Class 9 Exam > Class 9 Questions > you are provided with a mixture of nephthalen... Start Learning for Free
you are provided with a mixture of nephthalene and ammonium chloride by your teacher. suggest an activity to separate them with well labelled diagram.
?Most Upvoted Answer
you are provided with a mixture of nephthalene and ammonium chloride b...
Community Answer
you are provided with a mixture of nephthalene and ammonium chloride b...
Activity to Separate Naphthalene and Ammonium Chloride
Materials Required:
- Mixture of Naphthalene and Ammonium Chloride
- Beaker
- Funnel
- Filter Paper
- Bunsen Burner
- Tripod Stand
- Wire Gauze
- Glass Rod
Procedure:
1. Take the mixture of naphthalene and ammonium chloride in a beaker.
2. Heat the beaker with the help of a Bunsen Burner until all the naphthalene sublimes and forms vapors.
3. Place a funnel with a filter paper over another beaker.
4. The vapors of naphthalene will rise and get condensed on the funnel and filter paper.
5. The ammonium chloride remains in the original beaker.
6. After the sublimation of naphthalene is complete, turn off the Bunsen Burner and let the apparatus cool down.
7. Collect the naphthalene from the filter paper and store it in a clean and dry container.
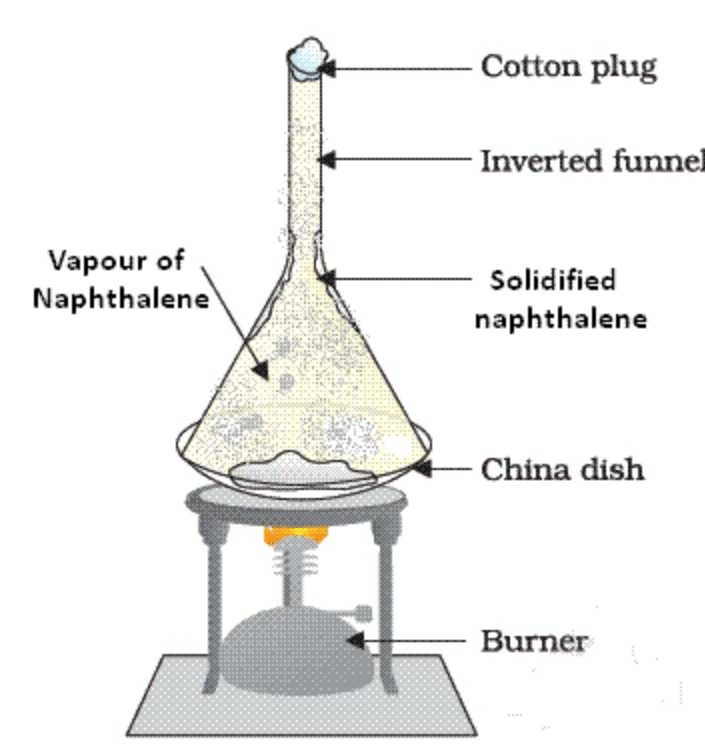
Diagram:

Explanation:
This activity is based on the principle of sublimation. Naphthalene, being a solid, sublimes on heating, i.e., it converts directly from solid to vapor state without melting. On the other hand, ammonium chloride remains in the beaker as it does not undergo sublimation on heating. The naphthalene vapors condense on the funnel and filter paper due to the difference in their boiling points. This process of separating a mixture of two or more substances by using their differences in volatilities is called sublimation.
Materials Required:
- Mixture of Naphthalene and Ammonium Chloride
- Beaker
- Funnel
- Filter Paper
- Bunsen Burner
- Tripod Stand
- Wire Gauze
- Glass Rod
Procedure:
1. Take the mixture of naphthalene and ammonium chloride in a beaker.
2. Heat the beaker with the help of a Bunsen Burner until all the naphthalene sublimes and forms vapors.
3. Place a funnel with a filter paper over another beaker.
4. The vapors of naphthalene will rise and get condensed on the funnel and filter paper.
5. The ammonium chloride remains in the original beaker.
6. After the sublimation of naphthalene is complete, turn off the Bunsen Burner and let the apparatus cool down.
7. Collect the naphthalene from the filter paper and store it in a clean and dry container.
Diagram:

Explanation:
This activity is based on the principle of sublimation. Naphthalene, being a solid, sublimes on heating, i.e., it converts directly from solid to vapor state without melting. On the other hand, ammonium chloride remains in the beaker as it does not undergo sublimation on heating. The naphthalene vapors condense on the funnel and filter paper due to the difference in their boiling points. This process of separating a mixture of two or more substances by using their differences in volatilities is called sublimation.
 | Explore Courses for Class 9 exam |  |
Question Description
you are provided with a mixture of nephthalene and ammonium chloride by your teacher. suggest an activity to separate them with well labelled diagram. Related: Short Answer Questions(2) : Matter in Our Surroundings? for Class 9 2026 is part of Class 9 preparation. The Question and answers have been prepared according to the Class 9 exam syllabus. Information about you are provided with a mixture of nephthalene and ammonium chloride by your teacher. suggest an activity to separate them with well labelled diagram. Related: Short Answer Questions(2) : Matter in Our Surroundings? covers all topics & solutions for Class 9 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for you are provided with a mixture of nephthalene and ammonium chloride by your teacher. suggest an activity to separate them with well labelled diagram. Related: Short Answer Questions(2) : Matter in Our Surroundings?.
you are provided with a mixture of nephthalene and ammonium chloride by your teacher. suggest an activity to separate them with well labelled diagram. Related: Short Answer Questions(2) : Matter in Our Surroundings? for Class 9 2026 is part of Class 9 preparation. The Question and answers have been prepared according to the Class 9 exam syllabus. Information about you are provided with a mixture of nephthalene and ammonium chloride by your teacher. suggest an activity to separate them with well labelled diagram. Related: Short Answer Questions(2) : Matter in Our Surroundings? covers all topics & solutions for Class 9 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for you are provided with a mixture of nephthalene and ammonium chloride by your teacher. suggest an activity to separate them with well labelled diagram. Related: Short Answer Questions(2) : Matter in Our Surroundings?.
Solutions for you are provided with a mixture of nephthalene and ammonium chloride by your teacher. suggest an activity to separate them with well labelled diagram. Related: Short Answer Questions(2) : Matter in Our Surroundings? in English & in Hindi are available as part of our courses for Class 9. Download more important topics, notes, lectures and mock test series for Class 9 Exam by signing up for free.
Here you can find the meaning of you are provided with a mixture of nephthalene and ammonium chloride by your teacher. suggest an activity to separate them with well labelled diagram. Related: Short Answer Questions(2) : Matter in Our Surroundings? defined & explained in the simplest way possible. Besides giving the explanation of you are provided with a mixture of nephthalene and ammonium chloride by your teacher. suggest an activity to separate them with well labelled diagram. Related: Short Answer Questions(2) : Matter in Our Surroundings?, a detailed solution for you are provided with a mixture of nephthalene and ammonium chloride by your teacher. suggest an activity to separate them with well labelled diagram. Related: Short Answer Questions(2) : Matter in Our Surroundings? has been provided alongside types of you are provided with a mixture of nephthalene and ammonium chloride by your teacher. suggest an activity to separate them with well labelled diagram. Related: Short Answer Questions(2) : Matter in Our Surroundings? theory, EduRev gives you an ample number of questions to practice you are provided with a mixture of nephthalene and ammonium chloride by your teacher. suggest an activity to separate them with well labelled diagram. Related: Short Answer Questions(2) : Matter in Our Surroundings? tests, examples and also practice Class 9 tests.
 | Explore Courses for Class 9 exam |  |
Top Courses for Class 9
Explore CoursesSignup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.






