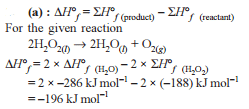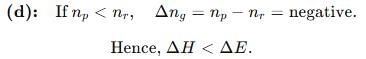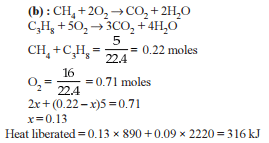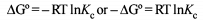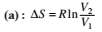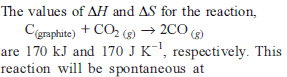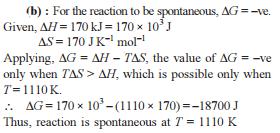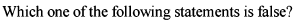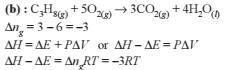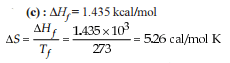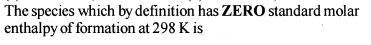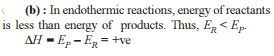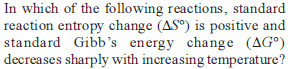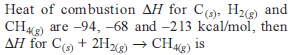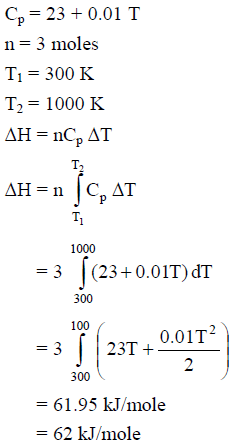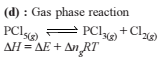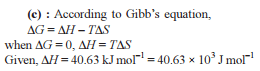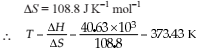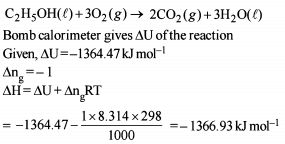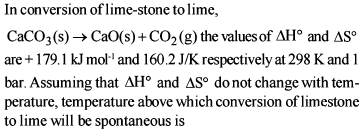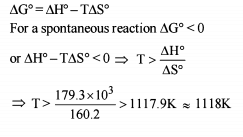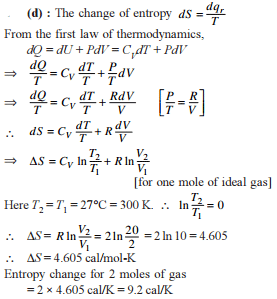Thermodynamics - 1 - Free MCQ Test with solutions for JEE Chemistry
MCQ Practice Test & Solutions: Thermodynamics - 1 (30 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Thermodynamics - 1". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
The pressure-volume work for an ideal gas can be calculated using the expression
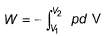
This type of work can also be calculated using the area under the curve within the specified limits. When an ideal gas is compressed, (I) reversibly or (II) irreversibly, then
Detailed Solution: Question 1
In the following case, find the option with the correct matching
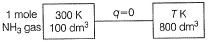
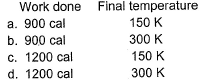
Detailed Solution: Question 2
1.0 mole of a monoatomic ideal gas is expanded from state I to state II at 300 K.
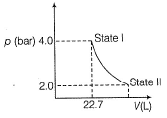
Thus, work done is
Detailed Solution: Question 3
1 mole of a diatomic gas is contained in a piston. It gains 50.0 J of heat and work is done on the surrounding by the system is -100 J. Thus,
Detailed Solution: Question 4
A sample containing 1.0 mole of an ideal gas is expanded isothermally and reversibly to ten time of its original volume, in two separate experiments. The expansion is carried out 300 K and at 600 K, respectively. Choose the correct option.
Detailed Solution: Question 5
Change in enthalpy for reaction,
2H2O2(l) → 2H2O(l) + O2(g)
if heat of formation of H2O2(l) and H2O(l) are −188 and −286 kJ/mol respectively, is
2H2O2(l) → 2H2O(l) + O2(g)
if heat of formation of H2O2(l) and H2O(l) are −188 and −286 kJ/mol respectively, is
Detailed Solution: Question 6
If ΔH is the change in enthalpy and ΔE, the change in internal energy accompanying a gaseous reaction, then
Detailed Solution: Question 7
During compression of a spring the work done is 10 kJ and 2 kJ escaped to the surroundings as heat. The change in internal energy, ΔU (in kJ) is :
Detailed Solution: Question 8
Two moles of an ideal gas is expanded isothermally and reversibly from 1 litre to 10 litre at 300 K. The enthalpy change (in kJ) for the process is
Detailed Solution: Question 9
When 5 litres of a gas mixture of methane and propane is perfectly combusted at 0°C and 1 atmosphere, 16 litres of oxygen at the same temperature and pressure is consumed. The amount of heat released from this combustion in kJ (ΔH_comb. (CH₄) = 890 kJ mol⁻¹, ΔH_comb. (C₃H₈) = 2220 kJ mol⁻¹) is
Detailed Solution: Question 10
Consider the following reactions:
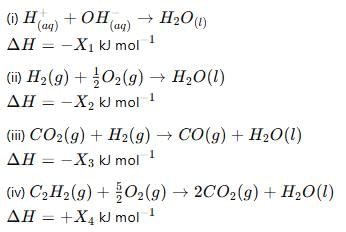
Enthalpy of formaton of H2O(l)
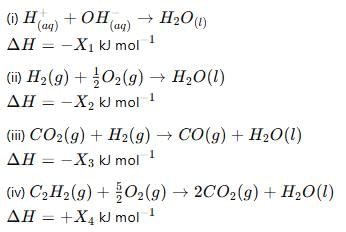
Detailed Solution: Question 11
The correct relationship between free energy change in a reaction and the corresponding equilibrium constant
Kc is
Kc is
Detailed Solution: Question 12
Detailed Solution: Question 13
Detailed Solution: Question 14
Detailed Solution: Question 15
Detailed Solution: Question 16
Detailed Solution: Question 17
Detailed Solution: Question 18
Detailed Solution: Question 19
Detailed Solution: Question 20
Detailed Solution: Question 21
Detailed Solution: Question 22
For silver, Cp(JK–1 mol–1) = 23 + 0.01 T. If the temperature (T) of 3 moles of silver is raised from 300 K to 1000 K at 1 atm pressure, the value of ΔH will be close to -
Detailed Solution: Question 23
Detailed Solution: Question 24
Detailed Solution: Question 25
Detailed Solution: Question 26
Detailed Solution: Question 27
Detailed Solution: Question 28
Detailed Solution: Question 29
Detailed Solution: Question 30
335 videos|699 docs|300 tests |