Physics Exam > Physics Questions > The ultraviolet Lyman αline of hydrogen... Start Learning for Free
The ultraviolet Lyman α line of hydrogen with wavelength 121.5 nm is emitted by an astronomical object. An observer on earth measures the wavelength of the light received from the object to be 607.5 nm. The observer can conclude that the object is moving with a radial velocity of :
- a)2.8 × 108 m/s away from Earth
- b)2.4 × 108 m/s towards Earth
- c)2.4 × 108 m/s away from Earth
- d)2.8 × 108 m/s towards Earth
Correct answer is option 'A'. Can you explain this answer?
Verified Answer
The ultraviolet Lyman αline of hydrogen with wavelength 121.5nm ...
Since the light received has a higher wavelength than the incident wavelength, one concludes that this is a red shift, that the object is moving away from the Earth.
where λ0 is the wavelength of the source.
The doppler effect equation gives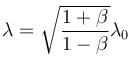
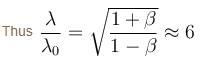
Solving this one gets β = 35/37
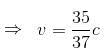
v = 2.8 × 108 m/s
away from the Earth.
The doppler effect equation gives
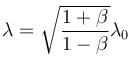
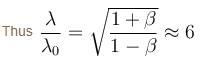
Solving this one gets β = 35/37
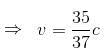
v = 2.8 × 108 m/s
away from the Earth.
The correct answer is: 2.8 × 108 m/s away from Earth
Most Upvoted Answer
The ultraviolet Lyman αline of hydrogen with wavelength 121.5nm ...
Series refers to a series of spectral lines in the ultraviolet region of the electromagnetic spectrum. These lines are associated with the transition of an electron in a hydrogen atom from a higher energy level to the ground state. The Lyman series is named after its discoverer, Theodore Lyman.
The Lyman series is characterized by a series of discrete lines with increasing wavelengths as the electron transitions from higher energy levels to the ground state. The first line in the series, known as Lyman alpha, is the transition from the n=2 energy level to the n=1 energy level. Subsequent lines in the series correspond to transitions from higher energy levels to the n=1 energy level.
The Lyman series is significant because it provides valuable information about the electronic structure of atoms, particularly hydrogen. By studying the wavelengths and energies of the Lyman series lines, scientists can determine the energy levels and transitions of electrons within atoms. This information is important for understanding atomic structure and behavior, as well as for various applications in fields such as astrophysics and spectroscopy.
The Lyman series is characterized by a series of discrete lines with increasing wavelengths as the electron transitions from higher energy levels to the ground state. The first line in the series, known as Lyman alpha, is the transition from the n=2 energy level to the n=1 energy level. Subsequent lines in the series correspond to transitions from higher energy levels to the n=1 energy level.
The Lyman series is significant because it provides valuable information about the electronic structure of atoms, particularly hydrogen. By studying the wavelengths and energies of the Lyman series lines, scientists can determine the energy levels and transitions of electrons within atoms. This information is important for understanding atomic structure and behavior, as well as for various applications in fields such as astrophysics and spectroscopy.
 | Explore Courses for Physics exam |  |
Question Description
The ultraviolet Lyman αline of hydrogen with wavelength 121.5nm is emitted by an astronomical object. An observer on earth measures the wavelength of the light received from the object to be 607.5nm. The observer can conclude that the object is moving with a radial velocity of :a)2.8 × 108m/saway from Earthb)2.4 × 108m/stowards Earthc)2.4 × 108m/saway from Earthd)2.8 × 108m/stowards EarthCorrect answer is option 'A'. Can you explain this answer? for Physics 2026 is part of Physics preparation. The Question and answers have been prepared according to the Physics exam syllabus. Information about The ultraviolet Lyman αline of hydrogen with wavelength 121.5nm is emitted by an astronomical object. An observer on earth measures the wavelength of the light received from the object to be 607.5nm. The observer can conclude that the object is moving with a radial velocity of :a)2.8 × 108m/saway from Earthb)2.4 × 108m/stowards Earthc)2.4 × 108m/saway from Earthd)2.8 × 108m/stowards EarthCorrect answer is option 'A'. Can you explain this answer? covers all topics & solutions for Physics 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for The ultraviolet Lyman αline of hydrogen with wavelength 121.5nm is emitted by an astronomical object. An observer on earth measures the wavelength of the light received from the object to be 607.5nm. The observer can conclude that the object is moving with a radial velocity of :a)2.8 × 108m/saway from Earthb)2.4 × 108m/stowards Earthc)2.4 × 108m/saway from Earthd)2.8 × 108m/stowards EarthCorrect answer is option 'A'. Can you explain this answer?.
The ultraviolet Lyman αline of hydrogen with wavelength 121.5nm is emitted by an astronomical object. An observer on earth measures the wavelength of the light received from the object to be 607.5nm. The observer can conclude that the object is moving with a radial velocity of :a)2.8 × 108m/saway from Earthb)2.4 × 108m/stowards Earthc)2.4 × 108m/saway from Earthd)2.8 × 108m/stowards EarthCorrect answer is option 'A'. Can you explain this answer? for Physics 2026 is part of Physics preparation. The Question and answers have been prepared according to the Physics exam syllabus. Information about The ultraviolet Lyman αline of hydrogen with wavelength 121.5nm is emitted by an astronomical object. An observer on earth measures the wavelength of the light received from the object to be 607.5nm. The observer can conclude that the object is moving with a radial velocity of :a)2.8 × 108m/saway from Earthb)2.4 × 108m/stowards Earthc)2.4 × 108m/saway from Earthd)2.8 × 108m/stowards EarthCorrect answer is option 'A'. Can you explain this answer? covers all topics & solutions for Physics 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for The ultraviolet Lyman αline of hydrogen with wavelength 121.5nm is emitted by an astronomical object. An observer on earth measures the wavelength of the light received from the object to be 607.5nm. The observer can conclude that the object is moving with a radial velocity of :a)2.8 × 108m/saway from Earthb)2.4 × 108m/stowards Earthc)2.4 × 108m/saway from Earthd)2.8 × 108m/stowards EarthCorrect answer is option 'A'. Can you explain this answer?.
Solutions for The ultraviolet Lyman αline of hydrogen with wavelength 121.5nm is emitted by an astronomical object. An observer on earth measures the wavelength of the light received from the object to be 607.5nm. The observer can conclude that the object is moving with a radial velocity of :a)2.8 × 108m/saway from Earthb)2.4 × 108m/stowards Earthc)2.4 × 108m/saway from Earthd)2.8 × 108m/stowards EarthCorrect answer is option 'A'. Can you explain this answer? in English & in Hindi are available as part of our courses for Physics. Download more important topics, notes, lectures and mock test series for Physics Exam by signing up for free.
Here you can find the meaning of The ultraviolet Lyman αline of hydrogen with wavelength 121.5nm is emitted by an astronomical object. An observer on earth measures the wavelength of the light received from the object to be 607.5nm. The observer can conclude that the object is moving with a radial velocity of :a)2.8 × 108m/saway from Earthb)2.4 × 108m/stowards Earthc)2.4 × 108m/saway from Earthd)2.8 × 108m/stowards EarthCorrect answer is option 'A'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of The ultraviolet Lyman αline of hydrogen with wavelength 121.5nm is emitted by an astronomical object. An observer on earth measures the wavelength of the light received from the object to be 607.5nm. The observer can conclude that the object is moving with a radial velocity of :a)2.8 × 108m/saway from Earthb)2.4 × 108m/stowards Earthc)2.4 × 108m/saway from Earthd)2.8 × 108m/stowards EarthCorrect answer is option 'A'. Can you explain this answer?, a detailed solution for The ultraviolet Lyman αline of hydrogen with wavelength 121.5nm is emitted by an astronomical object. An observer on earth measures the wavelength of the light received from the object to be 607.5nm. The observer can conclude that the object is moving with a radial velocity of :a)2.8 × 108m/saway from Earthb)2.4 × 108m/stowards Earthc)2.4 × 108m/saway from Earthd)2.8 × 108m/stowards EarthCorrect answer is option 'A'. Can you explain this answer? has been provided alongside types of The ultraviolet Lyman αline of hydrogen with wavelength 121.5nm is emitted by an astronomical object. An observer on earth measures the wavelength of the light received from the object to be 607.5nm. The observer can conclude that the object is moving with a radial velocity of :a)2.8 × 108m/saway from Earthb)2.4 × 108m/stowards Earthc)2.4 × 108m/saway from Earthd)2.8 × 108m/stowards EarthCorrect answer is option 'A'. Can you explain this answer? theory, EduRev gives you an ample number of questions to practice The ultraviolet Lyman αline of hydrogen with wavelength 121.5nm is emitted by an astronomical object. An observer on earth measures the wavelength of the light received from the object to be 607.5nm. The observer can conclude that the object is moving with a radial velocity of :a)2.8 × 108m/saway from Earthb)2.4 × 108m/stowards Earthc)2.4 × 108m/saway from Earthd)2.8 × 108m/stowards EarthCorrect answer is option 'A'. Can you explain this answer? tests, examples and also practice Physics tests.
 | Explore Courses for Physics exam |  |
Top Courses for Physics
Explore CoursesSignup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.






