Electrovalency is favoured when polarization is minimum. According to Fajans’ rules, high charge on ions and a small cation with a large anion increase polarization, thereby reducing ionic (electrovalent) character. Hence, 2 and 4 do not favour electrovalency.
JEE Main Chemistry Mock Test- 5 Free Online Test 2026
Full Mock Test & Solutions: JEE Main Chemistry Mock Test- 5 (25 Questions)
You can boost your JEE 2026 exam preparation with this JEE Main Chemistry Mock Test- 5 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of JEE 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Total Questions: 25
- - Analysis: Detailed Solutions & Performance Insights
- - Sections covered: Section I, Section II
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
1-Phenylethanol can be prepared by the reaction of benzaldehyde with
Detailed Solution: Question 1
Detailed Solution: Question 2
Which of the following sequence is correct as per Aufbau principle?
Detailed Solution: Question 3
Which of the following is the least polar molecule?
Detailed Solution: Question 4
Consider the ground state of Cr atom (Z = 24). The numbers of electrons with the azimuthal quantum numbers, l = 1 and 2 are, respectively :
Detailed Solution: Question 5
Which of the following alkyl group has the maximum + I effect?
Detailed Solution: Question 6
Which of the following factors do not favour electrovalency?
1. Low charge on ions
2. High charge on ions
3. Large cation and small anion
4. Small cation and large anion
1. Low charge on ions
2. High charge on ions
3. Large cation and small anion
4. Small cation and large anion
Detailed Solution: Question 7
Detailed Solution: Question 8
Detailed Solution: Question 9
Which of the following shows geometrical isomerism ?
Detailed Solution: Question 10
The co-ordination no. of a metal in a co-ordination compounds is
Detailed Solution: Question 11
Four successive members of the first row transition elements are listed below with their atomic no. Which of them is expected to have the highest third ionisation enthalpy?
Detailed Solution: Question 12
Detailed Solution: Question 13
In decinormal solution, CH₃COOH acid is ionised to the extent of 1.3%. If log 1.3=0.11. What is the pH of the solution?
Detailed Solution: Question 14
Sodium nitroprusside reacts with sulphide ion to give a purple colour due to the formation of :
Detailed Solution: Question 15
Detailed Solution: Question 16
Chlorination of toluene with excess of chlorine in the presence of light and heat followed by treatment with aqueous NaOH gives
Detailed Solution: Question 17
In a polymer, 30% of molecules have a molecular weight of 20,000, 40% of molecules have a molecular weight of 30,000, and the rest, 30% of molecules, have a molecular weight of 60,000. The weight average molecular weight of the polymer is
Detailed Solution: Question 18
Electrolysis of molten sodium chloride leads to the formation of
Detailed Solution: Question 19
In acidic medium dichromate ion oxidizes ferrous ion to ferric ion. If the gram molecular weight of potassium dichromate is 294 grams, its gram equivalent weight is....grams.
Detailed Solution: Question 20
The sum of number of unpaired electrons in
[CoCl6]–3, [Cr(NH3)6]+3, [Zn(NH3)4]+2
[CoCl6]–3, [Cr(NH3)6]+3, [Zn(NH3)4]+2
Detailed Solution: Question 21
Sum of number of ions in aqueous solution of CrCl3.5NH3 and CrCl3.4NH3.
Detailed Solution: Question 22
Find the planner species out of the following species
SF2, SF4, SF6, SO2, SO3
SF2, SF4, SF6, SO2, SO3
Detailed Solution: Question 23
The total number of contributing structures showing hyperconjugation (involving C–H bonds) for the following carbocation is :
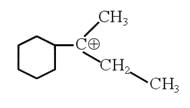

How many isomeric amines have the molecular formula C4H11N :
Detailed Solution: Question 25
|
360 docs|100 tests
|


