All Exams > UPSC > 1 Year Preparation for UPSC CSE > All Questions
All questions of Chemistry for UPSC CSE Exam
H2O is liquid and H2S is a gas because- a)oxygen forms stronger hydrogen level than sulphur.
- b)oxygen is less electronegative than sulphur.
- c)atomic radius of oxygen is less than that of sulphur.
- d)atomic radius of oxygen is greater than that of sulphur.
Correct answer is option 'A'. Can you explain this answer?
H2O is liquid and H2S is a gas because
a)
oxygen forms stronger hydrogen level than sulphur.
b)
oxygen is less electronegative than sulphur.
c)
atomic radius of oxygen is less than that of sulphur.
d)
atomic radius of oxygen is greater than that of sulphur.
| | Rahul Rathore answered |
In h2o there is hydrogen bonding because oxygen has a high electronegativity(only second to flourine). so, the hydrogen atoms from other molecules of water forms a hydrogen bonds with oxygen resulting in intermolecular hydrogen bonding. ... therefore at room temperature h2o is aliquid and h2s is a gas.
Hydrogen bomb is based on the principle of- a)controlled fusion reaction
- b)uncontrolled fusion reaction
- c)controlled fission reaction
- d)uncontrolled fission reaction
Correct answer is option 'B'. Can you explain this answer?
Hydrogen bomb is based on the principle of
a)
controlled fusion reaction
b)
uncontrolled fusion reaction
c)
controlled fission reaction
d)
uncontrolled fission reaction
| | Puja Roy answered |
Understanding the Hydrogen Bomb
The hydrogen bomb, also known as the thermonuclear bomb, operates on the principle of nuclear fusion. Unlike atomic bombs, which rely on fission, hydrogen bombs utilize uncontrolled fusion reactions.
Key Principles of the Hydrogen Bomb
- Uncontrolled Fusion Reaction: The hydrogen bomb is designed to create conditions that allow hydrogen isotopes (deuterium and tritium) to fuse together, releasing tremendous amounts of energy. This fusion occurs at extremely high temperatures and pressures, similar to those in the core of the sun.
- Initiation of Fusion: The initial explosion of a fission bomb (a smaller atomic bomb) serves as the trigger for the hydrogen bomb. The heat and pressure from this fission explosion compress the fusion fuel, leading to the fusion reaction.
- Release of Energy: The energy released during the fusion process is far greater than that released from fission reactions. This results in a massive explosion, making hydrogen bombs significantly more powerful than their fission counterparts.
Comparison with Other Reactions
- Controlled vs. Uncontrolled: While controlled fusion reactions are harnessed in experimental reactors (like tokamaks) for energy production, the hydrogen bomb operates on an uncontrolled basis, leading to explosive outcomes.
- Fission vs. Fusion: Fission reactions split heavy atomic nuclei to release energy, while fusion combines light nuclei. The hydrogen bomb's reliance on fusion is what distinguishes it from traditional nuclear weapons.
In summary, the hydrogen bomb's operation is primarily based on an uncontrolled fusion reaction, resulting in a powerful and destructive explosion.
The hydrogen bomb, also known as the thermonuclear bomb, operates on the principle of nuclear fusion. Unlike atomic bombs, which rely on fission, hydrogen bombs utilize uncontrolled fusion reactions.
Key Principles of the Hydrogen Bomb
- Uncontrolled Fusion Reaction: The hydrogen bomb is designed to create conditions that allow hydrogen isotopes (deuterium and tritium) to fuse together, releasing tremendous amounts of energy. This fusion occurs at extremely high temperatures and pressures, similar to those in the core of the sun.
- Initiation of Fusion: The initial explosion of a fission bomb (a smaller atomic bomb) serves as the trigger for the hydrogen bomb. The heat and pressure from this fission explosion compress the fusion fuel, leading to the fusion reaction.
- Release of Energy: The energy released during the fusion process is far greater than that released from fission reactions. This results in a massive explosion, making hydrogen bombs significantly more powerful than their fission counterparts.
Comparison with Other Reactions
- Controlled vs. Uncontrolled: While controlled fusion reactions are harnessed in experimental reactors (like tokamaks) for energy production, the hydrogen bomb operates on an uncontrolled basis, leading to explosive outcomes.
- Fission vs. Fusion: Fission reactions split heavy atomic nuclei to release energy, while fusion combines light nuclei. The hydrogen bomb's reliance on fusion is what distinguishes it from traditional nuclear weapons.
In summary, the hydrogen bomb's operation is primarily based on an uncontrolled fusion reaction, resulting in a powerful and destructive explosion.
Which one of the following pairs is correctly matched?- a)Silver iodide — Horn silver
- b)Silver chloride — Artificial rain
- c)Zinc phosphide — Rat poison
- d)Zinc sulphide — Philosopher’s wool
Correct answer is option 'C'. Can you explain this answer?
Which one of the following pairs is correctly matched?
a)
Silver iodide — Horn silver
b)
Silver chloride — Artificial rain
c)
Zinc phosphide — Rat poison
d)
Zinc sulphide — Philosopher’s wool
 | Kunal Roy answered |
Cerargyrite, also called Horn Silver, gray, very heavy halide mineral composed of silver chloride (AgCl); it is an ore of silver. Tiny particles of silver iodide are sprayed on a cloud from an aeroplane. The particles attract water drops from the cloud. When they form a drop that is large enough, it starts raining. Zinc phosphide is an inorganic compound that is used in pesticide products as a rodenticide.. Zinc oxide is also known as philosopher's wool.
The difference between a nuclear reactor and atomic bomb is that- a)no chain reaction takes place in nuclear reactor while in the atomic bomb there is a chain reaction.
- b)the chain reaction in nuclear reactor is controlled.
- c)the chain reaction in nuclear reactor is not controlled.
- d)no chain reaction takes place in atomic bomb while it takes place in nuclear reactor.
Correct answer is option 'B'. Can you explain this answer?
The difference between a nuclear reactor and atomic bomb is that
a)
no chain reaction takes place in nuclear reactor while in the atomic bomb there is a chain reaction.
b)
the chain reaction in nuclear reactor is controlled.
c)
the chain reaction in nuclear reactor is not controlled.
d)
no chain reaction takes place in atomic bomb while it takes place in nuclear reactor.
 | Pallavi Chakraborty answered |
There are two main fundamental differences between the design of an atomic bomb, and the design of a nuclear reactor. One difference is the way the fission reactions are controlled and the second difference stems from the enrichment of the fuel.
The order of appearance of the following with increasing temperature during the refining of crude oil is __________- a)Kerosene, gasoline, diesel
- b)Diesel, gasoline, kerosene
- c)Gasoline, kerosene, diesel
- d)Gasoline, diesel, kerosene
Correct answer is option 'C'. Can you explain this answer?
The order of appearance of the following with increasing temperature during the refining of crude oil is __________
a)
Kerosene, gasoline, diesel
b)
Diesel, gasoline, kerosene
c)
Gasoline, kerosene, diesel
d)
Gasoline, diesel, kerosene
 | Saanvi Reddy answered |
Petroleum products are usually grouped into three categories: light distillates (LPG, gasoline, naphtha), middle distillates (kerosene, diesel), heavy distillates and residuum (heavy fuel oil, lubricating oils, wax, asphalt). Hence, the correct option would be: Gasoline, kerosene, diesel.
The blue colour of water in the sea. What is the reason behind the phenomenon?- a)Refraction of the blue light by the impurities in sea water.
- b)Scattering of blue light by water molecules.
- c)Refraction of blue sky by sea water.
- d)Absorption of other colours except the blue colour by water molecules.
Correct answer is option 'B'. Can you explain this answer?
The blue colour of water in the sea. What is the reason behind the phenomenon?
a)
Refraction of the blue light by the impurities in sea water.
b)
Scattering of blue light by water molecules.
c)
Refraction of blue sky by sea water.
d)
Absorption of other colours except the blue colour by water molecules.
 | Anjana Nair answered |
Water molecules scatter blue wavelengths by absorbing the light waves, and then rapidly remitting the light waves in different directions. That is why there are mostly blue wavelengths that are reflected back to our eyes.
Helium is preferred to hydrogen in air balloons because it- a)is cheaper
- b)is less dense
- c)has greater lifting power
- d)does not form an explosive mixture with air
Correct answer is option 'D'. Can you explain this answer?
Helium is preferred to hydrogen in air balloons because it
a)
is cheaper
b)
is less dense
c)
has greater lifting power
d)
does not form an explosive mixture with air
 | Anshul Ghosh answered |
Hydrogen combines with oxygen with explosive force in the presence of a spark.Helium is an inert gas that will not burn or explode, so is much safer to use in balloons instead of hydrogen.
In the process of electroplating a utensil with zinc,- a)the utensil is made the cathode
- b)pure zinc is made the anode
- c)the utensil is made the cathode and pure zinc is made the anode
- d)the utensil is made the anode and pure zinc is made the cathode
Correct answer is option 'C'. Can you explain this answer?
In the process of electroplating a utensil with zinc,
a)
the utensil is made the cathode
b)
pure zinc is made the anode
c)
the utensil is made the cathode and pure zinc is made the anode
d)
the utensil is made the anode and pure zinc is made the cathode
| | Gauri Shah answered |
Understanding Electroplating with Zinc
Electroplating is a process used to deposit a layer of metal onto a surface, enhancing its appearance and corrosion resistance. In electroplating with zinc, the configuration of the cathode and anode is crucial.
Role of the Utensil as Cathode
- The utensil is made the cathode to attract zinc ions.
- As the cathode, it gains electrons during the electroplating process, allowing zinc to deposit onto its surface.
Role of Pure Zinc as Anode
- Pure zinc serves as the anode in the electroplating setup.
- When an electric current passes through the solution, zinc atoms from the anode oxidize and release zinc ions into the electrolyte solution.
Why Option C is Correct
- Option C states that the utensil is made the cathode and pure zinc is made the anode. This is the correct configuration for effective electroplating.
- The cathodic reaction involves the reduction of zinc ions from the solution, leading to their deposition on the utensil.
- The anodic reaction involves the oxidation of pure zinc, supplying a continuous flow of zinc ions into the solution, ensuring a consistent layer of zinc is deposited.
Conclusion
- The combination of the utensil as the cathode and pure zinc as the anode is essential for successful electroplating.
- This setup not only promotes uniform coating but also enhances the corrosion resistance of the utensil.
With this understanding, the electroplating process can be effectively implemented for various applications.
Electroplating is a process used to deposit a layer of metal onto a surface, enhancing its appearance and corrosion resistance. In electroplating with zinc, the configuration of the cathode and anode is crucial.
Role of the Utensil as Cathode
- The utensil is made the cathode to attract zinc ions.
- As the cathode, it gains electrons during the electroplating process, allowing zinc to deposit onto its surface.
Role of Pure Zinc as Anode
- Pure zinc serves as the anode in the electroplating setup.
- When an electric current passes through the solution, zinc atoms from the anode oxidize and release zinc ions into the electrolyte solution.
Why Option C is Correct
- Option C states that the utensil is made the cathode and pure zinc is made the anode. This is the correct configuration for effective electroplating.
- The cathodic reaction involves the reduction of zinc ions from the solution, leading to their deposition on the utensil.
- The anodic reaction involves the oxidation of pure zinc, supplying a continuous flow of zinc ions into the solution, ensuring a consistent layer of zinc is deposited.
Conclusion
- The combination of the utensil as the cathode and pure zinc as the anode is essential for successful electroplating.
- This setup not only promotes uniform coating but also enhances the corrosion resistance of the utensil.
With this understanding, the electroplating process can be effectively implemented for various applications.
Consider the following parts of spectra:
1. Visible
2. Infrared
3. Ultraviolet
4. Microwave Q. Which of the following is the correct sequence in which the wavelengths increase?- a)4, 3, 1, 2
- b)4, 1, 2, 3
- c)3, 2, 1, 4
- d)3, 1, 2, 4
Correct answer is option 'D'. Can you explain this answer?
Consider the following parts of spectra:
1. Visible
2. Infrared
3. Ultraviolet
4. Microwave
1. Visible
2. Infrared
3. Ultraviolet
4. Microwave
Q. Which of the following is the correct sequence in which the wavelengths increase?
a)
4, 3, 1, 2
b)
4, 1, 2, 3
c)
3, 2, 1, 4
d)
3, 1, 2, 4
 | Poulomi Nair answered |
Ultra-violet rays < visible light < infrared radiation < microwaves (0.1 micrometres) (0.7 micrometres ) (0.01 mm) (less than 10 cm)
Q.Which of the following substances is/are ozone depleting?
1. Chlorofluorocarbons
2. Halons
3. Carbon tetrachloride- a)1 only
- b)1 and 2 only
- c)2 and 3 only
- d)1, 2 and 3
Correct answer is option 'D'. Can you explain this answer?
Q.Which of the following substances is/are ozone depleting?
1. Chlorofluorocarbons
2. Halons
3. Carbon tetrachloride
1. Chlorofluorocarbons
2. Halons
3. Carbon tetrachloride
a)
1 only
b)
1 and 2 only
c)
2 and 3 only
d)
1, 2 and 3
 | Sagar Chakraborty answered |
All the three substances are responsible for ozone layer depletion in different ways. Which are: CFC—mostly used in refrigeration, air conditioning and heat pump systems. Halons—used historically as fire suppression agents and fire fighting, but now only allowed in very limited situations. Carbon tetrachloride (tetrachloromethane)—limited solvent used in laboratories and chemical and pharmaceutical industries.
What is “ Kiss of death” ?- a)A flower whose smell was the basis of a discovery on smell that led to award of 2004 Nobel Prize in Medicine.
- b)A chemical whose discovery ultimately won the scientists the 2004 Nobel Prize in Chemistry.
- c)A good wine flavour working on which scientist won the 2004 Nobel Prize in Chemistry.
- d)A network in brain associated with smell whose discovery led the scientists win the 2004 Nobel Prize in Physiology and Medicine.
Correct answer is option 'B'. Can you explain this answer?
What is “ Kiss of death” ?
a)
A flower whose smell was the basis of a discovery on smell that led to award of 2004 Nobel Prize in Medicine.
b)
A chemical whose discovery ultimately won the scientists the 2004 Nobel Prize in Chemistry.
c)
A good wine flavour working on which scientist won the 2004 Nobel Prize in Chemistry.
d)
A network in brain associated with smell whose discovery led the scientists win the 2004 Nobel Prize in Physiology and Medicine.
| | Saptarshi Patel answered |
Your question?
While tinning of brass utensils, the ammonium chloride powder used to clean the hot utensil produces fumes of- a)ammonia
- b)carbon monoxide
- c)hydrochloric acid
- d)ammonia and hydrochloric acid
Correct answer is option 'D'. Can you explain this answer?
While tinning of brass utensils, the ammonium chloride powder used to clean the hot utensil produces fumes of
a)
ammonia
b)
carbon monoxide
c)
hydrochloric acid
d)
ammonia and hydrochloric acid
 | Saanvi Reddy answered |
It produces fumes of ammonia and hydrochloric acid.
Hydrofluoric acid is not kept in glass bottles because it reacts with- a)visible light
- b)sodium oxide of glass
- c)aluminium oxide of glass
- d)silicon dioxide of glass
Correct answer is option 'D'. Can you explain this answer?
Hydrofluoric acid is not kept in glass bottles because it reacts with
a)
visible light
b)
sodium oxide of glass
c)
aluminium oxide of glass
d)
silicon dioxide of glass
 | Aashna Nambiar answered |
Hydrofluoric acid is one of the most dangerous acids known. It needs to be treated differently then even strong acids like sulphuric and hydrochloric. HF reacts with many materials, therefore, avoid contact with glass, concrete, metals, water, other acids, oxidizers, reducers, alkalis, combustibles, organics and ceramics.
Consider the following statements: If there were no phenomenon of capillarity
1. It would be difficult to use a kerosene lamp.
2. One would not be able to use a straw to consume a soft drink.
3. the blotting paper would fail to function.
4. the big trees that we see around would not have grown on the earth.Q. Which of the statements given above is/are correct?- a)1, 2 and 3 only
- b)1, 3 and 4 only
- c)2 and 4 only
- d)1, 2, 3 and 4
Correct answer is option 'B'. Can you explain this answer?
Consider the following statements: If there were no phenomenon of capillarity
1. It would be difficult to use a kerosene lamp.
2. One would not be able to use a straw to consume a soft drink.
3. the blotting paper would fail to function.
4. the big trees that we see around would not have grown on the earth.
1. It would be difficult to use a kerosene lamp.
2. One would not be able to use a straw to consume a soft drink.
3. the blotting paper would fail to function.
4. the big trees that we see around would not have grown on the earth.
Q. Which of the statements given above is/are correct?
a)
1, 2 and 3 only
b)
1, 3 and 4 only
c)
2 and 4 only
d)
1, 2, 3 and 4
 | Aashna Nambiar answered |
Except option (2), all are applications of capillary action. One would not be able to consume soft drink, if there is no atmospheric pressure, i.e., in vacuum.
Consider the following statements and select the correct code.
Assertion (A): A chemical reaction becomes faster at higher temperature.
Reason (R): At higher temperature, molecular motion becomes more rapid.- a)Both A and R are true and R is the correct explanation of A.
- b)Both A and R are true, but R is not correct explanation of A.
- c)A is true, but R is false.
- d)A is false, but R is true.
Correct answer is option 'A'. Can you explain this answer?
Consider the following statements and select the correct code.
Assertion (A): A chemical reaction becomes faster at higher temperature.
Reason (R): At higher temperature, molecular motion becomes more rapid.
Assertion (A): A chemical reaction becomes faster at higher temperature.
Reason (R): At higher temperature, molecular motion becomes more rapid.
a)
Both A and R are true and R is the correct explanation of A.
b)
Both A and R are true, but R is not correct explanation of A.
c)
A is true, but R is false.
d)
A is false, but R is true.
 | Avi Sengupta answered |
The rates of most reactions increase with a rise in temperature. Raising the temperature increases the fraction of molecules having very high kinetic energies. These are the ones most likely to react when they collide. The higher the temperature, the larger the fraction of molecules that can provide the activation energy needed for reaction.
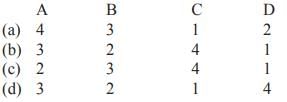
Match List-I with List-II and select the correct answer from the codes given below
- a)A
- b)B
- c)C
- d)D
Correct answer is option 'A'. Can you explain this answer?
Match List-I with List-II and select the correct answer from the codes given below


a)
A
b)
B
c)
C
d)
D
 | Meghana Shah answered |
Quarks have fractional electric charge values– either 1/2 or 2/3 times the elementary charge. The positron has an electric charge of +1e, a spin of 1/2, and has the same mass as an electron. A neutrino is an electrically neutral, weakly interacting elementary subatomic particle with halfinteger spin. All evidence suggest that neutrinos have mass but that their mass is tiny even by the standards of subatomic particles. Their mass has never been measured accurately.
Match List-I with List-II and select the correct answer from the codes given below:

- a)A
- b)B
- c)C
- d)D
Correct answer is option 'B'. Can you explain this answer?
Match List-I with List-II and select the correct answer from the codes given below:


a)
A
b)
B
c)
C
d)
D
 | Rithika Basak answered |
A nuclear reactor coolant — usually water but sometimes a gas or a liquid metal (like liquid sodium) or molten salt — is circulated past the reactor core to absorb the heat that it generates. The heat is carried away from the reactor and is then used to generate steam. Control rods that are made of a neutron poison are used to absorb neutrons. Absorbing more neutrons in a control rod means that there are fewer neutrons available to cause fission, so pushing the control rod deeper into the reactor will reduce its power output, and extracting the control rod will increase it. A neutron moderator is a medium that reduces the speed of fast neutrons, thereby turning them into thermal neutrons capable of sustaining a nuclear chain reaction involving uranium-235. Commonly used moderators include regular (light) water (roughly 75% of the world's reactors), solid graphite (20% of reactors) and heavy water (5% of reactors). Nuclear fuel is a material that can be 'burned' by nuclear fission or fusion to derive nuclear energy. Nuclear fuel can refer to the fuel itself, or to physical objects (for example, bundles composed of fuel rods) composed of the fuel material, mixed with structural, neutron moderating, or neutron reflecting materials. The most common fissile nuclear fuels are uranium-235 (235U) and plutonium-239 (239Pu).
Match List-I with List-II

- a)A
- b)B
- c)C
- d)D
Correct answer is option 'D'. Can you explain this answer?
Match List-I with List-II


a)
A
b)
B
c)
C
d)
D
 | Sagar Chakraborty answered |
The “plum pudding model” was put forward by J.J. Thomson in 1904. In this model, the atom is made up of negative electrons that float in a “soup” of positive charge, much like plums in a pudding or raisins in a fruit cake. In 1906, Thomson was awarded the Nobel Prize for his work in this field. Louise de Broglie suggested that electron exhibits a dual nature. Broglie also got Physics Noble Prize for his theory. In 1913, Bohr proposed his quantized shell model of the atom to explain how electrons can have stable orbits around the nucleus. Ernest Rutherford published his atomic theory describing the atom as having a central positive nucleus surrounded by negative orbiting electrons. This model suggested that most of the mass of the atom was contained in the small nucleus, and that the rest of the atom was mostly empty space.
Match List-I with List-II and select the correct answer from the codes given below:


- a)A
- b)B
- c)C
- d)D
Correct answer is option 'D'. Can you explain this answer?
Match List-I with List-II and select the correct answer from the codes given below:



a)
A
b)
B
c)
C
d)
D
 | Advait Chawla answered |
Morphine is an analgesic drug that is used to relieve severe pain. It was first isolated in 1804 by Friedrich Serturner, which is generally believed to be the first ever isolation of a natural plant alkaloid in history. Boric acid, also called orthoboric acid, is a weak acid of boron often used as an antiseptic or insecticide. Nickel silver, also known as German silver, is a copper alloy with nickel and often zinc. The usual formulation is 60% copper, 20% nickel and 20% zinc. Sodium is an extremely reactive metal and a powerful reducing agent. When exposed to air, it very quickly oxidizes. It also reacts violently to water. We store sodium under kerosene because kerosene is already highly reduced and won't react with sodium metal.
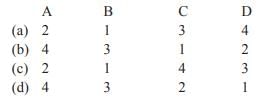
Match List-I with List-II and select the correct answer given below:
- a)A
- b)B
- c)C
- d)D
Correct answer is option 'A'. Can you explain this answer?
Match List-I with List-II and select the correct answer given below:


a)
A
b)
B
c)
C
d)
D
 | Avi Sengupta answered |
The electron was discovered in 1896, by the British physicist J. J. Thomson, using cathode rays while doing discharge tube experiments. In 1886, Eugen Goldstein discovered the existence of a new type of rays in discharge tube and named as anode rays or canal rays. Canal or anode rays travel in straight line and are deflected by electric field towards cathode which proves that they are composed of positively charged particles. These lightest positively charged particles were named and characterised as protons by Ernest Rutherford in 1919 who purposed Rutherford model for atom to explain the atomic structure. Anti-electron or in other words the positron was discovered in 1932 by Anderson, a physicists and he found that the bombardment of boron with an alpha particle resulted in the emission of this particle. In 1932, James Chadwick, an English physicist who had worked with Rutherford, detected neutrons.
Chapter doubts & questions for Chemistry - 1 Year Preparation for UPSC CSE 2026 is part of UPSC CSE exam preparation. The chapters have been prepared according to the UPSC CSE exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for UPSC CSE 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Chemistry - 1 Year Preparation for UPSC CSE in English & Hindi are available as part of UPSC CSE exam. Download more important topics, notes, lectures and mock test series for UPSC CSE Exam by signing up for free.
1 Year Preparation for UPSC CSE1013 videos|3871 docs|1305 tests |
Top Courses UPSC CSE
Related UPSC CSE Content
;
Signup to see your scores go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup

