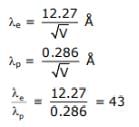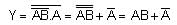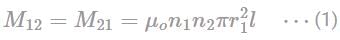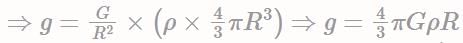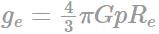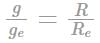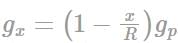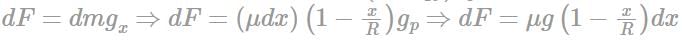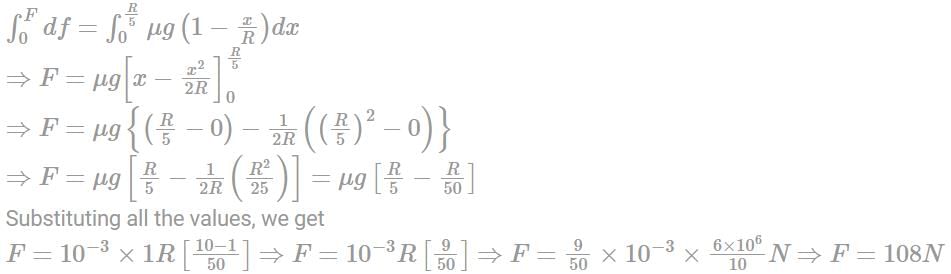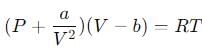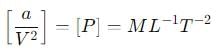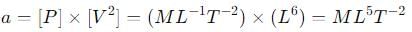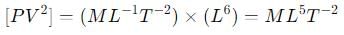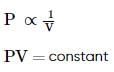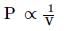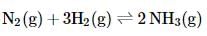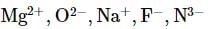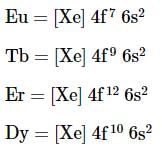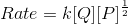COMEDK Mock Test - 5 - JEE MCQ
30 Questions MCQ Test COMEDK Mock Test Series - COMEDK Mock Test - 5
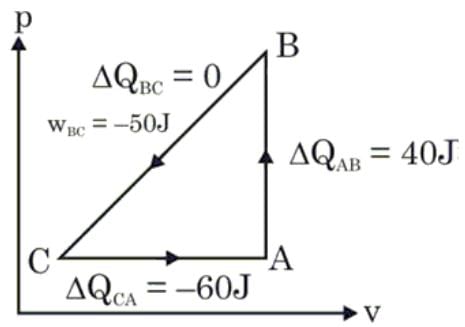
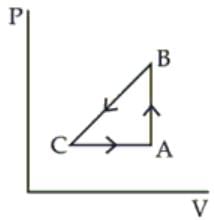
A sample of an ideal gas is taken through the cyclic process ABCA as shown in figure. It absorbs 40 J of heat during the part AB, no heat during BC and rejects 60 J of heat during CA. A work of 50 J is done on the gas during the part BC. The internal energy of the gas at A is 1560 J. The work done by the gas during the part CA is:

If wattless current flows in the AC circuit, then the circuit is:
The de-Broglie wavelength associated with an electron and a proton were calculated by accelerating them through same potential of 100 V. What should nearly be the ratio of their wavelengths? (mp = 1.00727 u, me = 0.00055 u)
Consider a two particle system with particles having masses m1 and m2. The first particle is pushed towards the centre of mass through a distance d. By what distance should the second particle be moved, so as to keep the centre of mass at the same position?
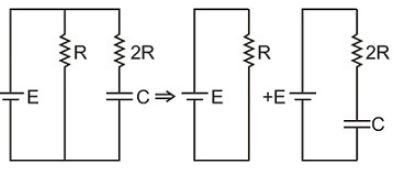
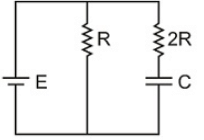
The time constant of charging of the capacitor shown in the diagram is
One end of a string of length l is connected to a particle of mass m and the other to a small peg on a smooth horizontal table. If the particle moves in a circle with speed v the net force on the particle ( directed towards the centre ) is:
Two long solenoids S1 and S2 have equal lengths and the solenoid S1 is placed co-axially inside the solenoid S2. If the current in both the solenoids is doubled, then the mutual inductance of both the solenoids will become:
A planet of radius R = 1/10 × ( radius of Earth) has the same mass density as Earth. Scientists dig a well of depth R/5 on it and lower a wire of the same length and of linear mass density 10-3 kg m-1 into it. If the wire is not touching anywhere, the force applied at the top of the wire by a person holding it in place is: (take the radius of Earth = 6 × 106 m and the acceleration due to gravity on Earth is 10 ms-2)
Find the amplitude of the S.H.M whose displacement y in cm is given by equation y= 3 sin157t + 4 cos157t, where t is time in seconds.
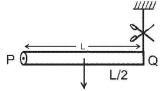
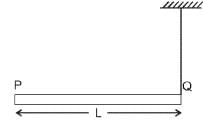
A rod PQ of mass M and length L is hinged at end P. The rod is kept horizontal by a massless string tied to point Q as shown in the figure. When the string is cut, the initial angular acceleration of the rod is:
[2013]
The Van der Waals equation for 1 mole of a real gas is
where P is the pressure, V is the volume, T is the absolute temperature, R is the molar gas constant and a and b are Van der Waal constants. The dimensions of a are the same as those of
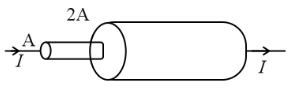
Two cylindrical rods of uniform cross-sectional area A and 2A, having free electrons per unit volume 2n and n respectively are joined in series. A constant current I flows through them in steady state. The ratio of the drift velocity of free electrons in the left rod to that of the right rod is ![]()
The length, breadth and thickness of a rectangular sheet of metal are 4.234 m, 1.005 m, and 2.01 cm respectively. Give the volume of the sheet to correct significant figures.
A metal conductor of length 1 m rotates vertically about one of its ends at angular velocity 5 radian per second. If the horizontal component of Earth's magnetic field is 0.2 x 10-4 T, then the emf developed between the two ends of the conductor is
Which of the following curves does not represent Boyle's law:
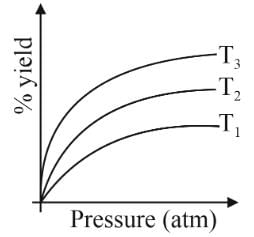
For the reaction N2(g)+3H2(g)⇌2NH3(g)
ΔH=−24 kJ/mole
The percentage yield of the reaction against pressure is plotted at three different temperature T1,T2,T3 as showning the adjacent figure. Pick out the correct statement among the following.
Among the lanthanide Eu, Tb, Er and Dy, which one readily forms stable divalent ions?
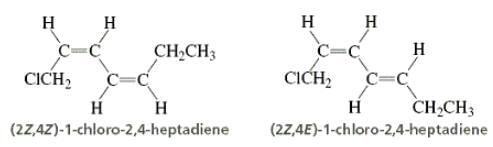
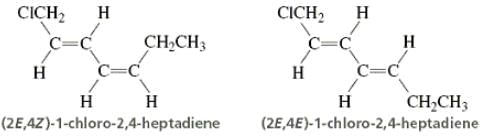
Directions: Consider the IUPAC names.
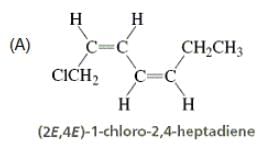
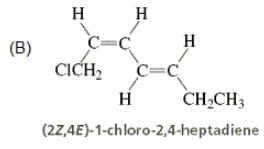
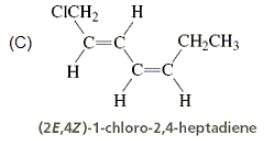
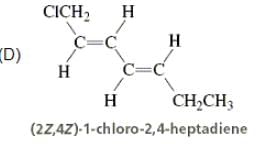
The incorrectly matched pairs are
Consider the reaction:
P + 2Q → R
When concentration of Q alone was tripled, the half-life did not change. When the concentration of P alone was made four times, the rate increased two times. The unit of rate constant for this reaction would be
In how many ways can 4 boys and 3 girls be seated in a row of 7 chairs if the boys and girls alternate?
When two dice are thrown, the probability of getting the sum 10 or 11 is
If α , β , γ are the roots of x3 − 2 x2 + 3x − 4 = 0, then the value of α 2β2 + β2γ2+ γ2α 2 is
Lines OA, OB are drawn from 0 with directions cosines proportional to < 1, -2, -1 > ans < 3, -2, 3 > respectively. The direction ratios of the normal to the plane AOB are
If the straight lines x = 1 + s, y = -3 - λs, z = 1 + λs and x = t/2, y = 1 + t, z = 2 - t, with parameters s and t respectively, are co-planar, then λ equals
If A and B are two independent events in a given sample space and the probability that both A and B occur is 0.16 while the probability that neither occurs is 0.36. The probabilities P(A) and P(B) are respectively
|
10 tests
|