Crystal Field Theory (CFT) & Colour of Complexes - Free MCQ Practice Test
MCQ Practice Test & Solutions: Test: Crystal Field Theory (CFT) & Colour of Complexes (25 Questions)
You can prepare effectively for NEET Chemistry Class 12 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Crystal Field Theory (CFT) & Colour of Complexes". These 25 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 35 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
Direction (Q. Nos. 1-10) This section contains 10 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q.
Among the following complexes the one which shows zero crystal field stabilisation energy is
Detailed Solution: Question 1
The wavelength of light absorbed is highest in
Detailed Solution: Question 2
The CFSE for octahedral [CoCI6]4- is 18000 cm-1. Then the CFSE for tetrahedral [CoCI4]2- will be
Detailed Solution: Question 3
The crystal field splitting energy (Δo) of
I. [CoBr6]3- II. [CoF6]3-
III. [Co(NCS)6]3- IV. [Co(CN)6]3- is in the order of
III. [Co(NCS)6]3- IV. [Co(CN)6]3- is in the order of
Detailed Solution: Question 4
A magnetic moment of 1.73 BM will be shown by one among the following
Detailed Solution: Question 5
The correct order o f ligand field strength is
Detailed Solution: Question 6
The crystal field splitting energies (CFSE) of high spin and low spin d6 metal complexes in octahedral complex in terms of Δo respectively are
Detailed Solution: Question 7
The Δt of the following complexes follows the order
I. [CoCI4]2-
II. [CoBr4]2-
III. [Co(NCS)4]2-
Detailed Solution: Question 8
In which of the following coordination entities, the magnitude Δo (CFSE in octahedral field) will be maximum ?
Detailed Solution: Question 9
The increasing order of wavelength of absorption for the complex ions
I. [Cr(NH3)6]3+
II. [CrCI6]3-
III. [Cr(H2O)6]3+
IV. [Cr(CN)6]3-
is :
IV. [Cr(CN)6]3-
Detailed Solution: Question 10
Direction (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q.
Which of the following metal ions cannot form both high spin and low spin octahedral complexes?
Detailed Solution: Question 11
Which is/are the correct statements?
Detailed Solution: Question 12
Detailed Solution: Question 13
Octahedral complex of Cr (III) will be
Detailed Solution: Question 14
Comprehension Type
Direction (Q. Nos. 16 and 17) This section contains a paragraph, describing theory, experiments, data, etc. Two questions related to the paragraph have.
Passage
In octahedral complexes due to repulsion between the ligands and d-orbitals, there is splitting of d-orbitals into two sets, i.e. two orbitals of higher energy called eg and three orbitals of lower energy called t2g. The difference of energy between the two sets of d-orbitals is called crystal field stabilisation energy denoted by Δo. For any given metal cation, the magnitude of Δo depends on the nature of ligands.
Q.
The CFSE for d7 configuration for strong ligand field is
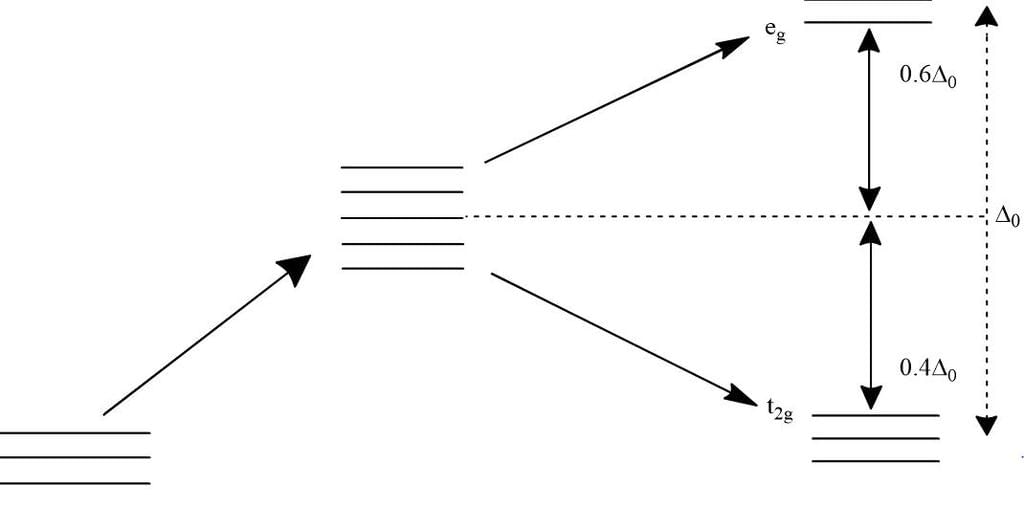

Detailed Solution: Question 15
In octahedral complexes due to repulsion between the ligands and d-orbitals, there is splitting of d-orbitals into two sets, i.e. two orbitals of higher energy called eg and three orbitals of lower energy called t2g. The difference of energy between the two sets of d-orbitals is called crystal field stabilisation energy denoted by Δo. For any given metal cation, the magnitude of Δo depends on the nature of ligands.
Q.
In the following complexes of manganese, the distribution of electrons in d-orbitals of manganese
i. [Mn(H2O)6]2+
ii. [Mn(CN)6]4-
Detailed Solution: Question 16
Matching List Type
Direction (Q. No. 18-19) Choices for the correct combination of elements from Column I and Column II are given as options (a), (b), (c) and (d) out of which one is correct.
Q.
Match the Column I with Column II and mark the correct option from the codes given below.


Detailed Solution: Question 17
Match the Column I with Column II and mark the correct option from the codes given below.






Detailed Solution: Question 18
One Integer Value Correct Type
Direction (Q. Nos. 20-24) This section contains 5 questions. When worked out will result in an integer from 0 to 9 (both inclusive).
Q.
Number of unpaired electrons in d6, low spin octahedral complex.
Detailed Solution: Question 19
The number of ligands which have strong crystal field splitting than
H2O among SCN-, NCS-, EDTA4- , 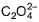 ,
, 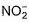 , Br-, PPh3, F-
, Br-, PPh3, F-
Detailed Solution: Question 20
Number of unpaired electrons in t2g and eg orbitals in weak octahedral ligand fields with d7 configuration.
Detailed Solution: Question 21
In tetrahedral complexes having weak field ligand, pairing starts with ...... electron.
Detailed Solution: Question 22
Statement Type
Direction (Q. No. 25) This section is based on Statement I and Statement II. Select the correct answer from the codes given below.
Q.
Statement I : The value of CFSE for the octahedral complex [Co(NH3)6]3+ is - 2.4Δo.
Statement II : The ammonia molecule is strong field ligand.
Detailed Solution: Question 23
Which of the following statements about unpaired electrons is correct?
Detailed Solution: Question 24
According to Crystal Field Theory, the colour of a coordination complex is mainly due to
Detailed Solution: Question 25
54 videos|290 docs|74 tests |



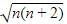 BM
BM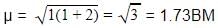
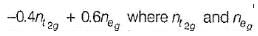 are the number of electron occupying the t2g and eg orbitatls respectively.
are the number of electron occupying the t2g and eg orbitatls respectively.