Class 12 Exam > Class 12 Questions > Aspirin is an acetylation product of [1998]a)... Start Learning for Free
Aspirin is an acetylation product of [1998]
- a)o-hydroxybenzoic acid
- b)o-dihydroxybenzene
- c)m-hydroxybenzoic acid
- d)p-dihydroxybenzene
Correct answer is option 'A'. Can you explain this answer?
Verified Answer
Aspirin is an acetylation product of [1998]a)o-hydroxybenzoic acidb)o-...
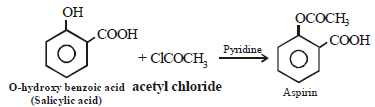
Most Upvoted Answer
Aspirin is an acetylation product of [1998]a)o-hydroxybenzoic acidb)o-...
Introduction:
Aspirin, also known as acetylsalicylic acid, is a commonly used medication for pain relief, fever reduction, and inflammation reduction. It is a derivative of salicylic acid and is synthesized through acetylation, which involves introducing an acetyl group (-COCH3) into the salicylic acid molecule. In this case, aspirin is the acetylation product of o-hydroxybenzoic acid.
Explanation:
To understand why the correct answer is option 'A' (o-hydroxybenzoic acid), let's break down the process of aspirin synthesis through acetylation.
Step 1: Starting Compound - Salicylic Acid
The starting compound for the synthesis of aspirin is salicylic acid. Salicylic acid is a white, crystalline solid that naturally occurs in the bark of willow trees and other plants. It has analgesic (pain-relieving), antipyretic (fever-reducing), and anti-inflammatory properties.
Step 2: Acetic Anhydride
Acetic anhydride is the acetylating agent used in the synthesis of aspirin. It is a colorless liquid with a strong, pungent odor. Acetic anhydride reacts with salicylic acid to introduce an acetyl group (-COCH3) into the molecule.
Step 3: Acetylation Reaction
The acetylation reaction involves the reaction between salicylic acid and acetic anhydride in the presence of a catalyst, such as sulfuric acid or phosphoric acid. During this reaction, the hydrogen atom (-H) on the hydroxyl group (OH) of salicylic acid is replaced by the acetyl group (-COCH3) from acetic anhydride.
Step 4: Formation of Aspirin
The product of the acetylation reaction is aspirin, also known as acetylsalicylic acid. Aspirin is a white, crystalline solid with a bitter taste. It is slightly soluble in water but readily dissolves in organic solvents.
Correct Answer - o-Hydroxybenzoic Acid
The correct answer is option 'A' (o-hydroxybenzoic acid). The structure of o-hydroxybenzoic acid is shown below:
H
|
OH
|
C6H4COOH
When acetic anhydride reacts with o-hydroxybenzoic acid, the acetyl group (-COCH3) replaces the hydrogen atom (-H) on the hydroxyl group (OH) of o-hydroxybenzoic acid, resulting in the formation of aspirin. Therefore, o-hydroxybenzoic acid is the precursor for the synthesis of aspirin through acetylation.
Aspirin, also known as acetylsalicylic acid, is a commonly used medication for pain relief, fever reduction, and inflammation reduction. It is a derivative of salicylic acid and is synthesized through acetylation, which involves introducing an acetyl group (-COCH3) into the salicylic acid molecule. In this case, aspirin is the acetylation product of o-hydroxybenzoic acid.
Explanation:
To understand why the correct answer is option 'A' (o-hydroxybenzoic acid), let's break down the process of aspirin synthesis through acetylation.
Step 1: Starting Compound - Salicylic Acid
The starting compound for the synthesis of aspirin is salicylic acid. Salicylic acid is a white, crystalline solid that naturally occurs in the bark of willow trees and other plants. It has analgesic (pain-relieving), antipyretic (fever-reducing), and anti-inflammatory properties.
Step 2: Acetic Anhydride
Acetic anhydride is the acetylating agent used in the synthesis of aspirin. It is a colorless liquid with a strong, pungent odor. Acetic anhydride reacts with salicylic acid to introduce an acetyl group (-COCH3) into the molecule.
Step 3: Acetylation Reaction
The acetylation reaction involves the reaction between salicylic acid and acetic anhydride in the presence of a catalyst, such as sulfuric acid or phosphoric acid. During this reaction, the hydrogen atom (-H) on the hydroxyl group (OH) of salicylic acid is replaced by the acetyl group (-COCH3) from acetic anhydride.
Step 4: Formation of Aspirin
The product of the acetylation reaction is aspirin, also known as acetylsalicylic acid. Aspirin is a white, crystalline solid with a bitter taste. It is slightly soluble in water but readily dissolves in organic solvents.
Correct Answer - o-Hydroxybenzoic Acid
The correct answer is option 'A' (o-hydroxybenzoic acid). The structure of o-hydroxybenzoic acid is shown below:
H
|
OH
|
C6H4COOH
When acetic anhydride reacts with o-hydroxybenzoic acid, the acetyl group (-COCH3) replaces the hydrogen atom (-H) on the hydroxyl group (OH) of o-hydroxybenzoic acid, resulting in the formation of aspirin. Therefore, o-hydroxybenzoic acid is the precursor for the synthesis of aspirin through acetylation.
 | Explore Courses for Class 12 exam |  |
Question Description
Aspirin is an acetylation product of [1998]a)o-hydroxybenzoic acidb)o-dihydroxybenzenec)m-hydroxybenzoic acidd)p-dihydroxybenzeneCorrect answer is option 'A'. Can you explain this answer? for Class 12 2026 is part of Class 12 preparation. The Question and answers have been prepared according to the Class 12 exam syllabus. Information about Aspirin is an acetylation product of [1998]a)o-hydroxybenzoic acidb)o-dihydroxybenzenec)m-hydroxybenzoic acidd)p-dihydroxybenzeneCorrect answer is option 'A'. Can you explain this answer? covers all topics & solutions for Class 12 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Aspirin is an acetylation product of [1998]a)o-hydroxybenzoic acidb)o-dihydroxybenzenec)m-hydroxybenzoic acidd)p-dihydroxybenzeneCorrect answer is option 'A'. Can you explain this answer?.
Aspirin is an acetylation product of [1998]a)o-hydroxybenzoic acidb)o-dihydroxybenzenec)m-hydroxybenzoic acidd)p-dihydroxybenzeneCorrect answer is option 'A'. Can you explain this answer? for Class 12 2026 is part of Class 12 preparation. The Question and answers have been prepared according to the Class 12 exam syllabus. Information about Aspirin is an acetylation product of [1998]a)o-hydroxybenzoic acidb)o-dihydroxybenzenec)m-hydroxybenzoic acidd)p-dihydroxybenzeneCorrect answer is option 'A'. Can you explain this answer? covers all topics & solutions for Class 12 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Aspirin is an acetylation product of [1998]a)o-hydroxybenzoic acidb)o-dihydroxybenzenec)m-hydroxybenzoic acidd)p-dihydroxybenzeneCorrect answer is option 'A'. Can you explain this answer?.
Solutions for Aspirin is an acetylation product of [1998]a)o-hydroxybenzoic acidb)o-dihydroxybenzenec)m-hydroxybenzoic acidd)p-dihydroxybenzeneCorrect answer is option 'A'. Can you explain this answer? in English & in Hindi are available as part of our courses for Class 12. Download more important topics, notes, lectures and mock test series for Class 12 Exam by signing up for free.
Here you can find the meaning of Aspirin is an acetylation product of [1998]a)o-hydroxybenzoic acidb)o-dihydroxybenzenec)m-hydroxybenzoic acidd)p-dihydroxybenzeneCorrect answer is option 'A'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of Aspirin is an acetylation product of [1998]a)o-hydroxybenzoic acidb)o-dihydroxybenzenec)m-hydroxybenzoic acidd)p-dihydroxybenzeneCorrect answer is option 'A'. Can you explain this answer?, a detailed solution for Aspirin is an acetylation product of [1998]a)o-hydroxybenzoic acidb)o-dihydroxybenzenec)m-hydroxybenzoic acidd)p-dihydroxybenzeneCorrect answer is option 'A'. Can you explain this answer? has been provided alongside types of Aspirin is an acetylation product of [1998]a)o-hydroxybenzoic acidb)o-dihydroxybenzenec)m-hydroxybenzoic acidd)p-dihydroxybenzeneCorrect answer is option 'A'. Can you explain this answer? theory, EduRev gives you an ample number of questions to practice Aspirin is an acetylation product of [1998]a)o-hydroxybenzoic acidb)o-dihydroxybenzenec)m-hydroxybenzoic acidd)p-dihydroxybenzeneCorrect answer is option 'A'. Can you explain this answer? tests, examples and also practice Class 12 tests.
 | Explore Courses for Class 12 exam |  |
Top Courses for Class 12
Explore CoursesSignup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.






