VITEEE PCBE Mock Test - 1 - JEE MCQ
30 Questions MCQ Test VITEEE: Subject Wise and Full Length MOCK Tests - VITEEE PCBE Mock Test - 1
Which of the following is an aquatic plant that does not use water for pollination?
During the propagation of a nerve impulse, the action potential results from the movement of
Which of the following are the two key concepts of Darwinian theory of evolution?
The cytoplasmic connections from cell to cell are known as
Which of the following is correct sequence of process of recombinant DNA technology-
i. Isolation of DNA
ii. Isolation of desired DNA fragment.
iii. Fragmentation of DNA by restriction endonuclease.
iv. Transferring of into host
v. Ligation of DNA fragment into vector
vi. Culturing in host cell to get desired product.
Which of the following is NOT a method by which CO₂ is transported in the blood?
Rate of breathing is controlled mainly by ________________.
Which one of the following represents the correct dimensions of the coefficient of viscosity?
The physical quantity having the dimensional formula [M-1L-3T3A2] is
In an experiment to measure the internal resistance of a cell by potentiometer, it is found that the balance point is at a length of 2 m when the cell is shunted by a 4 Ω resistance, and at 3 m when the cell is shunted by an 8 Ω resistance. The internal resistance of the cell is:
An aqueous solution of an ionic salt having molality equal to 0.1892 freezes at - 0.704°C. What is the van't Hoff factor of the salt? (Given Kf for water = 1.86 K/m)
On adding a few drops of dilute HCl to a freshly precipitated ferric hydroxide, a red-coloured colloidal solution is obtained. This phenomenon is called
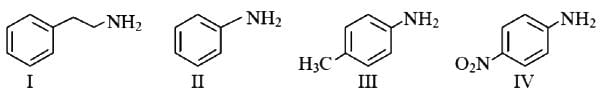
The correct order of basicity of the following amines is
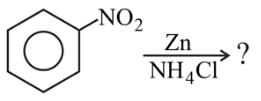
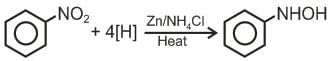
Directions: In the following question, two statements are given. One is Assertion and the other is Reason. Examine the statements carefully and mark the correct answer according to the instructions given below.
Assertion: Glycerol is purified by distillation under reduced pressure.
Reason: Glycerol is a trihydric alcohol.
Directions: In the following question, two statements are given. One is assertion and the other is reason. Examine the statements carefully and mark the correct answer accordingly.
Assertion: If β4 for [Cu(NH3)4]2+ is 2.1 x 1013, its instability constant is 4.76 x 10–14.
Reason: Overall dissociation equilibrium constant varies inversely with formation constant.
Among the following the one that gives positive iodoform test upon reaction with I2 and NaOH is
What is the author's opinion about the competition for customers among micro-financiers?
The sum of three consecutive odd numbers is 1383. What is the largest number?
|
1 videos|7 docs|63 tests
|
|
1 videos|7 docs|63 tests
|



 = [ML-1T-1]
= [ML-1T-1]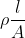
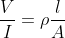
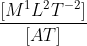 = [M1L2T-3A-1]
= [M1L2T-3A-1]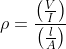 =
= 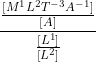 = [M1L3T-3A-2]
= [M1L3T-3A-2] is [M-1L-3T3A2]
is [M-1L-3T3A2]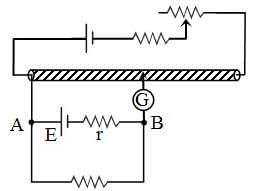
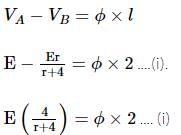
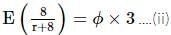
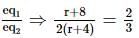
 =
= 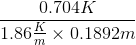 = 2
= 2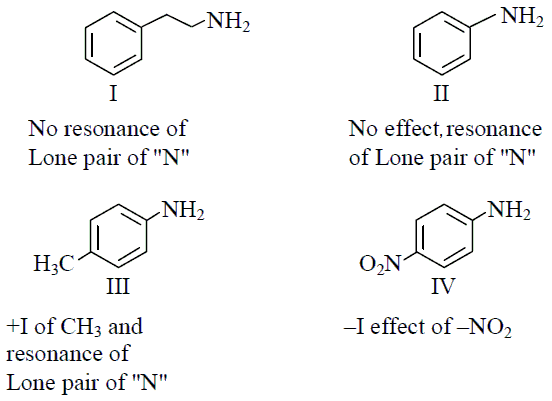
 = 2.1 x 1013
= 2.1 x 1013