Conformational Analysis - 1 - Free MCQ Practice Test with solutions, NEET
MCQ Practice Test & Solutions: Test: Conformational Analysis - 1 (10 Questions)
You can prepare effectively for NEET Chemistry Class 11 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Conformational Analysis - 1". These 10 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 15 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
Direction (Q. Nos. 1-10) This section contains 10 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
The molecular formula C5H12 contains how many isomeric alkanes?
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Detailed Solution: Question 1
Which of the following cycloalkanes exhibits the greatest molar heat of combustion per —CH2 — group?
Detailed Solution: Question 3
Which of the following correctly ranks the cycloalkanes in order of increasing ring strain per methylene group?
Detailed Solution: Question 4
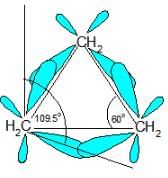
Which,of the following correctly lists the conformations of cyclohexane in order of increasing potential energies?
Detailed Solution: Question 5
Which is not true regarding conformers of ethane?
Detailed Solution: Question 6
The correct statement concerning conformers of 1,2-dichloroethane is
Detailed Solution: Question 7
Which of the following molecule exhibits conformational isomerism ?
Detailed Solution: Question 8
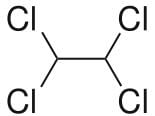
Which is true about conformers of 1,1,2,2-tetrachloroethane?
Which is true about conformers of 1,1,2,2-tetrachloroethane?
a) The most stable conformer has dihedral angle of 60° between all adjacent chlorine atoms
b) In the least stable conformer, two Cl-atoms are eclipsing one another while other two Cl-atoms are eclipsed to hydrogen atoms
c) In the most stable conformer, dihedral angle between hydrogen atoms is 60°
d) The most stable conformer is non-polar
a) The most stable conformer has dihedral angle of 60° between all adjacent chlorine atoms
b) In the least stable conformer, two Cl-atoms are eclipsing one another while other two Cl-atoms are eclipsed to hydrogen atoms
c) In the most stable conformer, dihedral angle between hydrogen atoms is 60°
d) The most stable conformer is non-polar
Detailed Solution: Question 9
What is relationship between the following Fischer Projections?
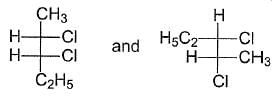
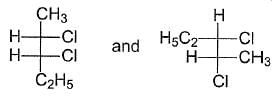
Detailed Solution: Question 10
98 videos|243 docs|71 tests |