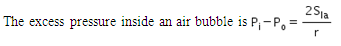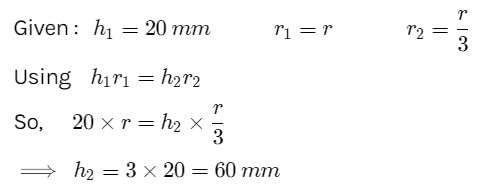Test: Surface Tension & Capillarity - NEET MCQ
10 Questions MCQ Test Physics Class 11 - Test: Surface Tension & Capillarity
The angle of contact for liquid on a solid surface is the angle between:
When impurity is added to a liquid, its surface tension
If drops and bubbles do not collapse under the effect of gravity, it indicates that
By which phenomenon does the water rise from roots to leaves of plants?
When a capillary tube of radius r is dipped in a liquid of density ρ and surface tension S, the liquid rises or falls through a distance
When an air bubble of radius R lies at a depth h below the free surface of a liquid of density ρ and surface tension Sla, then the excess pressure inside the bubble will be
Water rises to a height of 20 mm in a capillary. If the radius of the capillary is made 1/3 rd of its previous value, to what height will the water now rise in the tube?
The excess pressure inside a soap bubble is (Here, Sla is the surface tension between the liquid-air interface).
|
97 videos|379 docs|103 tests
|