Mendeleev's periodic table, Modern table - Free MCQ Practice Test
MCQ Practice Test & Solutions: Test: Mendeleev's periodic table, Modern periodic table (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Mendeleev's periodic table, Modern periodic table". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
Arrange the following in increasing order of ionic radii? 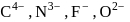
Detailed Solution: Question 1
The cause of periodicity of properties of elements is due to the repetition of similar:
Detailed Solution: Question 2
Tendency of an atom in a molecule to attract the shared pair of electron towards itself is called:
Detailed Solution: Question 3
Let electronegativity, ionisation energy and electron affinity be represented as EN, IP and EA respectively. Which one of the following equation is correct according to Mulliken?
Detailed Solution: Question 4
(X), (Y), (Z) are elements of the third period. Oxide of (X) is ionic, that of (Y) is amphoteric and of (Z) a giant molecule. (X), (Y) and (Z) will have atomic numbers in the order
Detailed Solution: Question 5
Which one of the following is correct order of second ionisation potential of 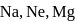 and
and 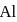 ?
?
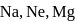 and
and 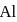 ?
?Detailed Solution: Question 6
Which one of the following arrangements represents the correct order of least negative to most negative electron gain enthalpy for 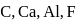 and
and 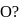
Detailed Solution: Question 7
The maximum covalency and highest oxidation state possible for an element with Z = 13 are respectively.
Detailed Solution: Question 8
Sum of first three ionization energies of 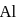 is
is 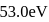 atom
atom 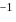 and the sum of first two ionization energies of
and the sum of first two ionization energies of 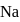 is
is 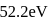 atom
atom 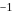 . Out of Al(III) and
. Out of Al(III) and 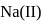
Detailed Solution: Question 9
The correct order of decreasing electronegatıvity values among the elements I-beryllium, II-oxygen, III-nitrogen and IV-magnesium is
Detailed Solution: Question 10
Which of the following sequence correctly represents the decreasing acidic nature of oxides?
Detailed Solution: Question 11
Correct order of first IP among following elements Be,B,C,N,O is
Detailed Solution: Question 12
Aqueous solutions of two compounds 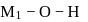 and
and 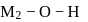 are prepared in two different beakers. If, the electronegativity of
are prepared in two different beakers. If, the electronegativity of 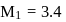 ,
, 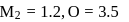 and
and 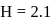 then the nature of two solutions will be respectively :
then the nature of two solutions will be respectively :
Detailed Solution: Question 13
An element having electronic configuration 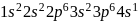 forms
forms
Detailed Solution: Question 14
The correct order of first ionisation enthalpy of group-13 elements is
Detailed Solution: Question 15
Which one of the following statements is incorrect in relation to ionisation enthalpy?
Detailed Solution: Question 16
If the ionization enthalpy and electron gain enthalpy of an element are 275 and  respectively, then the electronegativity of the elementon the Pauling scale is:
respectively, then the electronegativity of the elementon the Pauling scale is:
 respectively, then the electronegativity of the elementon the Pauling scale is:
respectively, then the electronegativity of the elementon the Pauling scale is:Detailed Solution: Question 17
The electronegativity order of the following elements is
Detailed Solution: Question 18
The successive ionization energy values of an element are 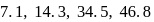 and
and 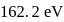 respectively. The element is likely to be:
respectively. The element is likely to be:
Detailed Solution: Question 19
Under normal condition which of the following electronic configuration is able to form dipositive ion
Detailed Solution: Question 20
335 videos|699 docs|300 tests |



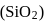 .
.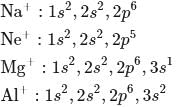
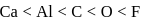
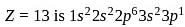
 is 6.
is 6.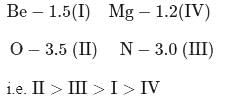

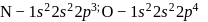
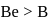 . Further IP of
. Further IP of 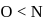 because atoms with fully or partly filled orbitals are most stable and hence have high ionisation energy.
because atoms with fully or partly filled orbitals are most stable and hence have high ionisation energy. and
and 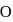 is
is 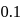 , which indicates
, which indicates 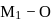 bond will be covalent, since
bond will be covalent, since 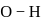 bond having more ionic character thus bond will break and
bond having more ionic character thus bond will break and 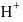 ions gets release and acidic solution is formed and whereas difference between electronegativity of
ions gets release and acidic solution is formed and whereas difference between electronegativity of 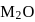 is
is 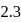 , thus,
, thus, 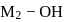 bond will break. Hence, solution will be basic in nature.
bond will break. Hence, solution will be basic in nature.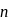 increases, the nuclear attraction over the outermost shell decreases, therefore removal of an electron will be easier.
increases, the nuclear attraction over the outermost shell decreases, therefore removal of an electron will be easier.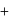 E.A.
E.A. 
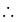 Electronegativity
Electronegativity 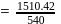
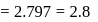
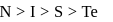 .
.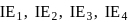 and
and 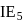 of an element are
of an element are 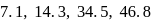 and
and 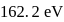 respectively. Therefore, We can see that there is a regular very close increment of ionization energy till the fourth ionization energy. After the fourth ionization energy, there is a sudden high jump in energy. It confirms that after losing four electrons, that element will achieve the noble gas configuration or stable configuration.
respectively. Therefore, We can see that there is a regular very close increment of ionization energy till the fourth ionization energy. After the fourth ionization energy, there is a sudden high jump in energy. It confirms that after losing four electrons, that element will achieve the noble gas configuration or stable configuration.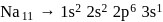
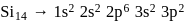
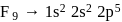
 and it is most likely to be silicon.
and it is most likely to be silicon.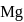 has only two electrons in the 3 s-orbital and hence its
has only two electrons in the 3 s-orbital and hence its 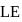 . is lowest, i.e. it has the maximum tendency to from di-positive ions.
. is lowest, i.e. it has the maximum tendency to from di-positive ions.