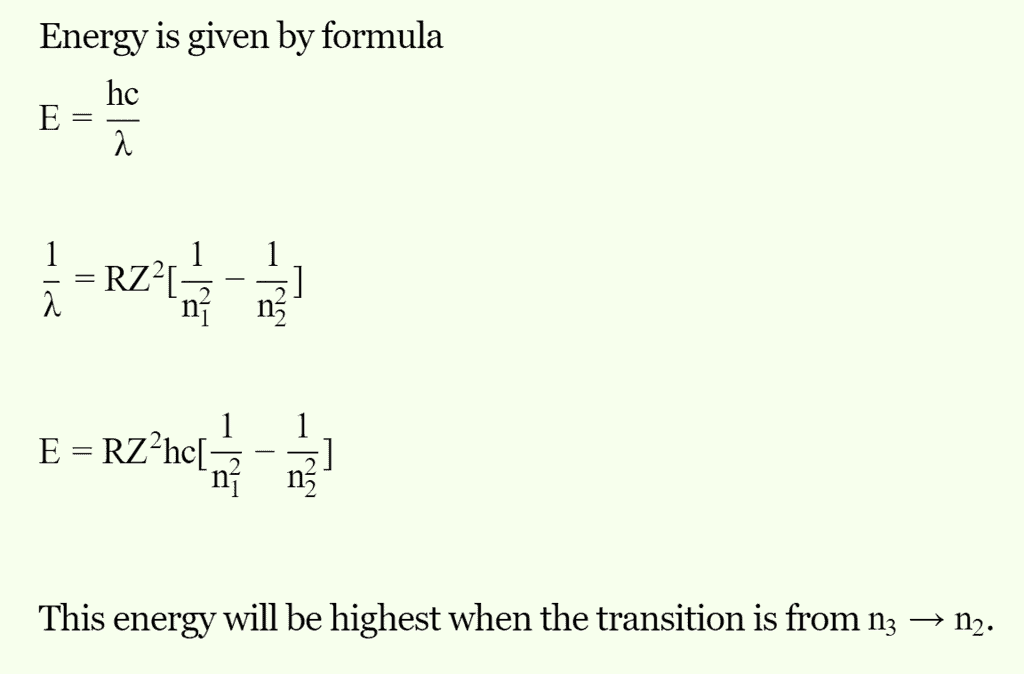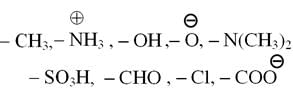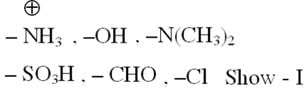JEE Main Chemistry Test- 1 Free Online Test 2026
MCQ Practice Test & Solutions: JEE Main Chemistry Test- 1 (25 Questions)
You can prepare effectively for JEE Mock Tests for JEE Main and Advanced 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "JEE Main Chemistry Test- 1". These 25 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
20 g of an ideal gas contains only atoms of S and O occupies 5.6 L at NPT. What is the mol. wt. of gas ?
Detailed Solution: Question 1
5.6 litres of a gas at N.T.P. are found to have a mass of 11 g. The molecular mass of the gas is
Detailed Solution: Question 2
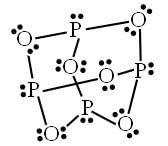
A crystalline salt Na2SO4.xH2O on heating losses 55.9 per cent of its mass. The formula of crystalline salt is---
Detailed Solution: Question 3
Vapour density of a metal chloride is 66. Its oxide contains 53% metal. The atomic weight of the metal is
Detailed Solution: Question 4
When two aqueous solutions react together to give off solid product, this reaction is known as
Detailed Solution: Question 5
At S.T.P. the density of CCl4 vapour in g/L will be nearest to
Detailed Solution: Question 6
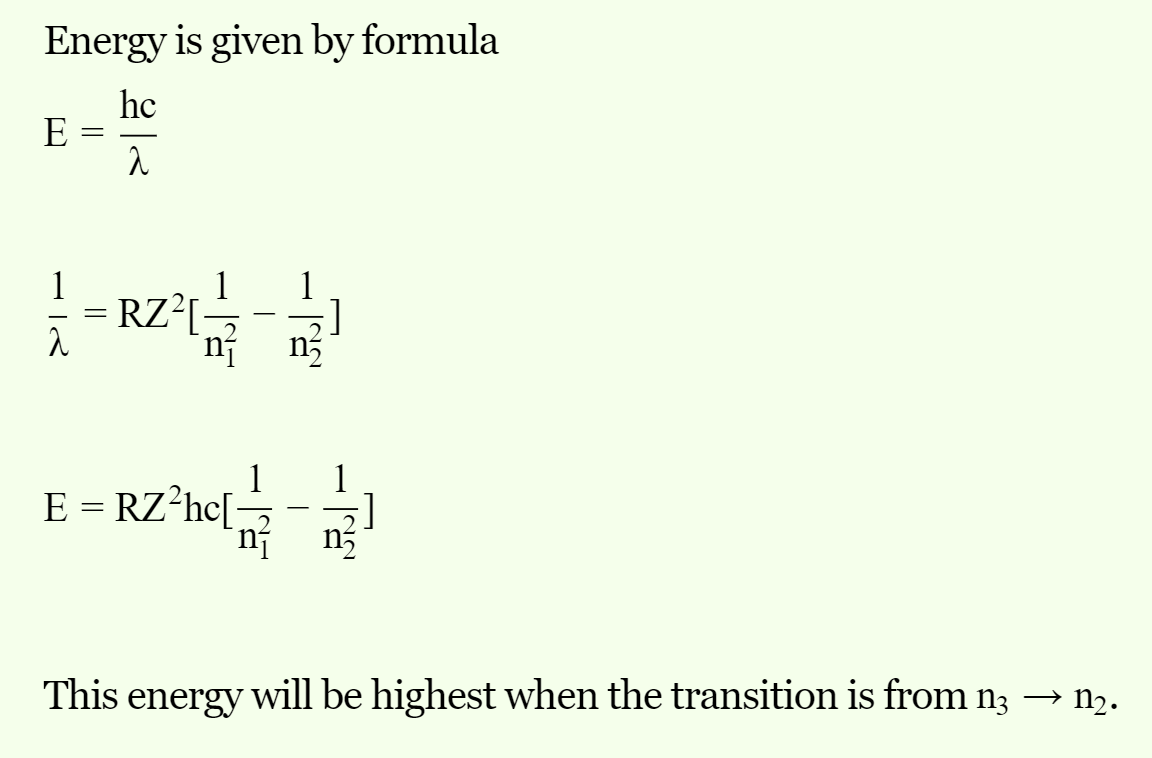
Which of the following transitions of electrons in the hydrogen atom will emit maximum energy?
Detailed Solution: Question 7
The uncertainty found from the uncertainty principle (Δx.Δp = h/4 π) is
Detailed Solution: Question 8
Detailed Solution: Question 9
The spectrum of He is expected to be similar to that of
Detailed Solution: Question 10
The electrons present in K-shell of the atom will differ in
Detailed Solution: Question 11
Which of the following statements about quantum numbers is wrong?
Detailed Solution: Question 12
Out of the two compounds shown below, the vapour pressing of B at a particular temperature is expected to be
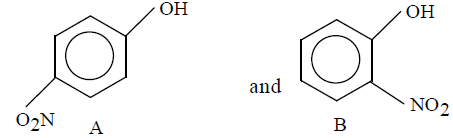
Detailed Solution: Question 13
In which of the following molecules, the central atom does not use sP3-hybrid orbitals in its bonding ?
Detailed Solution: Question 14
Detailed Solution: Question 15
Detailed Solution: Question 16
Detailed Solution: Question 17
From which of the following species it is easiest to remove one electron ?
Detailed Solution: Question 18
Detailed Solution: Question 19
Which element has the greatest tendency to lose electrons ?
Detailed Solution: Question 20
0.01 mole of iodoform (CHI3) reacts with Ag to produce a gas whose volume at NTP in ml is :-
2CHI3 + 6Ag —→ 6AgI(s) + C2H2(g)
2CHI3 + 6Ag —→ 6AgI(s) + C2H2(g)
Detailed Solution: Question 21
Detailed Solution: Question 22
How many maximum electrons can be accomodate in 7th shell of element X?
Detailed Solution: Question 23
Detailed Solution: Question 24
Detailed Solution: Question 25
360 docs|100 tests |



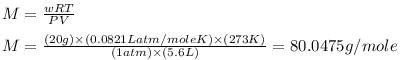 Therefore, the molar mass of the gas is, 80.0475 g/mole
Therefore, the molar mass of the gas is, 80.0475 g/mole