VITEEE PCBE Mock Test - 2 - JEE MCQ
30 Questions MCQ Test VITEEE: Subject Wise and Full Length MOCK Tests - VITEEE PCBE Mock Test - 2
Cardiac Muscle Tissue What unique structural feature of cardiac muscle tissue enhances its functionality in the heart?
How do bacteriophages differ from most plant-infecting viruses?
Assertion (A): Slime molds form fruiting bodies that can disperse spores.
Reason (R): The spores of slime molds are capable of surviving extreme conditions.
Reason (R): The spores of slime molds are capable of surviving extreme conditions.
The given figure shows an angiogram of the coronary blood vessel. Which one of the following statements correctly describes, what is being done?
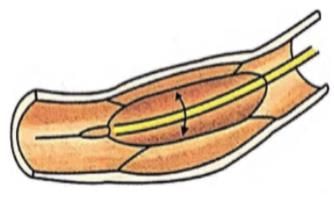
Read the following statements about the vascular bundles:
(a) In roots, xylem and phloem in a vascular bundle are arranged in an alternate manner along the different radii.
(b) Conjoint closed vascular bundles do not possess cambium
(c) In open vascular bundles, cambium is present in between xylem and phloem
(d) The vascular bundles of dicotyledonous stem possess endarch protoxylem
(e) In monocotyledonous root, usually there are more than six xylem bundles present
Choose the correct answer from the options given below :
Which of the following statements are correct?
(A). Depolarization of an axonal membrane is caused due to rise in stimulus-induced permeability to Na⁺ and its rapid influx into axoplasm.
(B). Diffusion of K⁺ outside the axonal membrane restores the resting potential of the membrane.
(C). Sodium-potassium pump maintains active transport of 2 Na⁺ outwards for 3 K⁺ into the axoplasm across the resting membrane.
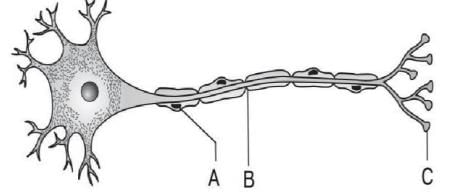
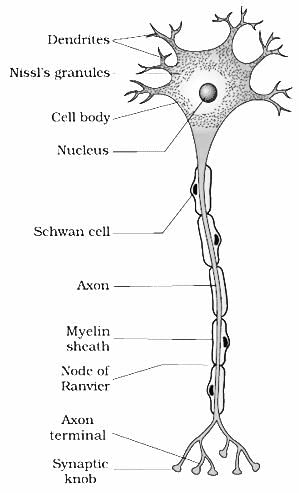
Refer to the given diagram of the structure of a neuron and identify A, B and C.
Which organelle is involved in the formation of cilia and flagella in animals?
The most common fungal partners of mycorrhiza are _________ species.
A proton moving with a speed u along the positive x-axis enters at y = 0, a region of uniform magnetic field B = B0 which exists to the right of y-axis as shown in the figure. The proton leaves the region after some time with a speed v at coordinate y. Then,
A current of 2 A flows through a system of resistors, as shown in the given figure. What is the value of 'V'?
A magnet of length 10 cm and magnetic moment 1 Am2 is placed along the side AB of an equilateral triangle ABC. If the length of side AB is 10 cm, then the magnetic field at point C is
For CE transistor amplifier, the audio signal voltage across the collector resistance of 2 kΩ is 4 V. If the current amplification factor of the transistor is 100 and the base resistance is 1 kΩ, then the input signal voltage is
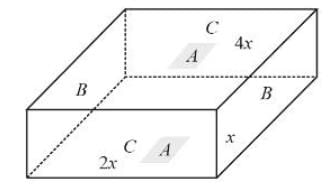
The following figure shows a rectangular block with dimensions x, 2x, and 4x. Electrical contacts can be made to the block between opposite pairs of faces (for example, between the faces labelled A−A, B−B, and C−C). Between which two faces would the maximum electrical resistance be obtained (A−A: Top and bottom faces, B−B: Left and right faces, and C−C: Front and rear faces)?
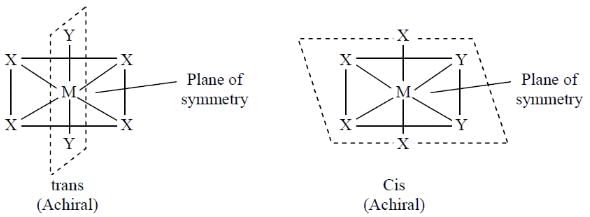
For an octahedral complex MX4Y2 (M = a transition metal, X and Y are monodentate achiral ligands), the correct statement, among the following, is
An amine (X) reacts with benzenesulphonyl chloride and the product thus obtained is soluble in KOH.
The amine (X) is
The pair of products formed during the reaction of yellow phosphorous with aqueous potash solution is
Among the second period elements, the actual first ionisation enthalpy are in order of? (Select the correct option from the following.)
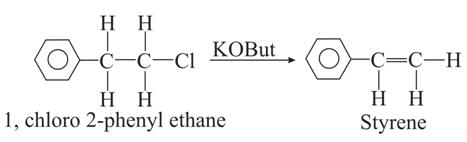
The polymer obtained by addition polymerisation of [x]. Which can be obtained by reaction between 1-chloro-2-phenyl ethane and potassium tertiary butoxide ?
Choose the word/group of words which is the most similar in meaning to the word/group of words printed in underline as used in the passage.
Pounding
Choose the word/group of words which is most opposite in meaning to the word/group of words printed in underline as used in the passage.
Coarse
Abhijit purchased a TV set for ₹18000 and a DVD player for ₹4000. He sold both the items together for ₹26400
₹26400. How much percent profit did he make?
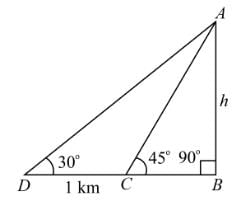
A person walking along a straight road observes a pole from two points that are 1 km (1000 m) apart. The angles of elevation of the pole from these two points are 30° and 45°. The height of the pole is:
20 women can do a piece of work in 16 days, while 16 men can complete the same work in 15 days. What is the ratio of the efficiency of a man to that of a woman?
|
1 videos|7 docs|63 tests
|
|
1 videos|7 docs|63 tests
|



 B); where q is the charge of the proton. The force F is perpendicular to both u and B. Since the force is perpendicular to the velocity of the particle, it does not do any work. Hence, the magnitude of the velocity of the particle will remain unchanged; only the direction of the velocity changes. Hence, v = u. Since u is perpendicular to B, the proton moves in a circular path. Since the charge of proton is positive, u is along positive x-axis and B is directed out of the page; the proton will move in a circle in the x-y plane in the clockwise direction. Hence, its y-coordinate will be negative, when it leaves the region. Thus, the correct choice is (4).
B); where q is the charge of the proton. The force F is perpendicular to both u and B. Since the force is perpendicular to the velocity of the particle, it does not do any work. Hence, the magnitude of the velocity of the particle will remain unchanged; only the direction of the velocity changes. Hence, v = u. Since u is perpendicular to B, the proton moves in a circular path. Since the charge of proton is positive, u is along positive x-axis and B is directed out of the page; the proton will move in a circle in the x-y plane in the clockwise direction. Hence, its y-coordinate will be negative, when it leaves the region. Thus, the correct choice is (4).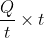 = Q. Hence, ampere-hour is the unit of electric charge.
= Q. Hence, ampere-hour is the unit of electric charge.
 volts = 3 volts
volts = 3 volts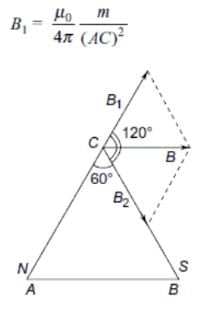

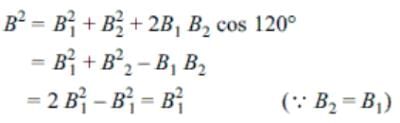


 = 2 mA
= 2 mA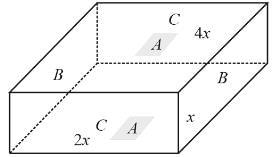
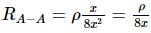
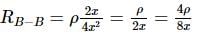
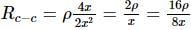
 Fe(SCN)3
Fe(SCN)3 

 43 = 28 +3ab × 4
43 = 28 +3ab × 4 64 = 28 + 12ab
64 = 28 + 12ab  64 - 28 = 12ab
64 - 28 = 12ab 36 = 12ab
36 = 12ab ab = 3
ab = 3















