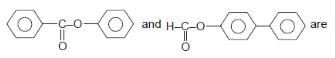Test: General Organic Chemistry - NEET MCQ
20 Questions MCQ Test NCERT Based Tests for NEET - Test: General Organic Chemistry
Which of the following structures is the correct structure of 3-ethyl-5, 5-diisopropyl-7- methylnonane
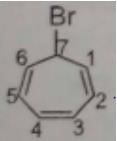
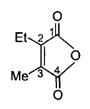
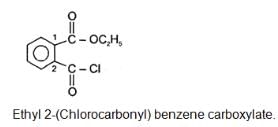
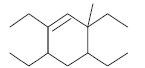
The correct IUPAC name of the folllowing compound is
A hydrocarbon (R) has six membered ring in which there is no unsaturation. Two alkyl groups are atttached to the ring adjacent to each other. One group has 3 carbon atoms with branching at 1st carbon atom of chain and another has 4 carbon atoms. The larger alkyl group has main chain of three carbon atoms of which second carbon is substituted. Correct IUPAC name of compound (R) is
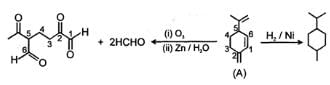
In the given sequence reaction which of the following is the correct structure of compounds A.
For the following reactions sequence
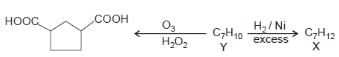
The structure consistent with X and Y are:
In which reaction a chiral reactant is giving a chiral product
Which of the following statements is true about the following conformer (Y)?
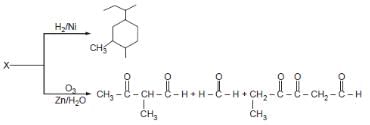
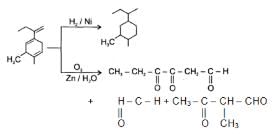
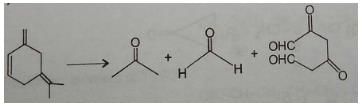
An unsaturated hydrocarbon on jcomplete hydrogenation gives 1-isopropyl-3 methylcyclohexane, after ozonolysis it gives one mole of formaldehyde, one mole of acetone and one mole of 2,4-Dioxohexanedial. The possible structures of the hydrocarbon maybe
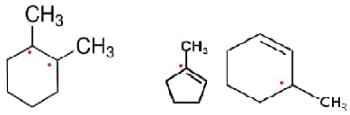
How many assymmetric carbon atoms are present in
(i) 2-Dimethyl cyclohexane
(ii) 3-Methyl cyclopentene
(iii) 3-Methylcyclohexene
Which of the following statements is not correct?
Identify the pair of enantiomers amongst the given pairs:
|
684 tests
|