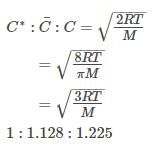JEE Main & Advanced Main 2013 Question Paper with Solutions Free Online
MCQ Practice Test & Solutions: JEE Main 2013 Question Paper with Solutions (90 Questions)
You can prepare effectively for JEE JEE Main & Advanced Mock Test Series 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "JEE Main 2013 Question Paper with Solutions". These 90 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 180 minutes
- - Number of Questions: 90
Sign up on EduRev for free to attempt this test and track your preparation progress.
PART A
Q. No. 1 -30 carry 4 marks each and 1 mark is deducted for every wrong answer.
Q. An unknown alcohol is treated with the “Lucas reagent” to determine whether the alcohol is primary,
secondary or tertiary. Which alcohol reacts fastest and by what mechanism:
secondary or tertiary. Which alcohol reacts fastest and by what mechanism:
Detailed Solution: Question 1
The first ionization potential of Na is 5.1 eV. The value of electron gain enthalpy of Na+ will be:
Detailed Solution: Question 2
Stability of the species 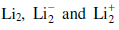 increases in the order of:
increases in the order of:
Detailed Solution: Question 3
The molarity of a solution obtained by mixing 750 mL of 0.5 (M) HCl with 250 mL of 2(M)HCl will be:
Detailed Solution: Question 4
Which of the following is the wrong statement?
Detailed Solution: Question 5
Four successive members of the first row transition elements are listed below with atomic numbers. Which
one of them is expected to have the highest 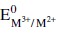 value?
value?
one of them is expected to have the highest
Detailed Solution: Question 6
A solution of (–) –1 – chloro –1 – phenylethane is toluene racemises slowly in the presence of a small
amount of SbCl5, due to the formation of :
amount of SbCl5, due to the formation of :
Detailed Solution: Question 7
The coagulating power of electrolytes having ions Na+, Al3+ and Ba2+ for arsenic sulphide sol increases in
the order:
the order:
Detailed Solution: Question 8
How many litres of water must be added to 1 litre of an aqueous solution of HCl with a pH of 1 to create an
aqueous solution with pH of 2?
aqueous solution with pH of 2?
Detailed Solution: Question 9
Which one of the following molecules is expected to exhibit diamagnetic behaviour?
Detailed Solution: Question 10
Which of the following arrangements does not represent the correct order of the property stated against it?
Detailed Solution: Question 11
Experimentally it was found that a metal oxide has formula M0.98O. Metal M, is present as M2+ and M3+ in
its oxide. Fraction of the metal which exists as M3+ would be:
its oxide. Fraction of the metal which exists as M3+ would be:
Detailed Solution: Question 12
compound with molecular mass 180 is acylated with CH3COCl to get a compound with molecular mass
390. The number of amino groups present per molecule of the former compound is:
390. The number of amino groups present per molecule of the former compound is:
Detailed Solution: Question 13
Given
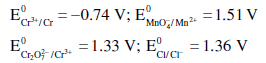
Based on the data given above, strongest oxidising agent will be:
Detailed Solution: Question 14
Arrange the following compounds in order of decreasing acidity:
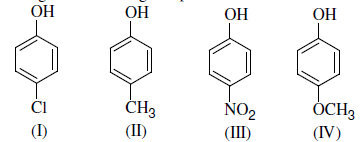
Detailed Solution: Question 15
The rate of a reaction doubles when its temperature changes from 300K to 310K. Activation energy of such
a reaction will be:
(R = 8.314 JK–1 mol–1 and log 2 = 0.301)
a reaction will be:
Detailed Solution: Question 16
Synthesis of each molecule of glucose in photosynthesis involves:
Detailed Solution: Question 17
Which of the following complex species is not expected to exhibit optical isomerism?
Detailed Solution: Question 18
A piston filled with 0.04 mol of an ideal gas expands reversibly from 50.0 mL to 375 mL at a constant
temperature of 37.00C. As it does so, it absorbs 208J of heat. The values of q and w for the process will be:
(R = 8.314 J/mol K) ( l n 7.5 = 2.01)
temperature of 37.00C. As it does so, it absorbs 208J of heat. The values of q and w for the process will be:
Detailed Solution: Question 19
A gaseous hydrocarbon gives upon combustion 0.72 g of water and 3.08 g of CO2. The empirical formula of
the hydrocarbon is:
the hydrocarbon is:
Detailed Solution: Question 20
The order of stability of the following carbocations:
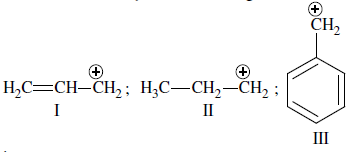 is
is
Detailed Solution: Question 21
Which of the following represents the correct order of increasing first ionization enthalpy for Ca, Ba, S, Se
and Ar?
and Ar?
Detailed Solution: Question 22
For gaseous state, if most probable speed is denoated by C*, average speed by 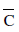 and mean square speed by C, then for a large number of molecules the ratios of these speeds are:
and mean square speed by C, then for a large number of molecules the ratios of these speeds are:
Detailed Solution: Question 23
The gas leaked from a storage tank of the Union Carbide plant in Bhopal gas tragedy was:
Detailed Solution: Question 24
Consider the following reaction:
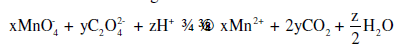
The values of x, y and z in the reaction are, respectively:
Detailed Solution: Question 25
Which of the following exists as covalent crystals in the solid state?
Detailed Solution: Question 26
Compound (A), C8H9Br, gives a white precipitate when warmed with alcoholic AgNO3. Oxidation of (A)
gives a acid (B), C8H6O4. (B) easily forms anhydride on heating. Identify the compound (A).
gives a acid (B), C8H6O4. (B) easily forms anhydride on heating. Identify the compound (A).
Detailed Solution: Question 27
Energy of an electron is given by 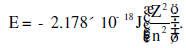 Wavelength of light required to excite an
Wavelength of light required to excite an
electron in an hydrogen atom from level n = 1 to n = 2 will be
(h = 6.62 × 10−34 Js and c = 3.0 × 108 ms−1)
electron in an hydrogen atom from level n = 1 to n = 2 will be
Detailed Solution: Question 28
An organic compound A upon reacting with NH3 gives B. On heating B gives C. C in presence of KOH
reacts with Br2 to give CH3CH2NH2. A is
reacts with Br2 to give CH3CH2NH2. A is
Detailed Solution: Question 29
In which of the following pairs of molecules/ions, both the species are not likely to exist?
Detailed Solution: Question 30
1 videos|238 docs|217 tests |