Van't Hoff Factor (Liquid Solution) - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Van't Hoff Factor (Liquid Solution) (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Van't Hoff Factor (Liquid Solution)". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 30 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
Only One Option Correct Type
This section contains 7 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct
Q.
The degree of dissociation (α ) o f weak electrolyte AxBy is related to van’t Hoff factor (i) by the expression
[AIEEE 2011]
This section contains 7 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct
Detailed Solution: Question 1
0.004 M Na2S04 aqueous solution is isotonic with 0.01 M glucose solution at 300 K. Thus, degree of dissociation of Na2S04 is
Detailed Solution: Question 2
When 20 g of naphthoic acid (C11H8O2) is dissolved in 50 g of benzene, a freezing point depression of 2 K is observed. [Kf (benzene) = 1.72Kmol-1 kg]. The van’t Hoff factor (i) is
[IIT - JEE 2007]
Detailed Solution: Question 3
An aqueous solution of a solute AB has boiling point of 101.08° C and freezes at -1 .80 °C . AB is found to be 100% ionised at boiling point. If Kb /Kf = 0.3, then AB
Detailed Solution: Question 4
Osmolarity of 0.02 M potassium ferrocyanide solution at 300 K is (assume solute is 100% ionised)
Detailed Solution: Question 5
AB and CD are two non -volatile solutes dissolved in same solvent, showing following behaviour
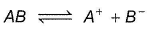
AB is ionised in solvent, α = degree of ionisation
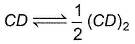
CD is dimerised in solvent, 3 = degree of dimerisation
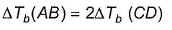
If α = 0.80, then β is
Detailed Solution: Question 6
Detailed Solution: Question 7
One or More than One Options Correct Type
This section contains 3 multiple type questions. Each question has 4 choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q.
Following solutions have been provided at temperature T K.
I. 1M aqueous glucose solution.
II. 1M aqueous sodium chloride solution.
III. 1M aqueous ammonium phosphate solution.
IV. 1M benzoic acid in benzene.
Select correct statements for the above solutions.
This section contains 3 multiple type questions. Each question has 4 choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
I. 1M aqueous glucose solution.
II. 1M aqueous sodium chloride solution.
III. 1M aqueous ammonium phosphate solution.
IV. 1M benzoic acid in benzene.
Select correct statements for the above solutions.
Detailed Solution: Question 8
Which of the following statements are correct?
Detailed Solution: Question 9
Matching List Type
Choices for the correct combination of elements from Column I and Column II are given as options (a), (b), (c) and (d), out of which one is correct
Q.
Match the different solutions in Column I with their ΔTb in Column II and select the answer from the codes given below.


Choices for the correct combination of elements from Column I and Column II are given as options (a), (b), (c) and (d), out of which one is correct
Detailed Solution: Question 10
In a 0.2 molal aqueous solution of a weak acid (HX), depression in freezing point is 0.383° Kf is 1.86° mol-1 kg. Assum e molarity equal to molality.
Match the parameters in Column I with their values in Column II and select the answer from the codes given below.


Match the parameters in Column I with their values in Column II and select the answer from the codes given below.
Detailed Solution: Question 11
Comprehension Type
This section contains a passage describing theory, experiments, data, etc. Two questions related to the paragraph have been given. Each question has only one correct answer out of the given 4 options (a), (b), (c) and (d)
Passage I
Hgl2 is insoluble in water. This solute is added into Kl solution.
Q.
In the above case,
This section contains a passage describing theory, experiments, data, etc. Two questions related to the paragraph have been given. Each question has only one correct answer out of the given 4 options (a), (b), (c) and (d)
Detailed Solution: Question 12
Hgl2 is insoluble in water. This solute is added into Kl solution.
Q.
Passage I van’t Hoff factor in this example
Detailed Solution: Question 13
Passage II
A solution containing 10 g of a dibasic acid in 1000 g of water freezes at -0.15° C. 10 mL of the acid is neutralised by 12 mL of 0.1 N NaOH solution. [Kf (H20 ) = 1.86° mol-1 kg]
Q.
van’t Hoff factor (/') of the dibasic acid is
Detailed Solution: Question 14
A solution containing 10 g of a dibasic acid in 1000 g of water freezes at -0.15° C. 10 mL of the acid is neutralised by 12 mL of 0.1 N NaOH solution. [Kf (H20 ) = 1.86° mol-1 kg]
Q.
pH of the solution of the acid is
Detailed Solution: Question 15
Passage III
A solution containing 0.684 g of cane sugar (C12H22011) in 100 g water freezes at - 0.037° C. A solution containing 0.585 g of NaCI in 100 g water freezes at - 0.342° C.
Q.
Apparent molecular weight of NaCI is
Detailed Solution: Question 16
Passage III
A solution containing 0.684 g of cane sugar (C12H22011) in 100 g water freezes at - 0.037° C. A solution containing 0.585 g of NaCI in 100 g water freezes at - 0.342° C.
Q.
van’t Hoff factor (i) is
Detailed Solution: Question 17
Direction:
This section contains 5 questions. When worked out will result in an integer from 0 to 9 {both inclusive).
Q.
Relative decrease in vapour pressure of an aqueous NaCI solution is 0.167. Thus, number of moles of NaCI present in 180 g of H20 is ... .
Detailed Solution: Question 18
Aqueous solution of barium phosphate which is 100% ionised has ΔTf / Kf as 0.40. Hence, given solution is x * 10-2 molal. What is the value of x?
Detailed Solution: Question 19
0.002 molal aqueous solution of an ionic compound [Co(NH3)5(N02)]CI freezes at - 0.00732° C. [Kf (H20) = 1.86° mol-1kg]. How many moles of ions does 1.0 mole of the salt produce on being dissolved in water?
Detailed Solution: Question 20
335 videos|697 docs|300 tests |