3x) (2x)
(let)
Total moles of equation
=1 − x + 3 − 3x + 2x = (4−2x)
Now, X(NH3) =
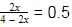
⇒ 2x = 2 − x
⇒ 3x = 2 ⇒ x = 0.66 =
32
Now, at equation, moles of N2= 1/3, moles of NH3 = 4/3
moles of H2 =3 − 2 = 1
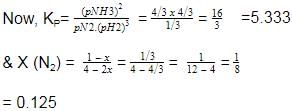
Passage lIOne of the reactions that takes place in producing steel from iron ore is the reduction of iron (II) oxide by carbon monoxide to give iron metal and carbon dioxide.
Initial partial pressure
CO(g) = 1.40 atm
CO2(g) = 0.80 atmQ. Under the given partial pressure, reaction is- a)displaced in forward side
- b)displaced in backward side
- c)in equilibrium
- d)incomplete in the absence of total pressure
Correct answer is option 'C'. Can you explain this answer?
Passage lI
One of the reactions that takes place in producing steel from iron ore is the reduction of iron (II) oxide by carbon monoxide to give iron metal and carbon dioxide.
Initial partial pressure
CO(g) = 1.40 atm
CO2(g) = 0.80 atm
Initial partial pressure
CO(g) = 1.40 atm
CO2(g) = 0.80 atm
Q. Under the given partial pressure, reaction is
a)
displaced in forward side
b)
displaced in backward side
c)
in equilibrium
d)
incomplete in the absence of total pressure
| | Preeti Iyer answered |
We know that with increase in pressure on one side, reaction shifts to that side which has less no. of moles(of gaseous species). However the no. of moles are same on both sides. So the REACTION WILL REMAIN IN EQUILIBRIUM.
Ammonium carbamate dissociates as,
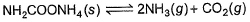
In a closed vessel containing ammonium carbamate in equilibrium with its vapour, ammonia is added such that partial pressure of NH3 now equals the original total pressure. Thus, ratio of the total pressure to the original pressure is- a)2:1
- b)31:27
- c)27 : 37
- d)
Correct answer is option 'B'. Can you explain this answer?
Ammonium carbamate dissociates as,
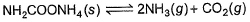
In a closed vessel containing ammonium carbamate in equilibrium with its vapour, ammonia is added such that partial pressure of NH3 now equals the original total pressure. Thus, ratio of the total pressure to the original pressure is
In a closed vessel containing ammonium carbamate in equilibrium with its vapour, ammonia is added such that partial pressure of NH3 now equals the original total pressure. Thus, ratio of the total pressure to the original pressure is
a)
2:1
b)
31:27
c)
27 : 37
d)
 | Imk Pathsala answered |
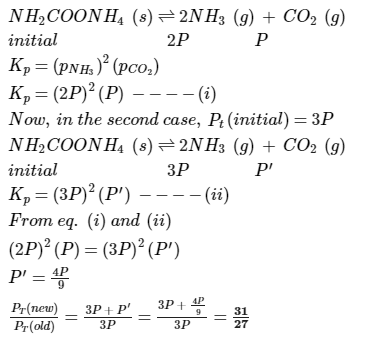
Direction (Q. Nos. 16-19) This section contains a paragraph, each describing theory, experiments, data etc. three Questions related to paragraph have been given.Each question have only one correct answer among the four given options (a),(b),(c),(d)Passage I
The equilibrium reaction 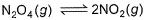 has been thoroughly studied Kp = 0.148 at 298 KIf the total pressure in a flask containing NO2 and N2O4 gas at 25°C is 1.50 atm, what fraction of the N2O4 has dissociated to NO2 ?
has been thoroughly studied Kp = 0.148 at 298 KIf the total pressure in a flask containing NO2 and N2O4 gas at 25°C is 1.50 atm, what fraction of the N2O4 has dissociated to NO2 ? - a)0.156
- b)0.844
- c)0.024
- d)0.076
Correct answer is option 'A'. Can you explain this answer?
Direction (Q. Nos. 16-19) This section contains a paragraph, each describing theory, experiments, data etc. three Questions related to paragraph have been given.Each question have only one correct answer among the four given options (a),(b),(c),(d)
Passage I
The equilibrium reaction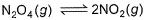 has been thoroughly studied Kp = 0.148 at 298 K
has been thoroughly studied Kp = 0.148 at 298 K
The equilibrium reaction
If the total pressure in a flask containing NO2 and N2O4 gas at 25°C is 1.50 atm, what fraction of the N2O4 has dissociated to NO2 ?
a)
0.156
b)
0.844
c)
0.024
d)
0.076
 | Lohit Matani answered |
The correct answer is Option A.
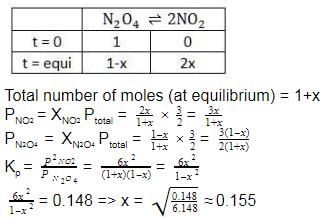
Fraction of N2O4 dissociated = x = 0.155 (x = mole fraction)
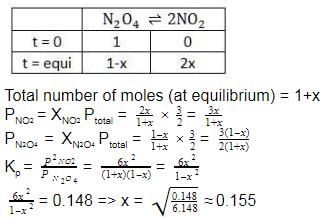
Fraction of N2O4 dissociated = x = 0.155 (x = mole fraction)
At 273 K and 1 atm, 1 L of N2O4 (g) decomposes to NO2(g)a s given,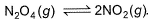 At equilibrium, original volume is 25% less than the existing volume. Percentage decomposition of N2O4 (g) is thus,
At equilibrium, original volume is 25% less than the existing volume. Percentage decomposition of N2O4 (g) is thus, - a)25%
- b)50%
- c)66.66%
- d)33.33%
Correct answer is option 'D'. Can you explain this answer?
At 273 K and 1 atm, 1 L of N2O4 (g) decomposes to NO2(g)a s given,
At equilibrium, original volume is 25% less than the existing volume. Percentage decomposition of N2O4 (g) is thus,
a)
25%
b)
50%
c)
66.66%
d)
33.33%
| | Suresh Reddy answered |
Let the initial volume of N2O4 be x and initial volume of NO2 is 0
If the degree of dissociation is a, then the final volume of N2O4 is x(1−a) and NO2 is 2ax.
Initial
It equilibrium
N2O4 ⟶ 2NO2
x 0
x(1−a) 2ax
Total initial volume =x+0=x
Final volume =x(1−a)+2ax=x+ax=x(1+a)
It is given that the initial volume is 25% less than the final volume
x=0.75×(1+a)
1+a=1.33
a=0.33
So %age dissociation = 33.33%
If the degree of dissociation is a, then the final volume of N2O4 is x(1−a) and NO2 is 2ax.
Initial
It equilibrium
N2O4 ⟶ 2NO2
x 0
x(1−a) 2ax
Total initial volume =x+0=x
Final volume =x(1−a)+2ax=x+ax=x(1+a)
It is given that the initial volume is 25% less than the final volume
x=0.75×(1+a)
1+a=1.33
a=0.33
So %age dissociation = 33.33%
Passage IIA 15 L flask at 300 K contains 64.4 g of a mixture of NO2 and N2O4 in equilibrium. Given,
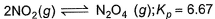 Q. Kc for the above equilibrium is
Q. Kc for the above equilibrium is - a)164.28
- b) 6.087x 10-3
- c)0.2708
- d)3.693
Correct answer is option 'A'. Can you explain this answer?
Passage II
A 15 L flask at 300 K contains 64.4 g of a mixture of NO2 and N2O4 in equilibrium. Given,
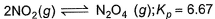
Q. Kc for the above equilibrium is
a)
164.28
b)
6.087x 10-3
c)
0.2708
d)
3.693
| | Pooja Shah answered |
Kp = Kc(RT)∆n
Kp = 6.67 ,
∆n = moles of products - moles of reactants = 1-2 = -1
R = 0.0821 L atm mol-¹K-¹
T = 300K
∆n = moles of products - moles of reactants = 1-2 = -1
R = 0.0821 L atm mol-¹K-¹
T = 300K
Substitute these values in the formula,
=> Kc = 6.67×0.0821×300
Kc = 164.28.
=> Kc = 6.67×0.0821×300
Kc = 164.28.
H2S (g) initially at a pressure of 10.0 atm and a temperature of 800 K, dissociates as
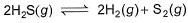 At equilibrium, the partial pressure of S2 vapour is 0.020 atm . Thus, Kp is
At equilibrium, the partial pressure of S2 vapour is 0.020 atm . Thus, Kp is - a)3.23x 10-7
- b)6.45x 10-7
- c)1.55x 106
- d)6.20x 107
Correct answer is option 'A'. Can you explain this answer?
H2S (g) initially at a pressure of 10.0 atm and a temperature of 800 K, dissociates as
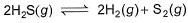
At equilibrium, the partial pressure of S2 vapour is 0.020 atm . Thus, Kp is
a)
3.23x 10-7
b)
6.45x 10-7
c)
1.55x 106
d)
6.20x 107
| | Geetika Shah answered |
The correct answer is Option A.
2H2S(g) ⇌ 2H2(g) + S2(g)
Pressure
at t=0 Pi − −
at eqm Pi−P 2P P
2H2S(g) ⇌ 2H2(g) + S2(g)
Pressure
at t=0 Pi − −
at eqm Pi−P 2P P
as P=0.02 thus Pi−P=10−0.02
Pi=10 2P=0.04

Kp = 3.23×10−7 atm.
Pi=10 2P=0.04

Kp = 3.23×10−7 atm.
Once the equilibrium is reached under given condition:- a)Cone, remains the same in spite of the change in temperature
- b)Cone, of all the substances presents do not change
- c)Cone, of reactants remairfs same
- d)Cone, of products remains same
Correct answer is option 'B'. Can you explain this answer?
Once the equilibrium is reached under given condition:
a)
Cone, remains the same in spite of the change in temperature
b)
Cone, of all the substances presents do not change
c)
Cone, of reactants remairfs same
d)
Cone, of products remains same
| | Raghav Bansal answered |
In a chemical reaction, chemical equilibrium is the state in which the forward reaction rate and the reverse reaction rate are equal. The result of this equilibrium is that the concentrations of the reactants and the products do not change. However, just because concentrations aren’t changing does not mean that all chemical reaction has ceased. Just the opposite is true; chemical equilibrium is a dynamic state in which reactants are being converted into products at all times, but at the exact rate that products are being converted back into reactants. The result of such a situation is analogous to a bridge between two cities, where the rate of cars going over the bridge in each direction is exactly equal. The result is that the net number of cars on either side of the bridge does not change.
At 90° C , the following equilibrium is established :

If 0.20 mole of hydrogen and 1.0 mole of sulphur are heated to 90°C in a 1.0 dm3 flask, what will be the partial pressure of H2S gas at equilibrium?- a)0.36 atm
- b)0.38 atm
- c)0.28 atm
- d)0.26 atm
Correct answer is option 'B'. Can you explain this answer?
At 90° C , the following equilibrium is established :

If 0.20 mole of hydrogen and 1.0 mole of sulphur are heated to 90°C in a 1.0 dm3 flask, what will be the partial pressure of H2S gas at equilibrium?
If 0.20 mole of hydrogen and 1.0 mole of sulphur are heated to 90°C in a 1.0 dm3 flask, what will be the partial pressure of H2S gas at equilibrium?
a)
0.36 atm
b)
0.38 atm
c)
0.28 atm
d)
0.26 atm
| | Geetika Shah answered |
Initial moles of H₂ = 0.2
Initial moles of S = 1
Kp = 6.8 * 10⁻²
Given equation:
H2(g) + S(s) ⇋ H2S(g)
Initial moles: 0.2 1
At equilibrium: (0.2-α) (1-α) α
Here, in the above equation we can see that hydrogen is the limiting reagent.
∴ Kp = α/(0.2 – α)
⇒ 6.8 * 10⁻² = α/(0.2 – α)
⇒ 1.36*10⁻² – (6.8*10⁻²)α = α
⇒ α + 0.068α = 1.36*10⁻²
⇒ α = 1.36*10⁻² / 1.068 = 1.273 * 10⁻² ← moles of H₂S
So, at equilibrium moles of H₂ = 0.2 – α = 0.2 – 1.273 * 10⁻² = 0.1873
Now, using the Ideal Gas equation,
PV = nRT ….. (i)
Where P = total pressure of the vessel
n = total no. of moles = (0.2-α) + (1-α) + α = 1.2 – α = 1.2 – 1.273*10⁻² = 1.1873
V = volume of vessel = 1 litre
R = Ideal gas constant = 0.082 L atm K⁻¹mol⁻¹
T = total temperature = 90℃ = 90+273 = 363 K
Substituting all the values in eq. (i), we get
P * 1 = 1.1873 * 0.082 * 363
⇒ P = 35.34 atm
Thus,
The partial pressure of H₂S at equilibrium
= (mole fraction of H₂S) * (total pressure)
= [1.273*10⁻² / 1.1873] * 35.34
= 0.3789 atm
≈ 0.38 atm
Initial moles of S = 1
Kp = 6.8 * 10⁻²
Given equation:
H2(g) + S(s) ⇋ H2S(g)
Initial moles: 0.2 1
At equilibrium: (0.2-α) (1-α) α
Here, in the above equation we can see that hydrogen is the limiting reagent.
∴ Kp = α/(0.2 – α)
⇒ 6.8 * 10⁻² = α/(0.2 – α)
⇒ 1.36*10⁻² – (6.8*10⁻²)α = α
⇒ α + 0.068α = 1.36*10⁻²
⇒ α = 1.36*10⁻² / 1.068 = 1.273 * 10⁻² ← moles of H₂S
So, at equilibrium moles of H₂ = 0.2 – α = 0.2 – 1.273 * 10⁻² = 0.1873
Now, using the Ideal Gas equation,
PV = nRT ….. (i)
Where P = total pressure of the vessel
n = total no. of moles = (0.2-α) + (1-α) + α = 1.2 – α = 1.2 – 1.273*10⁻² = 1.1873
V = volume of vessel = 1 litre
R = Ideal gas constant = 0.082 L atm K⁻¹mol⁻¹
T = total temperature = 90℃ = 90+273 = 363 K
Substituting all the values in eq. (i), we get
P * 1 = 1.1873 * 0.082 * 363
⇒ P = 35.34 atm
Thus,
The partial pressure of H₂S at equilibrium
= (mole fraction of H₂S) * (total pressure)
= [1.273*10⁻² / 1.1873] * 35.34
= 0.3789 atm
≈ 0.38 atm
Statement I 1 mole A(g) and 1 mole B(g)give 0.5 mole of C(g)and 0.5 mole D(g) at equilibrium.
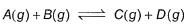
On taking 2 moles each of A(g)and B(g), percentage dissociation A(g)and B(g) is also doubled.Statement IIEquilibrium constant, Kc = 1- a)Both Statement I and Statement II are correct and Statement II is the corect explanation of Statement I
- b)Both Statement I and Statement II are correct and Statement II is not the correct explanation of Statement I
- c)Statement I is correct but Statement II is incorrect
- d)Statement II is incorrect but Statement II is correct
Correct answer is option 'D'. Can you explain this answer?
Statement I
1 mole A(g) and 1 mole B(g)give 0.5 mole of C(g)and 0.5 mole D(g) at equilibrium.
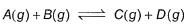
On taking 2 moles each of A(g)and B(g), percentage dissociation A(g)and B(g) is also doubled.
On taking 2 moles each of A(g)and B(g), percentage dissociation A(g)and B(g) is also doubled.
Statement II
Equilibrium constant, Kc = 1
a)
Both Statement I and Statement II are correct and Statement II is the corect explanation of Statement I
b)
Both Statement I and Statement II are correct and Statement II is not the correct explanation of Statement I
c)
Statement I is correct but Statement II is incorrect
d)
Statement II is incorrect but Statement II is correct
| | Soma Shankar answered |
No answer . in option"D" it has given that both statements AS II
In a state of dynamic equilibrium for the reaction A ⇌ B, which of the following observations would be made?
A. The concentration of A remains constant
B. The rate of conversion of A to B equals the rate of conversion of B to A
C. No molecules of A are converting to B
D. The system must be at constant temperature and pressure
E. The concentration of A must equal the concentration of B
Choose the correct answer from the options given below:- a)A, B and D only
- b)A, C and E only
- c)B, C and D only
- d)A, B and E only
Correct answer is option 'A'. Can you explain this answer?
A. The concentration of A remains constant
B. The rate of conversion of A to B equals the rate of conversion of B to A
C. No molecules of A are converting to B
D. The system must be at constant temperature and pressure
E. The concentration of A must equal the concentration of B
Choose the correct answer from the options given below:
a)
A, B and D only
b)
A, C and E only
c)
B, C and D only
d)
A, B and E only
 | Dr. Mohit Rajpoot answered |
The correct answer is Option A - A, B and D only
In a dynamic chemical equilibrium the rate of the forward reaction equals the rate of the reverse reaction, so there is no net change in amounts of reactants and products over time.
Because the forward and reverse rates are equal, the concentration of each species remains constant with time even though individual molecules continue to react in both directions.
The idea that no molecules are converting in either direction is incorrect because microscopic reactions continue; the equilibrium is dynamic, not static.
At equilibrium the macroscopic variables such as temperature and, in a closed steady system, pressure remain constant; additionally, the equilibrium constant is defined for a given temperature (so maintaining temperature is essential for a fixed equilibrium position).
There is no general requirement that concentrations of reactant and product be equal; they are only constant, not necessarily equal, unless dictated by stoichiometry or the value of the equilibrium constant.
Direction (Q. Nos. 14 and 15) This sectionis based on statement I and Statement II. Select the correct answer from the code given below.Q. Statement I If Kc = 4 for the following equilibrium,
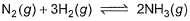
Then mixture of 1 mole N2, 3 moles H2 and 2 moles NH3 in 1L flask is in equilibrium.Statement IIReaction quotient of the given quantities is less than the equilibrium constant.- a)Both Statement I and Statement II are correct and Statement II is the corect explanation of Statement I
- b)Both Statement I and Statement II are correct and Statement II is not the correct explanation of Statement I
- c)Statement I is correct but Statement II is incorrect
- d)Statement II is incorrect but Statement II is correct
Correct answer is option 'D'. Can you explain this answer?
Direction (Q. Nos. 14 and 15) This sectionis based on statement I and Statement II. Select the correct answer from the code given below.
Q. Statement I
If Kc = 4 for the following equilibrium,
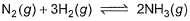
Then mixture of 1 mole N2, 3 moles H2 and 2 moles NH3 in 1L flask is in equilibrium.
Then mixture of 1 mole N2, 3 moles H2 and 2 moles NH3 in 1L flask is in equilibrium.
Statement II
Reaction quotient of the given quantities is less than the equilibrium constant.
a)
Both Statement I and Statement II are correct and Statement II is the corect explanation of Statement I
b)
Both Statement I and Statement II are correct and Statement II is not the correct explanation of Statement I
c)
Statement I is correct but Statement II is incorrect
d)
Statement II is incorrect but Statement II is correct
 | Ank answered |
B right ans
Solid ammonium carbonate (NH2CO2NH4) dissociates completely into ammonia and carbon dioxide when it evaporates :
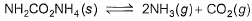
At 298 K, the total pressure of the gases in equilibrium with the solid is 0.116 atm. Derive the value of equilibrium constant Kp.- a)4p3/27
- b)6p3/27
- c)4p2/27
- d)2p3/27
Correct answer is option 'A'. Can you explain this answer?
Solid ammonium carbonate (NH2CO2NH4) dissociates completely into ammonia and carbon dioxide when it evaporates :
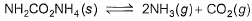
At 298 K, the total pressure of the gases in equilibrium with the solid is 0.116 atm. Derive the value of equilibrium constant Kp.
At 298 K, the total pressure of the gases in equilibrium with the solid is 0.116 atm. Derive the value of equilibrium constant Kp.
a)
4p3/27
b)
6p3/27
c)
4p2/27
d)
2p3/27
 | Sourav Dey answered |
A
Direction (Q. Nos. 11-14) This section contains a paragraph, wach describing theory, experiments, data etc. three Questions related to paragraph have been given.Each question have only one correct answer among the four given options (a),(b),(c),(d)Passage ISolid ammonium chloride is in equilibrium with ammonia and hydrogen chloride gases
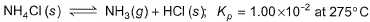
0.980 g of solid NH4CI is taken in a closed vessel of 1 L capacity and heated to 275° C.Q. Partial pressure of NH3(g) or HCI (g) at equilibrium is- a)0.10 atm
- b)1.0 atm
- c)0.01 atm
- d)1 x 10-4 atm
Correct answer is option 'A'. Can you explain this answer?
Direction (Q. Nos. 11-14) This section contains a paragraph, wach describing theory, experiments, data etc. three Questions related to paragraph have been given.Each question have only one correct answer among the four given options (a),(b),(c),(d)
Passage I
Solid ammonium chloride is in equilibrium with ammonia and hydrogen chloride gases
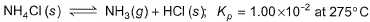
0.980 g of solid NH4CI is taken in a closed vessel of 1 L capacity and heated to 275° C.
0.980 g of solid NH4CI is taken in a closed vessel of 1 L capacity and heated to 275° C.
Q. Partial pressure of NH3(g) or HCI (g) at equilibrium is
a)
0.10 atm
b)
1.0 atm
c)
0.01 atm
d)
1 x 10-4 atm
| | Neha Joshi answered |
The reaction is as follow:-
NH4Cl(s) ⇋ NH3(g) + HCl(g)
Kp = (pNH3)(pHCl)
1×10-2 = p2 (SInce reactant dissociates into same ratio, so the partial pressure will be same for both)
100×10-4 = p2
Or p = 10×10-2 = 0.10
So, the partial pressure of NH3 and HCl are 0.10 atm.
NH4Cl(s) ⇋ NH3(g) + HCl(g)
Kp = (pNH3)(pHCl)
1×10-2 = p2 (SInce reactant dissociates into same ratio, so the partial pressure will be same for both)
100×10-4 = p2
Or p = 10×10-2 = 0.10
So, the partial pressure of NH3 and HCl are 0.10 atm.
For the following equilibrium,
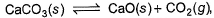


- a)a
- b)b
- c)c
- d)d
Correct answer is option 'A'. Can you explain this answer?
For the following equilibrium,
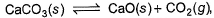

a)
a
b)
b
c)
c
d)
d
 | Raghav Shukla answered |
It's a very easy
you can solve it very easily with help of le Chatlier principle
clearly pressure is of co2 only as all other components are solid and there concentrations are taken as 1
clearly putting values with help of ideal gas equation, you will get the answer
you can solve it very easily with help of le Chatlier principle
clearly pressure is of co2 only as all other components are solid and there concentrations are taken as 1
clearly putting values with help of ideal gas equation, you will get the answer
On taking 60.0 g CH3COOH and 46.0 g CH3CH2OH in a 5 L flask in the presence of H30+ (catalyst), at 298 K 44.0 g of CH3COOC2H5 is formed at equilibrium.If amount of CH3COOH is doubled without affecting amount of CH3CH2OH, then CH3COOC2H5 formed is - a)20.33 g
- b)22.0 g
- c)44.0 g
- d)58.66 g
Correct answer is option 'D'. Can you explain this answer?
On taking 60.0 g CH3COOH and 46.0 g CH3CH2OH in a 5 L flask in the presence of H30+ (catalyst), at 298 K 44.0 g of CH3COOC2H5 is formed at equilibrium.
If amount of CH3COOH is doubled without affecting amount of CH3CH2OH, then CH3COOC2H5 formed is
a)
20.33 g
b)
22.0 g
c)
44.0 g
d)
58.66 g
 | Arjun Saini answered |
After doubling the amount,
Moles of CH3COOH => 120/60 = 2 moles.
Moles of CH3CH2OH => 46/46 = 1 moles.
POAC on Carbon,
2 * 2 + 1 * 2 = 2*n
we get,
n = 6/2 = 3 moles.
Mass of CH3COOC2H5 => 3 * 88 = > 264 gm
Passage IIA 15 L flask at 300 K contains 64.4 g of a mixture of NO2 and N2O4 in equilibrium. Given,
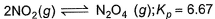 Q. Total pressure in the flask is
Q. Total pressure in the flask is - a)0.7427 atm
- b)1.3400 atm
- c)0.3714 atm
- d)0.6732 atm
Correct answer is option 'B'. Can you explain this answer?
Passage II
A 15 L flask at 300 K contains 64.4 g of a mixture of NO2 and N2O4 in equilibrium. Given,
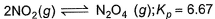
Q. Total pressure in the flask is
a)
0.7427 atm
b)
1.3400 atm
c)
0.3714 atm
d)
0.6732 atm
 | Top Rankers answered |
Step 1: Write the equilibrium reaction
The reaction between N2O4 and NO2 is:
N2O4(g) ⇌ 2NO2(g)
Step 2: Use the given data
We are given:
- Volume of the flask = 15 L
- Temperature = 300 K
- Mass of the mixture = 64.4 g (which is the mass of a mixture of N2O4 and NO2)
We need to find the total pressure in the flask.
Step 3: Calculate moles of N2O4 and NO2
Let's assume the mixture contains xxx grams of N2O4 and (64.4 − x) grams of NO2.
- Molar mass of N2O4 = 92 g/mol
- Molar mass of NO2 = 46 g/mol
The moles of N2O4 and NO2 can be calculated as:
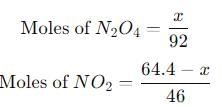
Step 4: Use the ideal gas law to calculate pressure
We can use the ideal gas law equation:
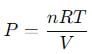
Where:
- P is the pressure,
- n is the total number of moles,
- R is the gas constant (0.0821 L·atm/mol·K),
- T is the temperature (300 K),
- V is the volume (15 L).
The total number of moles is the sum of the moles of N2O4 and NO2:
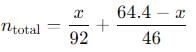
Now, we can use the ideal gas law equation:
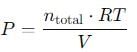
Substitute the values into the equation and solve for P.
Step 5: Calculate the total pressure
After performing the calculations, we find that the total pressure in the flask is approximately 1.3400 atm.
Direction (Q. Nos. 15 and 16) This section contains 2 questions. when worked out will result in an integer from 0 to 9 (both inclusive)Hot copper tunnings can be used as an “oxygen getter" for inert gas supplies by slowly passing the gas over the copper tunnings at 650 K.
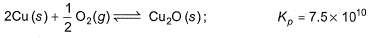 Q. How many molecules of CO2 are left in 1 L of a gas supply after equilibrium has been reached?
Q. How many molecules of CO2 are left in 1 L of a gas supply after equilibrium has been reached?
Correct answer is '2'. Can you explain this answer?
Direction (Q. Nos. 15 and 16) This section contains 2 questions. when worked out will result in an integer from 0 to 9 (both inclusive)
Hot copper tunnings can be used as an “oxygen getter" for inert gas supplies by slowly passing the gas over the copper tunnings at 650 K.
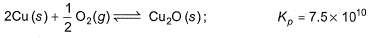
Q. How many molecules of CO2 are left in 1 L of a gas supply after equilibrium has been reached?
| | Aditya Oza answered |
Is it CO2 or Cu2O.
For the following equilibrium starting with 2 moles SO2 and 1 mole O2 in 1 L flask,
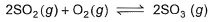
Equilibrium mixture required 0.4 mole 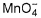 in acidic medium. Hence, Kc is
in acidic medium. Hence, Kc is - a)2.0
- b)0.4
- c)1.6
- d)2.6
Correct answer is option 'A'. Can you explain this answer?
For the following equilibrium starting with 2 moles SO2 and 1 mole O2 in 1 L flask,
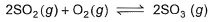
Equilibrium mixture required 0.4 mole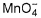 in acidic medium. Hence, Kc is
in acidic medium. Hence, Kc is
Equilibrium mixture required 0.4 mole
a)
2.0
b)
0.4
c)
1.6
d)
2.6
| | Ananya Datta answered |
0.4m of KMnO4 = 1 mole of SO2
= 1 mole of SO3
2SO2 + O2 ⇌ 2SO3
2 1 0
1 0.5 1
K = [SO3]2/[SO2]2 [O2]
= 12/(12*0.5)
= 2
= 1 mole of SO3
2SO2 + O2 ⇌ 2SO3
2 1 0
1 0.5 1
K = [SO3]2/[SO2]2 [O2]
= 12/(12*0.5)
= 2
Passage lIOne of the reactions that takes place in producing steel from iron ore is the reduction of iron (II) oxide by carbon monoxide to give iron metal and carbon dioxide.
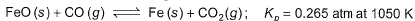
Initial partial pressure
CO(g) = 1.40 atm
CO2(g) = 0.80 atmQ. When equilibrium is attained,- a)
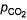 = Pco = 1-74 atm
= Pco = 1-74 atm - b)
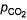 = Pco = 0.46 atm
= Pco = 0.46 atm - c)
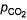 = 0-46 atm, pco = 1.74 atm
= 0-46 atm, pco = 1.74 atm - d)
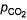 = 1.74 atm, pco = 0.46 atm
= 1.74 atm, pco = 0.46 atm
Correct answer is option 'C'. Can you explain this answer?
Passage lI
One of the reactions that takes place in producing steel from iron ore is the reduction of iron (II) oxide by carbon monoxide to give iron metal and carbon dioxide.
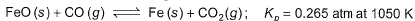
Initial partial pressure
CO(g) = 1.40 atm
CO2(g) = 0.80 atm
Initial partial pressure
CO(g) = 1.40 atm
CO2(g) = 0.80 atm
Q. When equilibrium is attained,
a)
b)
c)
d)
 | Ashwini Majumdar answered |
For the given reaction,

the reaction will proceed in the backward direction.
Therefore, we can say that the pressure of CO will increase while the pressure of CO
2
will decrease.Now, let the increase in pressure of CO = decrease in pressure of CO
2
be p.Then, we can write,
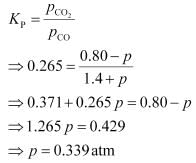
Therefore, equilibrium partial of
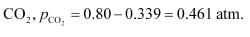
And, equilibrium partial pressure of
Direction (Q. Nos. 1-13) This section contains 13 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.Q. Given the reactions, 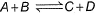 If one mole each of A and B are take in 5 L flask at 300 K, 0.7 mole of C are formed. Molar concentration of each species at equilibrium, when one mole of each are taken initially is
If one mole each of A and B are take in 5 L flask at 300 K, 0.7 mole of C are formed. Molar concentration of each species at equilibrium, when one mole of each are taken initially is
- a)a
- b)b
- c)c
- d)d
- e)None of the above
Correct answer is option 'E'. Can you explain this answer?
Direction (Q. Nos. 1-13) This section contains 13 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Q. Given the reactions, 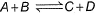 If one mole each of A and B are take in 5 L flask at 300 K, 0.7 mole of C are formed. Molar concentration of each species at equilibrium, when one mole of each are taken initially is
If one mole each of A and B are take in 5 L flask at 300 K, 0.7 mole of C are formed. Molar concentration of each species at equilibrium, when one mole of each are taken initially is
a)
a
b)
b
c)
c
d)
d
e)
None of the above
 | Lead Academy answered |

None of the provided options exactly match these concentrations.
Passage I
The equilibrium reaction 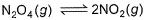 has been thoroughly studied Kp = 0.148 at 298 KQ. If the volume of the container is increased so that the total equilibrium pressure falls to 1.00 atm, then fraction of N2O4 dissociated is
has been thoroughly studied Kp = 0.148 at 298 KQ. If the volume of the container is increased so that the total equilibrium pressure falls to 1.00 atm, then fraction of N2O4 dissociated is- a) 0.036
- b)0.064
- c)0.911
- d)0.189
Correct answer is option 'D'. Can you explain this answer?
Passage I
The equilibrium reaction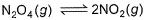 has been thoroughly studied Kp = 0.148 at 298 K
has been thoroughly studied Kp = 0.148 at 298 K
The equilibrium reaction
Q. If the volume of the container is increased so that the total equilibrium pressure falls to 1.00 atm, then fraction of N2O4 dissociated is
a)
0.036
b)
0.064
c)
0.911
d)
0.189
 | Ciel Knowledge answered |
Step 1: Understanding the equilibrium reaction
The equilibrium reaction is:
At equilibrium, we know:
- The equilibrium constant Kp = 0.148 at 298 K.
- The total equilibrium pressure is reduced to 1.00 atm due to an increase in the volume of the container.
Step 2: Use the equilibrium expression for Kp
The equilibrium expression for this reaction is:
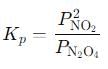
Where PNO2 is the partial pressure of NO2 and PN2O4 is the partial pressure of N2O4 at equilibrium.
Step 3: Let’s assume an initial amount of N2O4
Let’s assume the initial pressure of N2O4 before dissociation is P0, and the partial pressure of NO2 at equilibrium is PNO2. Let’s denote the fraction dissociated as x.
At equilibrium, the changes in pressures would be:
- The partial pressure of N2O4 will decrease by xP0.
- The partial pressure of NO2 will increase by 2xP0 (since two moles of NO2 are produced for each mole of N2O4).
Thus, the equilibrium pressures can be written as:
- PN2O4 = P0 − xP0
- PNO2 = 2xP0
Step 4: Total pressure at equilibrium
The total pressure at equilibrium is the sum of the partial pressures:
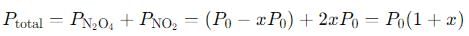
We are told that the total pressure is 1.00 atm, so:
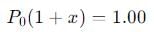
This equation allows us to solve for P0 in terms of x.
Step 5: Apply the equilibrium constant expression
Substitute the equilibrium pressures into the Kp expression:
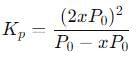
We know that Kp = 0.148, so:
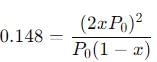
Simplify the equation:
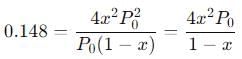
Now, substitute 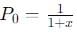 into this equation and solve for x.
into this equation and solve for x.
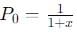 into this equation and solve for x.
into this equation and solve for x.Step 6: Solve for the fraction dissociated x
After solving the equation, you will find that the fraction dissociated xxx is approximately 0.189.
Chapter doubts & questions for Equilibrium - Chemistry - 4 Months Preparation for NEET 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Equilibrium - Chemistry - 4 Months Preparation for NEET in English & Hindi are available as part of NEET exam. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
;
Signup to see your scores go up
within 7 days!
within 7 days!
Takes less than 10 seconds to signup