NEET Exam > NEET Tests > NCERTs at Fingertips: Textbooks, Tests & Solutions > Test: Hydrogen Bonding (NCERT) - NEET MCQ
Test: Hydrogen Bonding (NCERT) - NEET MCQ
Test Description
10 Questions MCQ Test NCERTs at Fingertips: Textbooks, Tests & Solutions - Test: Hydrogen Bonding (NCERT)
Test: Hydrogen Bonding (NCERT) for NEET 2025 is part of NCERTs at Fingertips: Textbooks, Tests & Solutions preparation. The Test: Hydrogen Bonding (NCERT) questions and answers have been
prepared according to the NEET exam syllabus.The Test: Hydrogen Bonding (NCERT) MCQs are made for NEET 2025 Exam. Find important
definitions, questions, notes, meanings, examples, exercises, MCQs and online tests for Test: Hydrogen Bonding (NCERT) below.
Solutions of Test: Hydrogen Bonding (NCERT) questions in English are available as part of our NCERTs at Fingertips: Textbooks, Tests & Solutions for NEET & Test: Hydrogen Bonding (NCERT) solutions in
Hindi for NCERTs at Fingertips: Textbooks, Tests & Solutions course. Download more important topics, notes, lectures and mock
test series for NEET Exam by signing up for free. Attempt Test: Hydrogen Bonding (NCERT) | 10 questions in 10 minutes | Mock test for NEET preparation | Free important questions MCQ to study NCERTs at Fingertips: Textbooks, Tests & Solutions for NEET Exam | Download free PDF with solutions
Test: Hydrogen Bonding (NCERT) - Question 1
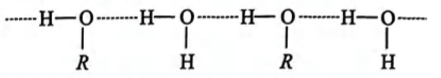
Which of the following compounds shows maximum hydrogen bonding?
Detailed Solution for Test: Hydrogen Bonding (NCERT) - Question 1
Detailed Solution for Test: Hydrogen Bonding (NCERT) - Question 2
Test: Hydrogen Bonding (NCERT) - Question 3
Which of the following statements is true about hydrogen bonding?
Detailed Solution for Test: Hydrogen Bonding (NCERT) - Question 3
Detailed Solution for Test: Hydrogen Bonding (NCERT) - Question 4
Test: Hydrogen Bonding (NCERT) - Question 5
Which of the following observations can be explained on the basis of hydrogen bonding?
(i) H—F has higher boiling point than other halogen acids.
(ii) H2O has highest boiling point among hydrides of group 16 elements.
(iii) NH3 has lower boiling point than PH3.
Detailed Solution for Test: Hydrogen Bonding (NCERT) - Question 5
Detailed Solution for Test: Hydrogen Bonding (NCERT) - Question 6
Test: Hydrogen Bonding (NCERT) - Question 7
Though covalent in nature, methanol is soluble in water, why?
Detailed Solution for Test: Hydrogen Bonding (NCERT) - Question 7
Detailed Solution for Test: Hydrogen Bonding (NCERT) - Question 8
Test: Hydrogen Bonding (NCERT) - Question 9
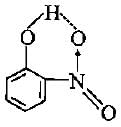
Which of the following molecules shows intramolecular hydrogen bonding?
Detailed Solution for Test: Hydrogen Bonding (NCERT) - Question 9
Detailed Solution for Test: Hydrogen Bonding (NCERT) - Question 10
|
237 docs|243 tests
|
Information about Test: Hydrogen Bonding (NCERT) Page
In this test you can find the Exam questions for Test: Hydrogen Bonding (NCERT) solved & explained in the simplest way possible.
Besides giving Questions and answers for Test: Hydrogen Bonding (NCERT), EduRev gives you an ample number of Online tests for practice