Class 12 Exam > Class 12 Questions > C and Si both have same lattice structure, ha... Start Learning for Free
C and Si both have same lattice structure, having 4 bonding electrons in each. However, C isinsulator whereas Si is intrinsic semiconductor.This is because : [2012]
- a)In case of C the valence band is notcompletely filled at absolute zerotemperature.
- b)In case of C the conduction band is partlyfilled even at absolute zero temperature.
- c)The four bonding electrons in the case of Clie in the second orbit, whereas in the caseof Si they lie in the third.
- d)The four bonding electrons in the case of Clie in the third orbit, whereas for Si they liein the fourth orbit.
Correct answer is option 'C'. Can you explain this answer?
Verified Answer
C and Si both have same lattice structure, having 4 bonding electrons ...
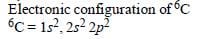
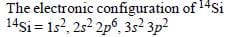
As they are away from Nucleus, so effect
of nucleus is low for Si even for Sn and Pb
are almost mettalic.
of nucleus is low for Si even for Sn and Pb
are almost mettalic.
Most Upvoted Answer
C and Si both have same lattice structure, having 4 bonding electrons ...
Explanation:
C and Si have the same lattice structure, but their electrical properties are different. The reason for this difference lies in the electronic configuration of the two elements.
Valence electrons
Both C and Si have 4 valence electrons, which are involved in bonding. The electronic configuration of C is 1s2 2s2 2p2, while that of Si is 1s2 2s2 2p6 3s2 3p2. This means that the valence electrons of C lie in the second orbit, whereas those of Si lie in the third orbit.
Bonding
The valence electrons of C and Si are involved in covalent bonding, which means that they share electrons with their neighboring atoms. In the case of C, each atom forms 4 covalent bonds, resulting in a very stable structure. However, this stable structure also means that the valence band is not completely filled at absolute zero temperature. Therefore, C is an insulator.
Intrinsic semiconductor
On the other hand, Si is an intrinsic semiconductor because its valence band is completely filled at absolute zero temperature, but there is a small energy gap between the valence band and the conduction band. This energy gap is small enough that thermal energy can cause electrons to jump from the valence band to the conduction band, allowing the material to conduct electricity. This property of semiconductors is utilized in many electronic devices.
Conclusion
In summary, the difference in electronic configuration between C and Si leads to differences in their electrical properties. While C is an insulator due to the incomplete filling of its valence band, Si is an intrinsic semiconductor due to the small energy gap between its valence and conduction bands.
C and Si have the same lattice structure, but their electrical properties are different. The reason for this difference lies in the electronic configuration of the two elements.
Valence electrons
Both C and Si have 4 valence electrons, which are involved in bonding. The electronic configuration of C is 1s2 2s2 2p2, while that of Si is 1s2 2s2 2p6 3s2 3p2. This means that the valence electrons of C lie in the second orbit, whereas those of Si lie in the third orbit.
Bonding
The valence electrons of C and Si are involved in covalent bonding, which means that they share electrons with their neighboring atoms. In the case of C, each atom forms 4 covalent bonds, resulting in a very stable structure. However, this stable structure also means that the valence band is not completely filled at absolute zero temperature. Therefore, C is an insulator.
Intrinsic semiconductor
On the other hand, Si is an intrinsic semiconductor because its valence band is completely filled at absolute zero temperature, but there is a small energy gap between the valence band and the conduction band. This energy gap is small enough that thermal energy can cause electrons to jump from the valence band to the conduction band, allowing the material to conduct electricity. This property of semiconductors is utilized in many electronic devices.
Conclusion
In summary, the difference in electronic configuration between C and Si leads to differences in their electrical properties. While C is an insulator due to the incomplete filling of its valence band, Si is an intrinsic semiconductor due to the small energy gap between its valence and conduction bands.
 | Explore Courses for Class 12 exam |  |
Question Description
C and Si both have same lattice structure, having 4 bonding electrons in each. However, C isinsulator whereas Si is intrinsic semiconductor.This is because : [2012]a)In case of C the valence band is notcompletely filled at absolute zerotemperature.b)In case of C the conduction band is partlyfilled even at absolute zero temperature.c)The four bonding electrons in the case of Clie in the second orbit, whereas in the caseof Si they lie in the third.d)The four bonding electrons in the case of Clie in the third orbit, whereas for Si they liein the fourth orbit.Correct answer is option 'C'. Can you explain this answer? for Class 12 2026 is part of Class 12 preparation. The Question and answers have been prepared according to the Class 12 exam syllabus. Information about C and Si both have same lattice structure, having 4 bonding electrons in each. However, C isinsulator whereas Si is intrinsic semiconductor.This is because : [2012]a)In case of C the valence band is notcompletely filled at absolute zerotemperature.b)In case of C the conduction band is partlyfilled even at absolute zero temperature.c)The four bonding electrons in the case of Clie in the second orbit, whereas in the caseof Si they lie in the third.d)The four bonding electrons in the case of Clie in the third orbit, whereas for Si they liein the fourth orbit.Correct answer is option 'C'. Can you explain this answer? covers all topics & solutions for Class 12 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for C and Si both have same lattice structure, having 4 bonding electrons in each. However, C isinsulator whereas Si is intrinsic semiconductor.This is because : [2012]a)In case of C the valence band is notcompletely filled at absolute zerotemperature.b)In case of C the conduction band is partlyfilled even at absolute zero temperature.c)The four bonding electrons in the case of Clie in the second orbit, whereas in the caseof Si they lie in the third.d)The four bonding electrons in the case of Clie in the third orbit, whereas for Si they liein the fourth orbit.Correct answer is option 'C'. Can you explain this answer?.
C and Si both have same lattice structure, having 4 bonding electrons in each. However, C isinsulator whereas Si is intrinsic semiconductor.This is because : [2012]a)In case of C the valence band is notcompletely filled at absolute zerotemperature.b)In case of C the conduction band is partlyfilled even at absolute zero temperature.c)The four bonding electrons in the case of Clie in the second orbit, whereas in the caseof Si they lie in the third.d)The four bonding electrons in the case of Clie in the third orbit, whereas for Si they liein the fourth orbit.Correct answer is option 'C'. Can you explain this answer? for Class 12 2026 is part of Class 12 preparation. The Question and answers have been prepared according to the Class 12 exam syllabus. Information about C and Si both have same lattice structure, having 4 bonding electrons in each. However, C isinsulator whereas Si is intrinsic semiconductor.This is because : [2012]a)In case of C the valence band is notcompletely filled at absolute zerotemperature.b)In case of C the conduction band is partlyfilled even at absolute zero temperature.c)The four bonding electrons in the case of Clie in the second orbit, whereas in the caseof Si they lie in the third.d)The four bonding electrons in the case of Clie in the third orbit, whereas for Si they liein the fourth orbit.Correct answer is option 'C'. Can you explain this answer? covers all topics & solutions for Class 12 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for C and Si both have same lattice structure, having 4 bonding electrons in each. However, C isinsulator whereas Si is intrinsic semiconductor.This is because : [2012]a)In case of C the valence band is notcompletely filled at absolute zerotemperature.b)In case of C the conduction band is partlyfilled even at absolute zero temperature.c)The four bonding electrons in the case of Clie in the second orbit, whereas in the caseof Si they lie in the third.d)The four bonding electrons in the case of Clie in the third orbit, whereas for Si they liein the fourth orbit.Correct answer is option 'C'. Can you explain this answer?.
Solutions for C and Si both have same lattice structure, having 4 bonding electrons in each. However, C isinsulator whereas Si is intrinsic semiconductor.This is because : [2012]a)In case of C the valence band is notcompletely filled at absolute zerotemperature.b)In case of C the conduction band is partlyfilled even at absolute zero temperature.c)The four bonding electrons in the case of Clie in the second orbit, whereas in the caseof Si they lie in the third.d)The four bonding electrons in the case of Clie in the third orbit, whereas for Si they liein the fourth orbit.Correct answer is option 'C'. Can you explain this answer? in English & in Hindi are available as part of our courses for Class 12. Download more important topics, notes, lectures and mock test series for Class 12 Exam by signing up for free.
Here you can find the meaning of C and Si both have same lattice structure, having 4 bonding electrons in each. However, C isinsulator whereas Si is intrinsic semiconductor.This is because : [2012]a)In case of C the valence band is notcompletely filled at absolute zerotemperature.b)In case of C the conduction band is partlyfilled even at absolute zero temperature.c)The four bonding electrons in the case of Clie in the second orbit, whereas in the caseof Si they lie in the third.d)The four bonding electrons in the case of Clie in the third orbit, whereas for Si they liein the fourth orbit.Correct answer is option 'C'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of C and Si both have same lattice structure, having 4 bonding electrons in each. However, C isinsulator whereas Si is intrinsic semiconductor.This is because : [2012]a)In case of C the valence band is notcompletely filled at absolute zerotemperature.b)In case of C the conduction band is partlyfilled even at absolute zero temperature.c)The four bonding electrons in the case of Clie in the second orbit, whereas in the caseof Si they lie in the third.d)The four bonding electrons in the case of Clie in the third orbit, whereas for Si they liein the fourth orbit.Correct answer is option 'C'. Can you explain this answer?, a detailed solution for C and Si both have same lattice structure, having 4 bonding electrons in each. However, C isinsulator whereas Si is intrinsic semiconductor.This is because : [2012]a)In case of C the valence band is notcompletely filled at absolute zerotemperature.b)In case of C the conduction band is partlyfilled even at absolute zero temperature.c)The four bonding electrons in the case of Clie in the second orbit, whereas in the caseof Si they lie in the third.d)The four bonding electrons in the case of Clie in the third orbit, whereas for Si they liein the fourth orbit.Correct answer is option 'C'. Can you explain this answer? has been provided alongside types of C and Si both have same lattice structure, having 4 bonding electrons in each. However, C isinsulator whereas Si is intrinsic semiconductor.This is because : [2012]a)In case of C the valence band is notcompletely filled at absolute zerotemperature.b)In case of C the conduction band is partlyfilled even at absolute zero temperature.c)The four bonding electrons in the case of Clie in the second orbit, whereas in the caseof Si they lie in the third.d)The four bonding electrons in the case of Clie in the third orbit, whereas for Si they liein the fourth orbit.Correct answer is option 'C'. Can you explain this answer? theory, EduRev gives you an ample number of questions to practice C and Si both have same lattice structure, having 4 bonding electrons in each. However, C isinsulator whereas Si is intrinsic semiconductor.This is because : [2012]a)In case of C the valence band is notcompletely filled at absolute zerotemperature.b)In case of C the conduction band is partlyfilled even at absolute zero temperature.c)The four bonding electrons in the case of Clie in the second orbit, whereas in the caseof Si they lie in the third.d)The four bonding electrons in the case of Clie in the third orbit, whereas for Si they liein the fourth orbit.Correct answer is option 'C'. Can you explain this answer? tests, examples and also practice Class 12 tests.
 | Explore Courses for Class 12 exam |  |
Top Courses for Class 12
Explore CoursesSignup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.






