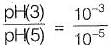Civil Engineering (CE) Exam > Civil Engineering (CE) Tests > Test: Quality & Characteristics of Sewage - 1 - Civil Engineering (CE) MCQ
Test: Quality & Characteristics of Sewage - 1 - Civil Engineering (CE) MCQ
Test Description
10 Questions MCQ Test - Test: Quality & Characteristics of Sewage - 1
Test: Quality & Characteristics of Sewage - 1 for Civil Engineering (CE) 2025 is part of Civil Engineering (CE) preparation. The Test: Quality & Characteristics of Sewage - 1 questions and answers have been prepared
according to the Civil Engineering (CE) exam syllabus.The Test: Quality & Characteristics of Sewage - 1 MCQs are made for Civil Engineering (CE) 2025 Exam.
Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests for Test: Quality & Characteristics of Sewage - 1 below.
Solutions of Test: Quality & Characteristics of Sewage - 1 questions in English are available as part of our course for Civil Engineering (CE) & Test: Quality & Characteristics of Sewage - 1 solutions in
Hindi for Civil Engineering (CE) course.
Download more important topics, notes, lectures and mock test series for Civil Engineering (CE) Exam by signing up for free. Attempt Test: Quality & Characteristics of Sewage - 1 | 10 questions in 30 minutes | Mock test for Civil Engineering (CE) preparation | Free important questions MCQ to study for Civil Engineering (CE) Exam | Download free PDF with solutions
Test: Quality & Characteristics of Sewage - 1 - Question 3
Gases, which are generally evolved during aerobic decomposition of sewage, are:
Detailed Solution for Test: Quality & Characteristics of Sewage - 1 - Question 3
Test: Quality & Characteristics of Sewage - 1 - Question 4
Imhoff cone is used to measure, in sewage:
Detailed Solution for Test: Quality & Characteristics of Sewage - 1 - Question 4
Detailed Solution for Test: Quality & Characteristics of Sewage - 1 - Question 5
Test: Quality & Characteristics of Sewage - 1 - Question 6
pH = 3, when compared to pH = 5, will be more acidic by
Detailed Solution for Test: Quality & Characteristics of Sewage - 1 - Question 6
Test: Quality & Characteristics of Sewage - 1 - Question 7
Chemical oxygen Demand (COD) of sewage is the
Detailed Solution for Test: Quality & Characteristics of Sewage - 1 - Question 7
Test: Quality & Characteristics of Sewage - 1 - Question 8
Minimum D.O. prescribed for a river stream, to avoid fish kills, is
Detailed Solution for Test: Quality & Characteristics of Sewage - 1 - Question 8
Test: Quality & Characteristics of Sewage - 1 - Question 9
BOD5 represents 5 days-biochemical oxygen demand at a temperature of
Test: Quality & Characteristics of Sewage - 1 - Question 10
Standard 5 day BOD at 20°C, when compared to ultimate BOD, is about
Detailed Solution for Test: Quality & Characteristics of Sewage - 1 - Question 10
Information about Test: Quality & Characteristics of Sewage - 1 Page
In this test you can find the Exam questions for Test: Quality & Characteristics of Sewage - 1 solved & explained in the simplest way possible.
Besides giving Questions and answers for Test: Quality & Characteristics of Sewage - 1, EduRev gives you an ample number of Online tests for practice
Download as PDF