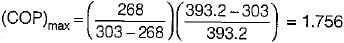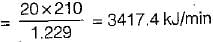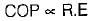Test: Refrigerants - 3 - Mechanical Engineering MCQ
15 Questions MCQ Test Thermodynamics - Test: Refrigerants - 3
The significant advantage of using Ammonia as a refrigerant is its.
The recommended material for use with ammonia as a refrigerants is
Which one eco-friendly refrigerant out of the following is used in many domestic and car air- conditioners now-a-days?
The use of refrigerant R-22 for temperatures below -30°C is not recommended due to its
Hydrogen is used in electrolux refrigeration system so that vapour pressure of ammonia in the evaporator should be
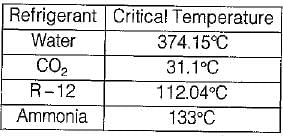
Which one of the following refrigerants has the highest critical temperature?
In an absorption type refrigerator, the heat is supplied to NH3 generator by condensing steam at 2 bar and 90% dry. The temperature in the refrigerator is to be maintained at - 5°C. If the refrigeration load is 20 tones and actual COP is 70% of the maximum then mass of steam required per hour is
(Take atmospheric temperature as 30°C saturation temperature of steam at pressure of 2 bar is 120.2°C, hfg at 2 bar is 2201.6 kJ/kg)
Match List-I (Refrigerant) with List-Il (Application) and select the correct answer using the codes given below the lists:
List-l List-II
A. R-11 1. Window type air conditioning system
B. R-12 2. Central air-conditioning system
C. R-22 3. Domestic refrigerator
D. NH3 4. Cold storages, ice plants
Codes:
A B C D
(a) 2 1 3 4
(b) 4 3 1 2
(c) 2 3 1 4
(d) 4 1 3 2
Match List-I (Property of refrigerant or absorbent) with List-II (Name of refrigerant) and select the correct answer using the code given below the lists
List-I List-II
A. Used for very low temperature 1. CF4
B. Used with centrifugal compressors 2. CCI3F
C. Used in vortex tube 3. Air
Codes: A B C
(a) 1 2 3
(b) 3 1 2
(c) 1 3 2
(d) 3 2 1
When the mixture of refrigerant is used to replace a refrigerant, if the COP increase, the reason attributed is
Match List-l with List-ll and select the correct answer using the codes given below the lists:
List-l List-ll
A. Sulphur candle test 1. Propane
B. Halide torch test 2. Ammonia
C. Soap and water test 3. Halocarbon refrigerants
D. Ammonia Swab test 4. Sulphur dioxide
Codes:
A B C D
(a) 2 3 1 4
(b) 4 1 3 2
(c) 2 1 3 4
(d) 4 3 1 2
Consider the following statements:
1. Azeotropes are the mixtures of refrigerants and behave like pure substances.
2. Isomers refrigerants are compounds with the same chemical formula but have different molecular structures.
3. The formula n + p + q = 2m is used for unsaturated chloro-fluorocarbon compounds (m, n, p and q are the number of atoms of carbon, hydrogen, fluorine and chlorine respectively.)
Q. Which of these statements are correct?
|
29 videos|66 docs|36 tests
|