SRMJEEE Subject Wise & Full Length Chemistry Mock Test - 6 Free Online
Full Mock Test & Solutions: SRMJEEE Chemistry Mock Test - 6 (35 Questions)
You can boost your JEE 2026 exam preparation with this SRMJEEE Chemistry Mock Test - 6 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of JEE 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 50 minutes
- - Total Questions: 35
- - Analysis: Detailed Solutions & Performance Insights
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
Which of the following will not from a yellow precipitate on heating with an alkaline solution of iodine?
Detailed Solution: Question 2
Detailed Solution: Question 4
When H₂S gas is passed through the HCl containing aqueous solution of CuCl₂, HgCl₂, BiCl₃ and coCl₂, it does not precipitate out
Detailed Solution: Question 5
Detailed Solution: Question 6
Electrophile in the case of chlorination of benzene in presence of FeCl₃ is
Detailed Solution: Question 7
The greater the s- character in an orbital the ... is its energy
Detailed Solution: Question 8
The acid showing salt like character in aqueous solutions is
Detailed Solution: Question 9
Detailed Solution: Question 11
60 J of heat flows out from 600 g of water at 30°C into surroundings at 25°C.The net entropy change in universe is approximately
Detailed Solution: Question 12
Detailed Solution: Question 13
A glass bulb is filled with NO₂ gas and immersed in an ice bath at 0°C which becomes colourless after some time . This colourless gas will be
Detailed Solution: Question 14
Calculate the heat of formation Δ H of CO (in kcal) from the following data :
C(graphite) + O2(g) → CO2 (g); ΔH = -94K. cal
CO(g) + 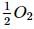 (g) → CO2 (g) ; ΔH = -68K. cal
(g) → CO2 (g) ; ΔH = -68K. cal
CO(g) +
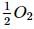 (g) → CO2 (g) ; ΔH = -68K. cal
(g) → CO2 (g) ; ΔH = -68K. calDetailed Solution: Question 15
Detailed Solution: Question 16
Detailed Solution: Question 18
Which one of the following reactions occur at the cathode?
Detailed Solution: Question 20
Which of the following applies to the reaction,
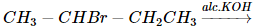
1. CH₃CH = CHCH₃(major product)
2. CH₂ = CH − CH₂CH₃(minor product)
2. CH₂ = CH − CH₂CH₃(minor product)
A compound having the formula NH₂CH₂COOH may behave
Detailed Solution: Question 22
A symmetrical molecule has 2 chiral carbon atoms. The no. of isomers is
Detailed Solution: Question 23
C-14 has a half-life of 5760 years. 100 mg of a sample containing C-14 is reduced to 25 mg in
Detailed Solution: Question 24
Detailed Solution: Question 25
Detailed Solution: Question 26
Which of the following is used for making horses, shoe heels and stoppers?
Detailed Solution: Question 28
In Duma's method, the gas which is collected in Nitrometer is
Detailed Solution: Question 29
Detailed Solution: Question 30
1 videos|4 docs|90 tests |
1 videos|4 docs|90 tests |


