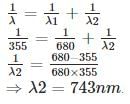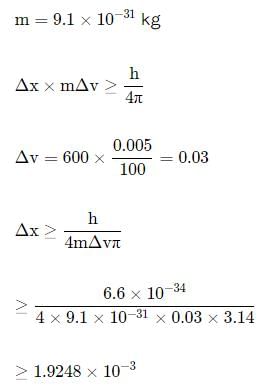Structure of Atom - Free MCQ Practice Test with solutions, NEET Chemistry
MCQ Practice Test & Solutions: Test: Structure of Atom (22 Questions)
You can prepare effectively for NEET Chemistry Class 11 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Structure of Atom". These 22 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 22 minutes
- - Number of Questions: 22
Sign up on EduRev for free to attempt this test and track your preparation progress.
Electronic configuration of the element having atomic number 24.
Detailed Solution: Question 1
According to quantum mechanics ψ2(r) the wave function squared gives
Detailed Solution: Question 2
Spin quantum number with two spin states of the electron represented by two arrows, ↑ (spin up) and ↓ (spin down) was introduced to account for
Detailed Solution: Question 3
Values of e/m (charge/mass) in the categories alpha particle (α), electron (e) and protons (p) increase in the order:
Detailed Solution: Question 4
A gas absorbs a photon of 355 nm and emits at two wavelengths. If one of the emissions is at 680 nm, the other is at:
Detailed Solution: Question 5
Detailed Solution: Question 6
The energy associated with the first orbit in the hydrogen atom is -2.18 x 10−18 J/atom. What is the energy associated with the fifth orbit?
Detailed Solution: Question 7
Detailed Solution: Question 8
Due to the presence of electrons in the inner shells, the electron in the outer shell will not experience the full positive charge of the nucleus (Ze). This is known as
Detailed Solution: Question 9
Maximum number of electrons in a subshell with l = 3 and n = 4 is
Detailed Solution: Question 10
Arrange the electrons present in the 4d, 3d, 4p and 3p orbitals in order of increasing energies
Detailed Solution: Question 11
Detailed Solution: Question 12
The Aufbau principle states : In the ground state of the atoms, the orbitals are filled in order of
Detailed Solution: Question 13
Radio frequency region of the electromagnetic spectrum is used for broadcasting. It is
Detailed Solution: Question 14
In an atom, an electron is moving with a speed of 600m/s with an accuracy of 0.005%. Certainity with which the position of the electron can be located is (h = 6.6 ×10−34 Js)
Detailed Solution: Question 15
Give the name and atomic number of the inert gas atom in which the total number of d-electrons is equal to the difference between the numbers of total p and total s electrons.
Detailed Solution: Question 16
Detailed Solution: Question 17
Detailed Solution: Question 18
Detailed Solution: Question 19
An cation A3+ has 18 electrons. Write the atomic number of A.
Detailed Solution: Question 20
Emission spectrum of a material results from the material's (atom or molecules)
Detailed Solution: Question 21
Dalton's atomic theory could not explain one of the following
Detailed Solution: Question 22
98 videos|243 docs|71 tests |