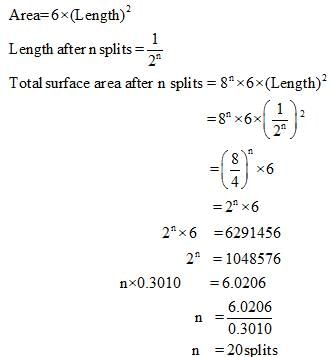Surface Chemistry (Old NCERT) - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: P. Bahadur Test: Surface Chemistry (Old NCERT) (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "P. Bahadur Test: Surface Chemistry (Old NCERT)". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 30 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
When a graph is plotted between log x/m and log p, it is straight line With an angle 45° and intercept 0.3010 on y-axis. If initial pressure is 0.3 atm, what will be the amount of gas adsorbed per gm of adsorbent :
Detailed Solution: Question 1
Which of the following statements about physical adsorption is not correct ?
Detailed Solution: Question 2
Following is the variation of physical adsorption with temperature:
Detailed Solution: Question 3
Finally divided catalyst has greater surface area and has greater catalytic activity then the compact solid. If a total surface area of 6291456 cm is required for adsorption of gaseous reaction in a catalysed reaction, then how many splits should be made of cube exactly 1 cm in length.
Detailed Solution: Question 4
Which of the following is not characteristic of chemisorption?
Detailed Solution: Question 5
A colloidal solution can be purified following the method of
Detailed Solution: Question 6
Gold number of a lyophilic sol is such property that:
Detailed Solution: Question 7
100 mL of a colloidal solution is completely precipitated by addition of 5 mL of 1 M Nacl solution . Calculate the coagulation value of Nacl .
Detailed Solution: Question 8
Detailed Solution: Question 9
Some type of gels like gelatin loose water slowly. The process is known as :
Detailed Solution: Question 10
During the adsorption of Krypton on activated charcoal at low temperature
Detailed Solution: Question 11
Size of colloidal particles may range from
Detailed Solution: Question 12
Detailed Solution: Question 13
Gold number of haemoglobin is 0.03. Hence, 100 mL of gold sol will require haemoglobin so that gold is not coagulated by 10 mL of 10% NaCl solution:
Detailed Solution: Question 14
Which one of the following statements is correct:
Detailed Solution: Question 15
What can adsorb larger volume of hydrogen gas :
Detailed Solution: Question 16
Statement-1 : All colloidal dispersions give very low osmotic pressure and show very small freezing point depression or boiling pointelevation.
Statement-2 : Tydall effect is due to scattering of light from the surface of colloidal particles.
Detailed Solution: Question 17
Statement-1 : The Brownian movement is due to the bombardment of collodial particles by the molecules of dispersion medium which are in the constant motion like molecules in a gas.
Statement-2 :Brownian movement provides a visible proof of the random kinetic motion of molecules in a liquid.
Detailed Solution: Question 18
Statement-1: In the coagulation of negatively charged arsenic sulphide soil, the coagulating power decreases in the order, Al3+ > Ba2+ > Na+.
Statement-2 : Generally greater the valence of coagulating ion, the greater is its power of coagulation.
Detailed Solution: Question 19
Assertion: Isoelectric point is pHpH at which colloidal can move towards either of electrode.
Reason: At isoelectric point coolidal particles becomes electrically netural .
Reason: At isoelectric point coolidal particles becomes electrically netural .
Detailed Solution: Question 20
335 videos|699 docs|300 tests |