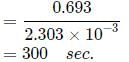Chemical Kinetics - Free MCQ Practice Test with solutions, JEE Chemistry
MCQ Practice Test & Solutions: R.C. Mukherjee Test: Chemical Kinetics (30 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "R.C. Mukherjee Test: Chemical Kinetics". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 45 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
The rate of a reaction is expressed in different ways as follows :
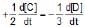 =
= 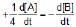
The reaction is :
Detailed Solution: Question 1
The rate constant for the forward reaction A(g) 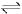 2B(g) is 1.5 × 10-3 s-1 at 100 K. If 10-5 moles of A and 100 moles of B are present in a 10 litre vessel at equilibrium then rate constant for the backward reaction at this temperature is
2B(g) is 1.5 × 10-3 s-1 at 100 K. If 10-5 moles of A and 100 moles of B are present in a 10 litre vessel at equilibrium then rate constant for the backward reaction at this temperature is
Detailed Solution: Question 2
Reaction A + B → C + D follow's following rate law : rate = k[A]+1/2[B]1/2. Starting with initial conc. of 1 M of A and B each, what is the time taken for concentration of A of become 0.25 M.
Given : k = 2.303 × 10-3 sec-1.
Detailed Solution: Question 3
Consider the following first order competing reactions :
X 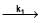 A+ B and y
A+ B and y 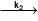 C+D
C+D
if 50% of the reaction of X was completed when 96% of the reaction of Y was completed, the ratio of their rate constants 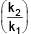 is
is
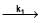 A+ B and y
A+ B and y 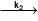 C+D
C+DDetailed Solution: Question 4
Units of rate constant for first and zero order reactions in terms of molarity (M) are respectively.
Detailed Solution: Question 5
The rate of a chemical reaction doubles for every 10°C rise of temperature. If the temperature is raised by 50°C, the rate of the reaction increases by about
Detailed Solution: Question 6
For a reaction pA + qB → products, the rate law expression is r = k[A]l[B]m, then :
Detailed Solution: Question 7
In the reaction : A + 2B → 3C + D, which of the following expression does not describe changes in the conc. of various species as a function of time :
Detailed Solution: Question 8
A first order reaction is 87.5% complete in an hour. The rate constant of the reaction is
Detailed Solution: Question 9
Half-life period of a second order reaction is
Detailed Solution: Question 10
A first order reaction is 50% completed in 20 minutes at 27°C and in 5 min at 47°C. The energy of activation of the reaction is
Detailed Solution: Question 11
For the first order reaction A → B + C, carried out at 27°C if 3.8 × 10-16% of the reactant molecules exists in the activated state, the Ea (activation energy) of the reaction is
Detailed Solution: Question 12
The rate constant, the activation energy and the Arrhenius parameter (A) of a chemical reaction at 25°C are 3.0 × 10-4 s-1, 104.4 kJ mol-1 and 6.0 × 10-4s-1respectively. The value of the rate constant at T → ∞ is
Detailed Solution: Question 13
The following mechanism has been proposed for the exothermic catalyzed complex reaction.
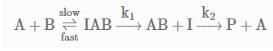
If k1 is much smaller than k2. The most suitable qualitative plot of potential energy (P.E.) versus reaction coordinate for the above reaction.
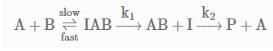
Detailed Solution: Question 14
The activation energy of a reaction at a given temperature is found to be 2.303 RT J mol–1. The ratio of rate constant to the Arrhenius factor is
Detailed Solution: Question 15
Consider  + heat, If activation energy for forward reaction is 100 kJ/mole then activation energy for backward reaction and heat of reaction is :
+ heat, If activation energy for forward reaction is 100 kJ/mole then activation energy for backward reaction and heat of reaction is :
 + heat, If activation energy for forward reaction is 100 kJ/mole then activation energy for backward reaction and heat of reaction is :
+ heat, If activation energy for forward reaction is 100 kJ/mole then activation energy for backward reaction and heat of reaction is :Detailed Solution: Question 16
In a reaction, the thershold energy is equal to :
Detailed Solution: Question 17
The first order rate constant k is related to temperature as log k = 15.0 - (106/T). Which of the following pair of value is correct ?
Detailed Solution: Question 18
When a graph between log K and 1/T is drawn a straight line is obtained. The temperature at which line cuts y-axis and x-axis.
Detailed Solution: Question 19
The rate constant, the activation energy and the frequency factor of a chemical reaction at 25°C are 3.0 × 10-2 s-1, 104.4 KJ mol-1 and 6.0 × 1014 s-1 respectively. The value of the rate constant as T → ∞ is :
Detailed Solution: Question 20
The rate data for the net reaction at 25°C for the reaction X + 2Y → 3Z are given below :
[X0] [Y0] Time required for [Z] to increase by 0.005 mol per litre.
0.01 0.01 72 sec
0.02 0.005 36 sec
0.02 0.01 18 sec
The intial rate (as given by Z) is :
Detailed Solution: Question 21
The rate of production of NH3 in N2 + 3H2 → 2NH3 is 3.4 kg min-1. The rate of consumption of H2 is :
Detailed Solution: Question 22
For a given reaction of first order it takes 20 min. for the conc. to drop from 1.0 M to 0.60 M. The time required for the conc. to drop from 0.60 M to 0.36 M will be :
Detailed Solution: Question 23
For a first order reaction, the concentration of reactant :
Detailed Solution: Question 24
Radioactivity of a sample (z = 22) decreases 90% after 10 years. What will be the half-life of the sample?
Detailed Solution: Question 25
Mathematical representation for t1/4 life for first order reaction is over is given by :
Detailed Solution: Question 26
For a reaction A → Products, the conc. of reactant C0, aC0, a2C0, a3C0............ after time interval 0, t, 2t ............ where 'a' is constant. Then :
Detailed Solution: Question 27
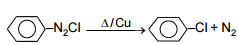 Half-life is independent of conc. of A. After 10 minutes volume N2 gas is 10 L and after complete reaction 50 L. Hence rate constant in min-1 :
Half-life is independent of conc. of A. After 10 minutes volume N2 gas is 10 L and after complete reaction 50 L. Hence rate constant in min-1 :
Detailed Solution: Question 28
In a zero-order reaction for every 10° rise of temperature, the rate is doubled. If the temperature is increased from 10°C to 100°C, the rate of the reaction will become
Detailed Solution: Question 29
In acidic medium the rate of reaction between (BrO3)- & Br- ions is given by the expression, –[d(BrO3-) /dt] = K[BrO3-][Br-][H +]2 It means :
Detailed Solution: Question 30
335 videos|697 docs|300 tests |