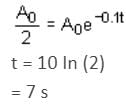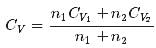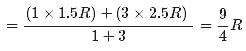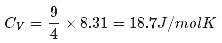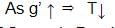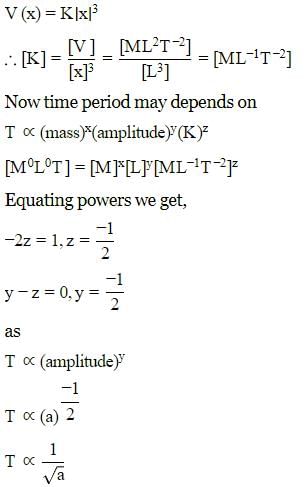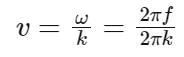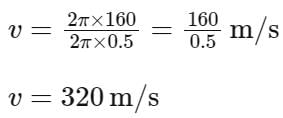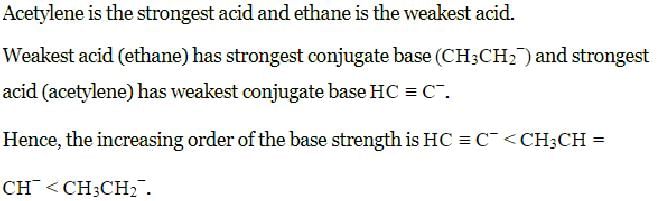JEE Main Part Test - 3 Free Online Test 2026
MCQ Practice Test & Solutions: JEE Main Part Test - 3 (75 Questions)
You can prepare effectively for JEE Mock Tests for JEE Main and Advanced 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "JEE Main Part Test - 3". These 75 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 180 minutes
- - Number of Questions: 75
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
A 14.5 kg mass, fastened to the end of a steel wire of unstretched length 1.0 m, is whirled in a vertical circle with an angular velocity of 2 rev/s at the bottom of the circle. The cross-sectional area of the wire is 0.065 cm2. Calculate the elongation of the wire when the mass is at the lowest point of its path.
Detailed Solution: Question 2
Two bullets A and B are fired horizontally with speed v and 2v respectively.which of the following is true
Detailed Solution: Question 3
The displacement of a damped harmonic oscillator is given by:
x(t) = e-0.1t cos (10πt + φ). Hence, t is in seconds.
The time taken for its amplitude of vibration to drop to half of its initial value is close to:
x(t) = e-0.1t cos (10πt + φ). Hence, t is in seconds.
Detailed Solution: Question 4
1 mole of a monoatomic gas is mixed with 3 moles of a diatomic gas. What is the molecular specific heat of the mixture at constant volume?
Detailed Solution: Question 5
The average distance a molecule can travel without colliding is called the
Detailed Solution: Question 6
Value of gas constant, R for one mole of a gas is independent of the
Detailed Solution: Question 7
According to kinetic theory of gases, 0K is that temperature at which for an ideal gas
Detailed Solution: Question 8
Four moles of an ideal diatomic gas is heated at constant volume from 20° C to 30° C. The molar specific heat of the gas at constant pressure (Cp) is 30.3 Jmol-1K-1 and the universal gas constant (R) is 8.3 Jmol-1K-1. The increase in internal energy of the gas is
Detailed Solution: Question 9
Three moles of an ideal monoatomic gas is initially in the state A shown in the adjoining pressure-temperature graph. It is taken to state B without changing its pressure. If R is the universal gas constant, the work done by the gas in this process is
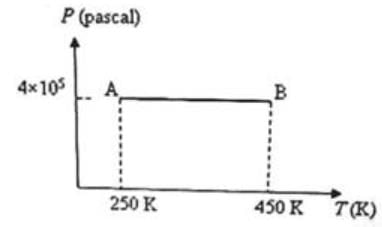

Detailed Solution: Question 10
A second pendulum is mounted in a space shuttle. Its period of oscillations will decrease when rocket is:
Detailed Solution: Question 11
Find the amplitude of the S.H.M whose displacement y in cm is given by equation y= 3 sin157t + 4 cos157t, where t is time in seconds.
Detailed Solution: Question 12
A particle executes linear simple harmonic motion with an amplitude of 2 cm. When the particle is at 1 cm from the mean position, the magnitude of its velocity is equal to that of its acceleration. Then its time period in seconds is:
Detailed Solution: Question 13
What is the maximum Kinetic energy and minimum potential energy of a harmonic oscillator with amplitude 0.03m, force constant 4×105 N/m and total mechanical energy of 230 J.
Detailed Solution: Question 14
The velocity and acceleration amplitudes of body executing simple harmonic motion is
Detailed Solution: Question 15
A particle of mass m is executing oscillation about the origin on X-axis. Its potential energy is V(x)=K∣x∣3. Where K is a positive constant. If the amplitude of oscillation is a, then its time period T is proportional to.
Detailed Solution: Question 16
Energy is supplied to the damped oscillatory system at the same rate at which it is dissipating energy, then the amplitude of such oscillations would become constant. Such oscillations are called
Detailed Solution: Question 17
Detailed Solution: Question 18
In the ideal case of zero damping, the amplitude of simple harmonic motion at resonance is:
Detailed Solution: Question 19
The necessary condition for phenomenon of interference to occur is
Detailed Solution: Question 20
The equation of wave is given by
Y = 10-2 sin 2π (160t – 0.5x + π/4)
where x and Y are in m and t in s. The speed of the wave is _______ km h-1.
Detailed Solution: Question 21
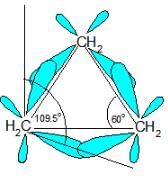
Which of the following correctly ranks the cycloalkanes in order of increasing ring strain per methylene group?
Detailed Solution: Question 22
Which of the following cycloalkanes exhibits the greatest molar heat of combustion per —CH2 — group?
Detailed Solution: Question 23
Detailed Solution: Question 24
Detailed Solution: Question 25
Arrange in increasing order of basicity. HC≡C−, CH3CH=CH−, CH3CH2−
Detailed Solution: Question 26
What are the hybridization and shapes of the following molecules?
(i) CH3F
(ii) HC ≡ N
(i) CH3F
(ii) HC ≡ N
Detailed Solution: Question 27
Direction (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
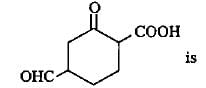
Q. Consider the following compounds.

The correct statement regarding properties of above mentioned compounds is/are
Detailed Solution: Question 28
Detailed Solution: Question 29
The compound shown below evolve hydrogen gas when refluxed with potassium metal, why?
Detailed Solution: Question 30
360 docs|100 tests |