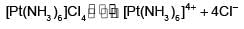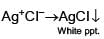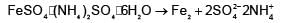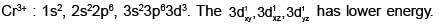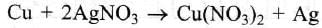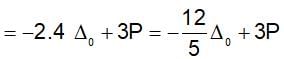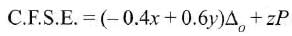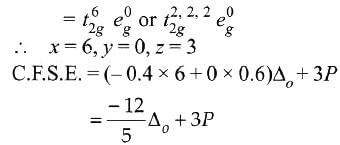Coordination Compounds - Free MCQ Test with solutions for Class 12
MCQ Practice Test & Solutions: Coordination Compounds (41 Questions)
You can prepare effectively for Class 12 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Coordination Compounds ". These 41 questions have been designed by the experts with the latest curriculum of Class 12 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 45 minutes
- - Number of Questions: 41
Sign up on EduRev for free to attempt this test and track your preparation progress.
The EAN of platinum in potassium hexachloroplatinate (IV) is:
Detailed Solution: Question 1
Detailed Solution: Question 2
Detailed Solution: Question 3
Detailed Solution: Question 4
Which will give a white precipitate with AgNO3 in aqueous solution?
Detailed Solution: Question 5
Detailed Solution: Question 6
The number of ions formed on dissolving one molecule of FeSO4.(NH4)2SO4.6H2O in water is:
Detailed Solution: Question 7
Detailed Solution: Question 8
Detailed Solution: Question 9
Which possesses tetrahedral shape (sp3 - hybridization of central atom?
Detailed Solution: Question 10
Detailed Solution: Question 11
Which complex has square planar shape & dsp2- hybridization?
Detailed Solution: Question 12
Detailed Solution: Question 13
The number of precipitable halide ions in [Pt(NH3)CI2Br]CI is:
Detailed Solution: Question 14
Which statement about coordination number of metal ion is true?
Detailed Solution: Question 15
The correct name of the compound [Cu(NH3)4](NO3)2, according to IUPAC system is:
Detailed Solution: Question 16
The formula of a carbonyl complex of (CO)n CO(CO)n, in which there is a single covalent Co-Co bond is?
Detailed Solution: Question 17
Of the following complexes, the one with the largest value of the crystal field splitting is:
Detailed Solution: Question 18
Detailed Solution: Question 19
In which complex is the transition metal in zero oxidation state?
Detailed Solution: Question 20
Detailed Solution: Question 21
From the stability constant (hypothetical values) given below, predict which is the strongest ligand?
Detailed Solution: Question 22
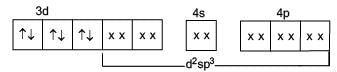
Which of the following complex species do not involve d2sp3-hybridization?
Detailed Solution: Question 23
Which of the following compounds would exhibit coordination isomerism?
Detailed Solution: Question 24
Detailed Solution: Question 25
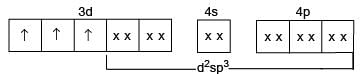
[Cr(H2O6]CI3 (at. no. of Cr=24) has a magnetic moment of 3.83 BM. The correct distribution of 3d-electrons in the chromium of the complex:
Detailed Solution: Question 26
The hypothetical complex chloro diaquatriammine cobalt(III) chloride can be represented as:
Detailed Solution: Question 27
In the silver plating of Cu, K[Ag(CN)2] is used instead of AgNO3. The reason is:
Detailed Solution: Question 28
Detailed Solution: Question 29
Low spin complex of d6-cation in an octahedral field will have the following energy:
Detailed Solution: Question 30