SRMJEEE Subject Wise & Full Length Chemistry Mock Test - 7 Free Online
Full Mock Test & Solutions: SRMJEEE Chemistry Mock Test - 7 (35 Questions)
You can boost your JEE 2026 exam preparation with this SRMJEEE Chemistry Mock Test - 7 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of JEE 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 50 minutes
- - Total Questions: 35
- - Analysis: Detailed Solutions & Performance Insights
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
Detailed Solution: Question 1
Which of the following reaction cannot be used for the conversion RCO → RCH₂
Detailed Solution: Question 2
Detailed Solution: Question 3
Which one of the following has the minimum boiling point?
Detailed Solution: Question 4
Which of the following has the same mass as that of an electron?
Detailed Solution: Question 5
A one litre flask is full of brown bromine vapours. The intensity of brown colour of vapour will not decrease appreciably on adding to the flask some
Detailed Solution: Question 6
Detailed Solution: Question 7
The maximum amount of BaCl₄ precipitated on mixing BaCl₂ (0.5 M) with H₂SO₄ (1M) will correspond to
Detailed Solution: Question 8
The energy of C-C triple bond in acetylene in k.cals is
Detailed Solution: Question 9
Lower carboxylic acid are soluble in water due to
Detailed Solution: Question 10
Detailed Solution: Question 11
The difference between ΔH and ΔE for the combustion of methane at 27°C will be (in J mol-1)
Detailed Solution: Question 12
The chemical equilibrium of a reversible reaction is not influenced by
Detailed Solution: Question 13
If initial concentration is doubled, the time for half reaction is also doubled, the order of reaction is
Detailed Solution: Question 14
Given that (i) C + O₂ → CO₂ ; ∆H° = − xkJ, (ii) 2CO + O₂ → 2CO₂ ; ∆H° = −ykJ . The enthalpy of formation of carbon monoxide will be
Detailed Solution: Question 15
Detailed Solution: Question 16
Which of the following is isoelectronic as well as has the same structure as that of N₂O?
Detailed Solution: Question 18
The amount of Aluminium deposited when 0.1 Faraday current is passed through aluminium chloride will be (R = 27)
Detailed Solution: Question 19
In the ideal gas equation, the gas constant R has dimension of
Detailed Solution: Question 20
4gram of hydrocarbon on complete combustion gave 12.571 gram of CO2 and 5.143 gram of water. What is the empirical formula of the hydrocarbon
Detailed Solution: Question 21
Detailed Solution: Question 22
Which of the following compounds will show metamerism?
Detailed Solution: Question 23
A radioactive isotope decays at such a rate that after 192 minutes, only 1/16 of the original amount remains. The half-life of the radioactive isotope is
Detailed Solution: Question 24
The major product (70%-80%) of reaction between m-dinitrobenzene with (NH4)2Sx is
Detailed Solution: Question 25
Which of the following will not give the iodoform test?
Detailed Solution: Question 26
A mixture contains four solid organic compounds A, B, C and D. On heating only C changes from solid to vapour state. C can be separated from the rest in the mixture by
Detailed Solution: Question 28
Detailed Solution: Question 29
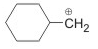
Following carbocation changes to more stable carbocation :
1 videos|4 docs|90 tests |
1 videos|4 docs|90 tests |


