JEE Exam > JEE Questions > PASSAGE - 1The noble gases have closed-shell ...
Start Learning for Free
PASSAGE - 1
The noble gases have closed-shell electronic configuration and are monoatomic gases under normal conditions. The low boiling points of the lighter noble gases are due to weak dispersion forces between the atoms and the absence of other interatomic interactions.
The direct reaction of xenon with fluorine leads to a series of compounds with oxidation numbers +2, +4 and +6. XeF4 reacts violently with water to given XeO3. The compounds of xenon exhibit rich stereochemistry and their geometries can be deduced considering the total number of electron pairs in the valence shell.
Q. XeF4 and XeF6 are expected to be
- a)oxidizing
- b)reducing
- c)unreactive
- d)strongly basic
Correct answer is option 'A'. Can you explain this answer?
| FREE This question is part of | Download PDF Attempt this Test |
Verified Answer
PASSAGE - 1The noble gases have closed-shell electronic configuration ...
All xenon fluorides are strongly oxidizing, XeF4 can act as reducing agent (with F2) as well as oxidizing agent but XeF6 can only function as an oxidizing agent.
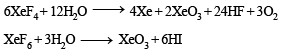
Most Upvoted Answer
PASSAGE - 1The noble gases have closed-shell electronic configuration ...
Answer:
The compounds XeF4 and XeF6 are expected to be oxidizing agents.
Explanation:
1. Noble Gases and their Electronic Configuration:
- Noble gases are a group of elements that have closed-shell electronic configurations, meaning that their outermost energy level is fully occupied with electrons.
- This configuration makes noble gases highly stable and unreactive under normal conditions.
2. Boiling Points of Noble Gases:
- The boiling points of noble gases increase with increasing atomic number.
- This is because the strength of the dispersion forces (also known as London forces or Van der Waals forces) increases with increasing molecular weight.
- Dispersion forces are the attractive forces between temporary dipoles that occur due to the constant motion of electrons.
- In the lighter noble gases, such as helium and neon, the dispersion forces are weak, resulting in low boiling points.
3. Xenon Compounds:
- Xenon (Xe) is a noble gas that can form compounds with other elements, particularly fluorine (F).
- The direct reaction of xenon with fluorine leads to the formation of compounds with oxidation numbers of 2, 4, and 6.
- XeF4 and XeF6 are examples of such compounds.
4. Oxidizing Agents:
- Oxidizing agents are substances that have a tendency to gain electrons or cause the oxidation of other substances.
- XeF4 and XeF6 are strong oxidizing agents because xenon has a high electronegativity and can readily accept electrons from other substances.
- In the case of XeF4, it reacts violently with water to give XeO3, indicating its ability to oxidize water.
Conclusion:
- Based on the above information, it can be concluded that XeF4 and XeF6 are expected to be oxidizing agents due to the high electronegativity of xenon and their ability to accept electrons from other substances.
The compounds XeF4 and XeF6 are expected to be oxidizing agents.
Explanation:
1. Noble Gases and their Electronic Configuration:
- Noble gases are a group of elements that have closed-shell electronic configurations, meaning that their outermost energy level is fully occupied with electrons.
- This configuration makes noble gases highly stable and unreactive under normal conditions.
2. Boiling Points of Noble Gases:
- The boiling points of noble gases increase with increasing atomic number.
- This is because the strength of the dispersion forces (also known as London forces or Van der Waals forces) increases with increasing molecular weight.
- Dispersion forces are the attractive forces between temporary dipoles that occur due to the constant motion of electrons.
- In the lighter noble gases, such as helium and neon, the dispersion forces are weak, resulting in low boiling points.
3. Xenon Compounds:
- Xenon (Xe) is a noble gas that can form compounds with other elements, particularly fluorine (F).
- The direct reaction of xenon with fluorine leads to the formation of compounds with oxidation numbers of 2, 4, and 6.
- XeF4 and XeF6 are examples of such compounds.
4. Oxidizing Agents:
- Oxidizing agents are substances that have a tendency to gain electrons or cause the oxidation of other substances.
- XeF4 and XeF6 are strong oxidizing agents because xenon has a high electronegativity and can readily accept electrons from other substances.
- In the case of XeF4, it reacts violently with water to give XeO3, indicating its ability to oxidize water.
Conclusion:
- Based on the above information, it can be concluded that XeF4 and XeF6 are expected to be oxidizing agents due to the high electronegativity of xenon and their ability to accept electrons from other substances.
Attention JEE Students!
To make sure you are not studying endlessly, EduRev has designed JEE study material, with Structured Courses, Videos, & Test Series. Plus get personalized analysis, doubt solving and improvement plans to achieve a great score in JEE.

|
Explore Courses for JEE exam
|

|
Similar JEE Doubts
PASSAGE - 1The noble gases have closed-shell electronic configuration and are monoatomic gases under normal conditions. The low boiling points of the lighter noble gases are due to weak dispersion forces between the atoms and the absence of other interatomic interactions.The direct reaction of xenon with fluorine leads to a series of compounds with oxidation numbers +2, +4 and +6. XeF4 reacts violently with water to given XeO3. The compounds of xenon exhibit rich stereochemistry and their geometries can be deduced considering the total number of electron pairs in the valence shell.Q.XeF4 and XeF6 are expected to bea)oxidizingb)reducingc)unreactived)strongly basicCorrect answer is option 'A'. Can you explain this answer?
Question Description
PASSAGE - 1The noble gases have closed-shell electronic configuration and are monoatomic gases under normal conditions. The low boiling points of the lighter noble gases are due to weak dispersion forces between the atoms and the absence of other interatomic interactions.The direct reaction of xenon with fluorine leads to a series of compounds with oxidation numbers +2, +4 and +6. XeF4 reacts violently with water to given XeO3. The compounds of xenon exhibit rich stereochemistry and their geometries can be deduced considering the total number of electron pairs in the valence shell.Q.XeF4 and XeF6 are expected to bea)oxidizingb)reducingc)unreactived)strongly basicCorrect answer is option 'A'. Can you explain this answer? for JEE 2024 is part of JEE preparation. The Question and answers have been prepared according to the JEE exam syllabus. Information about PASSAGE - 1The noble gases have closed-shell electronic configuration and are monoatomic gases under normal conditions. The low boiling points of the lighter noble gases are due to weak dispersion forces between the atoms and the absence of other interatomic interactions.The direct reaction of xenon with fluorine leads to a series of compounds with oxidation numbers +2, +4 and +6. XeF4 reacts violently with water to given XeO3. The compounds of xenon exhibit rich stereochemistry and their geometries can be deduced considering the total number of electron pairs in the valence shell.Q.XeF4 and XeF6 are expected to bea)oxidizingb)reducingc)unreactived)strongly basicCorrect answer is option 'A'. Can you explain this answer? covers all topics & solutions for JEE 2024 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for PASSAGE - 1The noble gases have closed-shell electronic configuration and are monoatomic gases under normal conditions. The low boiling points of the lighter noble gases are due to weak dispersion forces between the atoms and the absence of other interatomic interactions.The direct reaction of xenon with fluorine leads to a series of compounds with oxidation numbers +2, +4 and +6. XeF4 reacts violently with water to given XeO3. The compounds of xenon exhibit rich stereochemistry and their geometries can be deduced considering the total number of electron pairs in the valence shell.Q.XeF4 and XeF6 are expected to bea)oxidizingb)reducingc)unreactived)strongly basicCorrect answer is option 'A'. Can you explain this answer?.
PASSAGE - 1The noble gases have closed-shell electronic configuration and are monoatomic gases under normal conditions. The low boiling points of the lighter noble gases are due to weak dispersion forces between the atoms and the absence of other interatomic interactions.The direct reaction of xenon with fluorine leads to a series of compounds with oxidation numbers +2, +4 and +6. XeF4 reacts violently with water to given XeO3. The compounds of xenon exhibit rich stereochemistry and their geometries can be deduced considering the total number of electron pairs in the valence shell.Q.XeF4 and XeF6 are expected to bea)oxidizingb)reducingc)unreactived)strongly basicCorrect answer is option 'A'. Can you explain this answer? for JEE 2024 is part of JEE preparation. The Question and answers have been prepared according to the JEE exam syllabus. Information about PASSAGE - 1The noble gases have closed-shell electronic configuration and are monoatomic gases under normal conditions. The low boiling points of the lighter noble gases are due to weak dispersion forces between the atoms and the absence of other interatomic interactions.The direct reaction of xenon with fluorine leads to a series of compounds with oxidation numbers +2, +4 and +6. XeF4 reacts violently with water to given XeO3. The compounds of xenon exhibit rich stereochemistry and their geometries can be deduced considering the total number of electron pairs in the valence shell.Q.XeF4 and XeF6 are expected to bea)oxidizingb)reducingc)unreactived)strongly basicCorrect answer is option 'A'. Can you explain this answer? covers all topics & solutions for JEE 2024 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for PASSAGE - 1The noble gases have closed-shell electronic configuration and are monoatomic gases under normal conditions. The low boiling points of the lighter noble gases are due to weak dispersion forces between the atoms and the absence of other interatomic interactions.The direct reaction of xenon with fluorine leads to a series of compounds with oxidation numbers +2, +4 and +6. XeF4 reacts violently with water to given XeO3. The compounds of xenon exhibit rich stereochemistry and their geometries can be deduced considering the total number of electron pairs in the valence shell.Q.XeF4 and XeF6 are expected to bea)oxidizingb)reducingc)unreactived)strongly basicCorrect answer is option 'A'. Can you explain this answer?.
Solutions for PASSAGE - 1The noble gases have closed-shell electronic configuration and are monoatomic gases under normal conditions. The low boiling points of the lighter noble gases are due to weak dispersion forces between the atoms and the absence of other interatomic interactions.The direct reaction of xenon with fluorine leads to a series of compounds with oxidation numbers +2, +4 and +6. XeF4 reacts violently with water to given XeO3. The compounds of xenon exhibit rich stereochemistry and their geometries can be deduced considering the total number of electron pairs in the valence shell.Q.XeF4 and XeF6 are expected to bea)oxidizingb)reducingc)unreactived)strongly basicCorrect answer is option 'A'. Can you explain this answer? in English & in Hindi are available as part of our courses for JEE.
Download more important topics, notes, lectures and mock test series for JEE Exam by signing up for free.
Here you can find the meaning of PASSAGE - 1The noble gases have closed-shell electronic configuration and are monoatomic gases under normal conditions. The low boiling points of the lighter noble gases are due to weak dispersion forces between the atoms and the absence of other interatomic interactions.The direct reaction of xenon with fluorine leads to a series of compounds with oxidation numbers +2, +4 and +6. XeF4 reacts violently with water to given XeO3. The compounds of xenon exhibit rich stereochemistry and their geometries can be deduced considering the total number of electron pairs in the valence shell.Q.XeF4 and XeF6 are expected to bea)oxidizingb)reducingc)unreactived)strongly basicCorrect answer is option 'A'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of
PASSAGE - 1The noble gases have closed-shell electronic configuration and are monoatomic gases under normal conditions. The low boiling points of the lighter noble gases are due to weak dispersion forces between the atoms and the absence of other interatomic interactions.The direct reaction of xenon with fluorine leads to a series of compounds with oxidation numbers +2, +4 and +6. XeF4 reacts violently with water to given XeO3. The compounds of xenon exhibit rich stereochemistry and their geometries can be deduced considering the total number of electron pairs in the valence shell.Q.XeF4 and XeF6 are expected to bea)oxidizingb)reducingc)unreactived)strongly basicCorrect answer is option 'A'. Can you explain this answer?, a detailed solution for PASSAGE - 1The noble gases have closed-shell electronic configuration and are monoatomic gases under normal conditions. The low boiling points of the lighter noble gases are due to weak dispersion forces between the atoms and the absence of other interatomic interactions.The direct reaction of xenon with fluorine leads to a series of compounds with oxidation numbers +2, +4 and +6. XeF4 reacts violently with water to given XeO3. The compounds of xenon exhibit rich stereochemistry and their geometries can be deduced considering the total number of electron pairs in the valence shell.Q.XeF4 and XeF6 are expected to bea)oxidizingb)reducingc)unreactived)strongly basicCorrect answer is option 'A'. Can you explain this answer? has been provided alongside types of PASSAGE - 1The noble gases have closed-shell electronic configuration and are monoatomic gases under normal conditions. The low boiling points of the lighter noble gases are due to weak dispersion forces between the atoms and the absence of other interatomic interactions.The direct reaction of xenon with fluorine leads to a series of compounds with oxidation numbers +2, +4 and +6. XeF4 reacts violently with water to given XeO3. The compounds of xenon exhibit rich stereochemistry and their geometries can be deduced considering the total number of electron pairs in the valence shell.Q.XeF4 and XeF6 are expected to bea)oxidizingb)reducingc)unreactived)strongly basicCorrect answer is option 'A'. Can you explain this answer? theory, EduRev gives you an
ample number of questions to practice PASSAGE - 1The noble gases have closed-shell electronic configuration and are monoatomic gases under normal conditions. The low boiling points of the lighter noble gases are due to weak dispersion forces between the atoms and the absence of other interatomic interactions.The direct reaction of xenon with fluorine leads to a series of compounds with oxidation numbers +2, +4 and +6. XeF4 reacts violently with water to given XeO3. The compounds of xenon exhibit rich stereochemistry and their geometries can be deduced considering the total number of electron pairs in the valence shell.Q.XeF4 and XeF6 are expected to bea)oxidizingb)reducingc)unreactived)strongly basicCorrect answer is option 'A'. Can you explain this answer? tests, examples and also practice JEE tests.

|
Explore Courses for JEE exam
|

|
Suggested Free Tests
Signup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.
























