Metallurgy of Some Important Elements (Al, Zn, Au) (Old NCERT) - Free MCQ
MCQ Practice Test & Solutions: Test: Metallurgy of Some Important Elements (Al, Zn, Au) (Old NCERT) (25 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Metallurgy of Some Important Elements (Al, Zn, Au) (Old NCERT)". These 25 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 35 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
Only One Option Correct Type
Direction (Q. Nos. 1-10) This section contains 10 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q.
In the electrolytic refining of zinc
Detailed Solution: Question 1
When compared to ΔG° for the formation of Al2O3 the ΔG° for the formation of Cr2O3 is
Detailed Solution: Question 2
During the extraction of gold the following reactions take place
Au + CN- + H2O 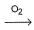 X,
X,
X + Zn → Y + Au
Here, X and Y respectively are
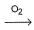 X,
X,X + Zn → Y + Au
Detailed Solution: Question 3
Gold is extracted by hydrometallurgical process based on its property
Detailed Solution: Question 4
During Hoope’s process for electrolytic refining of Al, the middle layer is of
Detailed Solution: Question 5
Thermite is a mixture of iron oxide and
Detailed Solution: Question 6
Which is an alloy of aluminium?
Detailed Solution: Question 7
Which one of the following metals has the largest abundance in the earth’s crust?
Detailed Solution: Question 8
The processes used in the refining of aluminium and zinc metals respectively are
Detailed Solution: Question 9
In sulphatizing roasting of ZnS products are
Detailed Solution: Question 10
One or More than One Options Correct Type
Direction (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q.
Purification of alumina is called
Detailed Solution: Question 11
Among the following statements the correct one is
Detailed Solution: Question 12
The alloys having zinc metal are
Detailed Solution: Question 13
Which is a zinc compound?
Detailed Solution: Question 14
Which is not a mineral of Al?
Detailed Solution: Question 15
Comprehension Type
Direction (Q. Nos. 16 and 17) This section contains a paragraph, describing theory, experiments, data, etc. Two questions related to the paragraph have been given. Each question has only one correct answer among the four given options (a), (b), (c) and (d).
Passage
The chief ore of zinc is ZnS.The ore is purified by floatation process and then heated in air which converts ZnS into ZnO. ZnO can be reduced with coke in Belgian process to get zinc which is purified by electrolysis.
Alternately zinc can be extracted from calamine .This after purification by levigation, subjected to calcination to get oxide. This on reduction by Belgian method followed by electrolytic refining yields pure zinc.
Q.
Zinc oxide is converted into zinc by
Alternately zinc can be extracted from calamine .This after purification by levigation, subjected to calcination to get oxide. This on reduction by Belgian method followed by electrolytic refining yields pure zinc.
Detailed Solution: Question 16
Passage
The chief ore of zinc is ZnS.The ore is purified by floatation process and then heated in air which converts ZnS into ZnO. ZnO can be reduced with coke in Belgian process to get zinc which is purified by electrolysis.
Alternately zinc can be extracted from calamine .This after purification by levigation, subjected to calcination to get oxide. This on reduction by Belgian method followed by electrolytic refining yields pure zinc.
Q.
Which of the following graph in the given figure is the Ellingham diagram of ZnO?
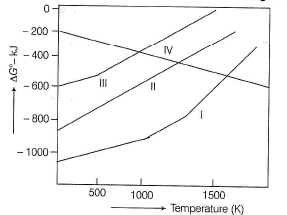
Alternately zinc can be extracted from calamine .This after purification by levigation, subjected to calcination to get oxide. This on reduction by Belgian method followed by electrolytic refining yields pure zinc.
Detailed Solution: Question 17
Matching List Type
Direction (Q. Nos. 18 and 19) Choices for the correct combination of elements from Column I and Column II are given as options (a), (b), (c) and (d), out of which one is correct.
Q.
Match the Column I with Column II and mark the correct option from the codes given below.

Detailed Solution: Question 18
Match the Column I with Column II and mark the correct option from the codes given below.
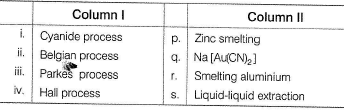
Detailed Solution: Question 19
One Integer Value Correct Type
Direction (Q, Nos. 20-24) This section contains 5 questions. When worked out will result in an integer from 0 to 9 (both inclusive).
Q.
Gibbsite formula is Al2O3 . xH2O. Here, value of x is
Detailed Solution: Question 20
Gold is soluble in aqua-regia which is cone. HNO3 and cone. HCI in 1: x ratio. Then the value of x is
Detailed Solution: Question 21
Duralumin is an important alloy of aluminium .This has 95% Al and ...... % copper.
Detailed Solution: Question 22
Al is amphoteric metal and liberate hydrogen by reacting with both with dilute acids and bases . 2 moles of Al metal when reacted with either acid or base, the number of moles of dihydrogen liberated is ......
Detailed Solution: Question 23
During the Hall-Heroult process of preparing Al metal from purified bauxite, if 3 moles of O2 is formed at anode the number of moles of Al formed at cathode is
Detailed Solution: Question 24
Statement Type
Direction (Q. No. 25) This section is based,on Statement I and Statement II. Select the correct answer from the codes given below.
Q.
Statement I : Many gem stones are impure forms of alumina (Al2O3).
Statement II : In cyanide process gold (Au) is treated with a cone, solution of sodium or potassium cyanide.
Detailed Solution: Question 25
332 videos|699 docs|300 tests |


