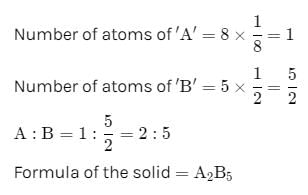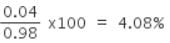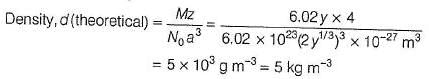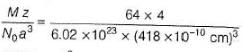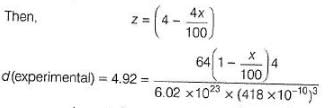Imperfections in Solids (Old NCERT) - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Imperfections in Solids (Old NCERT) (21 Questions)
You can prepare effectively for JEE Chapter-wise Tests for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Imperfections in Solids (Old NCERT)". These 21 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 30 minutes
- - Number of Questions: 21
Sign up on EduRev for free to attempt this test and track your preparation progress.
Only One Option Correct Type
This section contains 9 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q.
This section contains 9 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Detailed Solution: Question 1
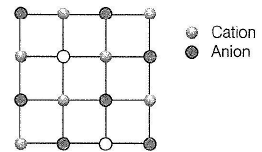
Structure shown in the figure represents
Detailed Solution: Question 2
Which of the following defects is also known as dislocation defect?
Detailed Solution: Question 3
Cations are present in the interstitial sites in
Detailed Solution: Question 4
Select the correct statement about non-stoichiometric compounds
Detailed Solution: Question 5
In a face-centred cubic lattice, atom A occupies the corners of the cube and atom B occupies the face-centred positions. If one atom of B is missing from one of the face-centred points, the formula of the compound is
Detailed Solution: Question 6
An element crystallises in fcc lattice having edge length 40 0 pm. Maximum radius of the atom which can be placed in the interstitial site without distorting the structure is
Detailed Solution: Question 7
Experimentally it was found that a metal oxide has formula M0.98O. Metal M, is present as M2+ and M3+ in its oxide. The fraction of the metal which exists as M3+ would be:
Detailed Solution: Question 8
If NaCI is doped with 10-3 mole % of SrCI2 then , number of cation ic vacancies is
Detailed Solution: Question 9
One or More than One Options Correct Type
This section contains 7 multiple type questions. Each question has 4 choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q.
AgBr(s) crystals can exhibit one or more types of defect which are
This section contains 7 multiple type questions. Each question has 4 choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Detailed Solution: Question 10
Substitution of foreign atom in the site of parent atom in crystal is a?
Detailed Solution: Question 11
As grain size of metal increases, Strength of metal:
Detailed Solution: Question 12
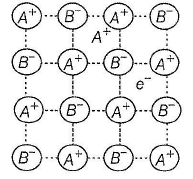
Select the correct statements about the given structure.
Detailed Solution: Question 13
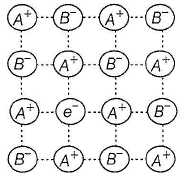
Select the correct statement(s) about following structure.
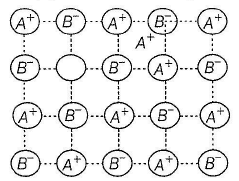
Detailed Solution: Question 14
Which of the following statement(s) is/are correct?
Detailed Solution: Question 15
Comprehension Type
This section contains a passage describing theory, experiments, data, etc. Two questions related to the paragraph have been given. Each question has only one correct answer out of the given 4 options (a), (b), (c) and (d).
Passage I
The crystal AB (rock salt structure) has m ole cular w eight 6.02 y u, where y is an arbitrary unit in u. Also, given minimum distance between cation and anion = y 1/3 nm and observed density = 20 kg m-3.
Q.
Theoretical density of AB is
This section contains a passage describing theory, experiments, data, etc. Two questions related to the paragraph have been given. Each question has only one correct answer out of the given 4 options (a), (b), (c) and (d).
Detailed Solution: Question 16
Passage
The crystal AB (rock salt structure) has m ole cular w eight 6.02 y u, where y is an arbitrary unit in u. Also, given minimum distance between cation and anion = y 1/3 nm and observed density = 20 kg m-3.
Q.
Defect present in the crystal is of the type
Detailed Solution: Question 17
Passage II
Titanium (II) oxide has a rock salt structure. X-ray diffraction data shows that the length of one edge of the cubic unit cell for TiO with 1:1 ratio of Ti to O is 418 pm and the density is 4.92 g cm-3. ( Atomic mass of Ti = 48).
Q.
Defect present in the crystal is of the type
Detailed Solution: Question 18
Passage II
Titanium (II) oxide has a rock salt structure. X-ray diffraction data shows that the length of one edge of the cubic unit cell for TiO with 1:1 ratio of Ti to O is 418 pm and the density is 4.92 g cm-3. ( Atomic mass of Ti = 48).
Q.
% of vacancies (defects) in the structure is
Detailed Solution: Question 19
Matching List Type
Choices for the correct combination of elements from Column I and Column II are given as options (a), (b), (c) and (d), out of which one is correct.


Choices for the correct combination of elements from Column I and Column II are given as options (a), (b), (c) and (d), out of which one is correct.
Detailed Solution: Question 20
One Integer Value Correct Type
This section contains 1 question, when worked out will result in an integer value from 0 to 9 (both inclusive)
Q.
Calcium crystallises in a face-centred cubic unit cell with a = 0.556 nm and density 1.4848 g/cm3. Percentage of Schottky defects in this crystal is ... .
This section contains 1 question, when worked out will result in an integer value from 0 to 9 (both inclusive)
Detailed Solution: Question 21
446 docs|929 tests |