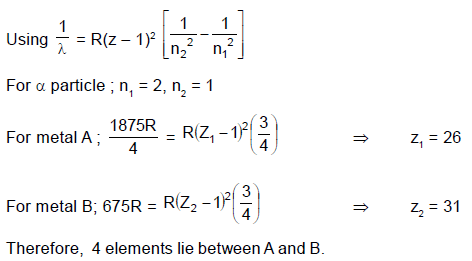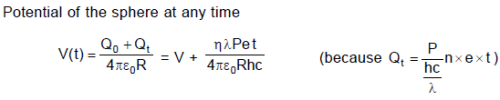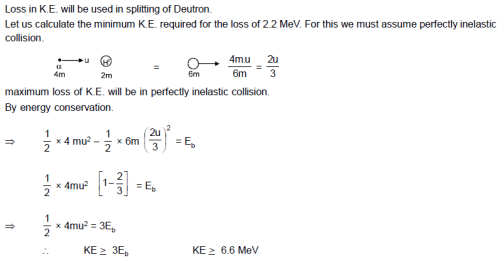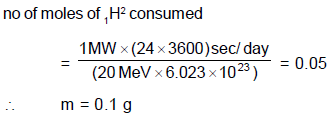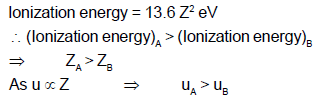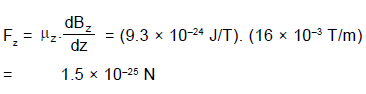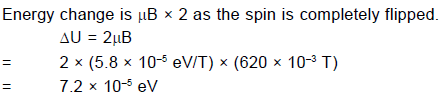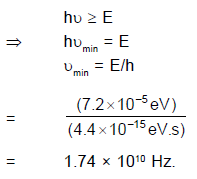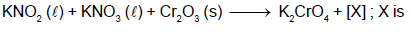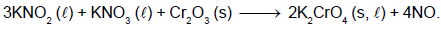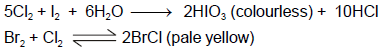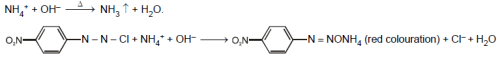JEE Advanced Test- 6 - JEE MCQ
30 Questions MCQ Test Mock Tests for JEE Main and Advanced 2025 - JEE Advanced Test- 6
The wavelengths of Kα x-rays of two metals ‘A’ and ‘B’ are 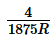 and
and 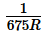 respectively, where ‘R’ is
respectively, where ‘R’ is
Rydberg constant. The number of elements lying between ‘A’ and ‘B’ according to their atomic numbers is
Rydberg constant. The number of elements lying between ‘A’ and ‘B’ according to their atomic numbers is
A free neutron decays to a proton but a free proton does not decay to a neutron out side nucleus. This is beacuse
An α a particle with a kinetic energy of 2.1 eV makes a head on collision with a hydrogen atom moving towards it with a kinetic energy of 8.4 eV. The collision.
A monochromatic radiation of wavelength λ is incident on a sample containing He+. As a result the Helium sample starts radiating. A part of this radiation is allowed to pass through a sample of atomic hydrogen gas in ground state. It is noticed that the hydrogen sample has started emitting electrons whose maximum Kinetic Energy is 37.4 eV. (hc = 12400 eV Å) Then λ is -
The work function of a certain metal is When a monochromatic light of wavelength λ < λ0 is
incident such that the plate gains a total power P. If the efficiency of photoelectric emission is η%
and all the emitted photoelectrons are captured by a hollow conducting sphere of radius R already
charged to potential V, then neglecting any interaction between plate and the sphere, expression of
potantial of the sphere at time t is :
The Kinetic energy must an α-particle possess to split a deutron H2 whose binding energy is Eb = 2.2 MeV -
Assuming that about 20 MeV of energy is released per fusion reaction, 1H2 + 1H3 → 0n1 + 2He4, the mass of 1H2 consumed per day in a future fusion reactor of power 1 MW would be approximately
Nuclei of radioactive element A are produced at rate ' t2 ' at any time t. The element A has decay constant λ. Let N be the number of nuclei of element A at any time t. At time t = t0, is minimum. Then the number of nuclei of element A at time t = t0 is
If nuclear charge is doubled in a hydrogen like atom, which of the following statement(s) are consistent with Bohr’s theory?
Ionization energy of a hydrogen-like ion B is less than that of hydrogen like ion A. Let r, u, E and L represent the radius of the orbit, speed of the electron, energy of the atom and orbital angular momentum of the electron respectively. In ground state
An electron makes a transition from n = 2 to n = 1 state in a hydrogen like atom.
Statement–1 : undergoes 2α decays, 2β decays (negative β) and 2 γ decays. As a result the
daughter product is .
Statement–2 : In α decay the mass number decreases by 4 unit and atomic number decreases by 2 unit.
In β decay (negative β) the mass number remains unchanged and atomic number increases by 1 unit. In
γ decay, mass number and atomic number remains unchanged.
Statement–1 : In the duration electron jumps from first excited state to ground state in a stationary isolated hydrogen atom, angular momentum of the electron about the nucleus is conserved.
Statement–2 : As the electron jumps from first excited state to ground state, in a hydrogen atom, the electrostatic force on electron is always directed towards the nucleus.
The orbital and spin angular momentum of the atom influence its magnetic structure and these properties are most directly studied by placing the atom in a magnetic field. Also, a magnetic field can affect the wavelengths of the emitted photons.
The angular momentum vector associated with an atomic state can take up only certain specified directions in space. This concept of space quantization was shown by Otto Stern and Walthor Gerlach in their experiment.
In the experiment, silver is vapourized in an electric oven and silver atoms spray into the evacuated apparatus through a small hole in the oven wall. The atoms which are electrically neutral but have a magnetic moment, are formed into a narrow beam as they pass through a slit in a screen. The beam, thus collimated, then passes between the poles of an electromagnet and finally, deposits its silver atoms on a glass plate that serves as a detector. The pole faces of the magnet are shaped to make the magnetic field as nonuniform as possible.
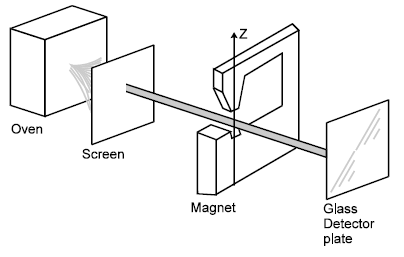
In a non-uniform magnetic field, there is a net force on a magnetic dipole. Its magnitude and direction depends on the orientation of the dipole. Thus the silver atoms in the beam are deflected up or down, depending on the orientation of their magnetic dipole moments with respect to the z–direction.
The potential energy of a magnetic dipole in a magnetic field where
is magnetic dipole moment of the atom. From symmetry, the magnetic field at the beam position has no x or y components i.e.
The net force Fz on the dipole is
Thus, the net force depends, not on the magnitude of the field itself, but on its spatial derivative or gradient.
The Results
If space quantization did not exist, then could take on any value from +
to –
, the result would be a spreading out of the beam when the magnet was turned ON. However, the beam was split cleanly into two subbeams, each subbeam corresponding to one of the two permitted orientations of the magnetic moment of the silver atom, as shown.
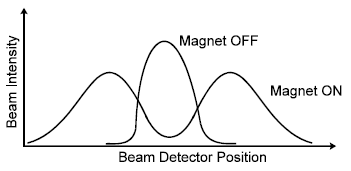
In a silver atom, all the spin and orbital magnetic moments of the electrons cancel, except for those of the atom's single valance electron. For this electron the orbital magnetic moment is zero because orbital angular momentum is zero (because for electrons of s–orbit, L = 0), leaving only the spin magnetic moment. This can take up only two orientations in a magnetic field, corresponding to ms = +1/2 and ms = – 1/2. Hence there are two subbeams – and not some other number.
Q. A hydrogen atom in ground state passes through a magnetic field that has a gradient of 16mT/m in the vertical direction. If vertical component magnetic moment of the atom is 9.3 × 10–24 J/T, then force on it due to the magnetic moment of the electron is :
The orbital and spin angular momentum of the atom influence its magnetic structure and these properties are most directly studied by placing the atom in a magnetic field. Also, a magnetic field can affect the wavelengths of the emitted photons.
The angular momentum vector associated with an atomic state can take up only certain specified directions in space. This concept of space quantization was shown by Otto Stern and Walthor Gerlach in their experiment.
In the experiment, silver is vapourized in an electric oven and silver atoms spray into the evacuated apparatus through a small hole in the oven wall. The atoms which are electrically neutral but have a magnetic moment, are formed into a narrow beam as they pass through a slit in a screen. The beam, thus collimated, then passes between the poles of an electromagnet and finally, deposits its silver atoms on a glass plate that serves as a detector. The pole faces of the magnet are shaped to make the magnetic field as nonuniform as possible.

In a non-uniform magnetic field, there is a net force on a magnetic dipole. Its magnitude and direction depends on the orientation of the dipole. Thus the silver atoms in the beam are deflected up or down, depending on the orientation of their magnetic dipole moments with respect to the z–direction.
The potential energy of a magnetic dipole in a magnetic field where
is magnetic dipole moment of the atom. From symmetry, the magnetic field at the beam position has no x or y components i.e.
The net force Fz on the dipole is
Thus, the net force depends, not on the magnitude of the field itself, but on its spatial derivative or gradient.
The Results
If space quantization did not exist, then could take on any value from +
to –
, the result would be a spreading out of the beam when the magnet was turned ON. However, the beam was split cleanly into two subbeams, each subbeam corresponding to one of the two permitted orientations of the magnetic moment of the silver atom, as shown.

In a silver atom, all the spin and orbital magnetic moments of the electrons cancel, except for those of the atom's single valance electron. For this electron the orbital magnetic moment is zero because orbital angular momentum is zero (because for electrons of s–orbit, L = 0), leaving only the spin magnetic moment. This can take up only two orientations in a magnetic field, corresponding to ms = +1/2 and ms = – 1/2. Hence there are two subbeams – and not some other number.
Q. A hydrogen atom in ground state passes through a magnetic field that has a gradient of 16mT/m in the vertical direction. If vertical component magnetic moment of the atom is 9.3 × 10–24 J/T, then force on it due to the magnetic moment of the electron is :
The orbital and spin angular momentum of the atom influence its magnetic structure and these properties are most directly studied by placing the atom in a magnetic field. Also, a magnetic field can affect the wavelengths of the emitted photons.
The angular momentum vector associated with an atomic state can take up only certain specified directions in space. This concept of space quantization was shown by Otto Stern and Walthor Gerlach in their experiment.
In the experiment, silver is vapourized in an electric oven and silver atoms spray into the evacuated apparatus through a small hole in the oven wall. The atoms which are electrically neutral but have a magnetic moment, are formed into a narrow beam as they pass through a slit in a screen. The beam, thus collimated, then passes between the poles of an electromagnet and finally, deposits its silver atoms on a glass plate that serves as a detector. The pole faces of the magnet are shaped to make the magnetic field as nonuniform as possible.

In a non-uniform magnetic field, there is a net force on a magnetic dipole. Its magnitude and direction depends on the orientation of the dipole. Thus the silver atoms in the beam are deflected up or down, depending on the orientation of their magnetic dipole moments with respect to the z–direction.
The potential energy of a magnetic dipole in a magnetic field where
is magnetic dipole moment of the atom. From symmetry, the magnetic field at the beam position has no x or y components i.e.
The net force Fz on the dipole is
Thus, the net force depends, not on the magnitude of the field itself, but on its spatial derivative or gradient.
The Results
If space quantization did not exist, then could take on any value from +
to –
, the result would be a spreading out of the beam when the magnet was turned ON. However, the beam was split cleanly into two subbeams, each subbeam corresponding to one of the two permitted orientations of the magnetic moment of the silver atom, as shown.

In a silver atom, all the spin and orbital magnetic moments of the electrons cancel, except for those of the atom's single valance electron. For this electron the orbital magnetic moment is zero because orbital angular momentum is zero (because for electrons of s–orbit, L = 0), leaving only the spin magnetic moment. This can take up only two orientations in a magnetic field, corresponding to ms = +1/2 and ms = – 1/2. Hence there are two subbeams – and not some other number.
Q. A hydrogen atom in ground state passes through a magnetic field that has a gradient of 16mT/m in the vertical direction. If vertical component magnetic moment of the atom is 9.3 × 10–24 J/T, then force on it due to the magnetic moment of the electron is :
In column-I, consider each process just before and just after it occurs. Initial system is isolated from all other bodies. Consider all product particles (even those having rest mass zero) in the system. Match the system in column-I with the result they produce in column-II.

A radioactive sample has 12.0 × 1018 active nuclei at a certain instant. Number of nuclei still in the same active state after two half-lives is n ×1018. Find n.
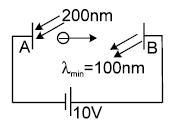
In the figure shown electromagnetic radiations of wavelength 200nm are incident on the metallic plate A. The photo electrons are accelerated by a potential difference 10V. These electrons strike another metal plate B from which electromagnetic radiations are emitted. The minimum wavelength of the emitted photons is 100nm. The work function of the metal ‘A’ is x eV then find x + 2.2 use hc = 12400 eVÅ, use Rch = 13.6 eV.
An X-ray tube is working at potential of 20 kV. The potential difference is decreased to 10 kV. It is found that
the difference of the wavelength of Kα X-ray and the most energetic continuous X-ray becomes 4 times the
difference before the change of voltage. Find the atomic number of the target element. Take b = 1 and =0.54.
Which of the following statement is correct with respect to the metal carbonyls of Ist transition series?
Concentrated sulphuric acid is added followed by heating of each of the following test tubes labelled (i) to (v).

Which of the following statement is incorrect about these observations ?
The following flow diagram represents the extraction of magnesium from sea water.
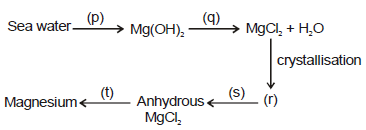
Which of the following options describes the correct reactants, products and reaction conditions ?
When CS2 layer containing both Br2 and I2 (2 : 1) is shaken with excess of Cl2 water, the violet colour due to I2 disappears and a pale yellow colour appears in the solution. The disappearance of violet colour and appearance of pale yellow colour is due to the formation of :
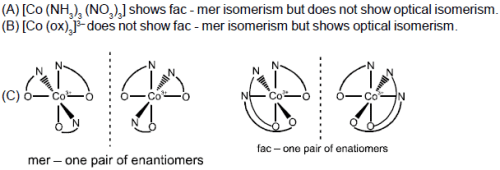
Which of the following has both face-mer and optical isomers ?
An inorganic chloride (X) on heating with a sodium hydroxide solution liberates a colourless and non-inflammable gas having characterisitic odour. The salt (X) gives a red colouration with 4-nitrobenzene-diazonium chloride in the presence of sodium hydroxide solution. X is :
A metal nitrate (X) gives a white precipitate with ammonia solution but the precipitate gets dissolved on adding ammonium salts. Lead dioxide and concentrated nitric acid on boiling with a dilute solution of metal nitrate (X) produces a violet-red (or purple) colour solution. Small amount of metal nitrate (X) gives a amethyst-red bead with borax in oxidising flame when cold. The cation of metal nitrate is :
|
356 docs|142 tests
|