JEE Exam > JEE Questions > The decreasing values of bond angles from NH3...
Start Learning for Free
The decreasing values of bond angles from NH3 (106º) to SbH3 (101º) down group-15 of the periodic table is due to [2006]
- a)decreasing lp-bp repulsion
- b)decreasing electronegativity
- c)increasing bp-bp repulsion
- d)increasing p-orbital character in sp3
Correct answer is option 'B'. Can you explain this answer?
| FREE This question is part of | Download PDF Attempt this Test |
Verified Answer
The decreasing values of bond angles from NH3 (106º) to SbH3 (101...
The bond angle decreases on moving down the group due to decrease in bond pair-bond pair repulsion.
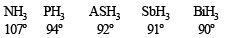
NOTE : This can also be explained by the fact that as the size of central atom increases sp3 hybrid orbital becomes more distinct with increasing size of central atom i.e. pure p- orbitals are utilized in M–H bonding
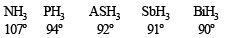
NOTE : This can also be explained by the fact that as the size of central atom increases sp3 hybrid orbital becomes more distinct with increasing size of central atom i.e. pure p- orbitals are utilized in M–H bonding
Most Upvoted Answer
The decreasing values of bond angles from NH3 (106º) to SbH3 (101...
Explanation:
Electronegativity:
- The decreasing values of bond angles from NH3 to SbH3 down Group-15 of the periodic table can be attributed to the decreasing electronegativity of the central atom.
- Electronegativity is the ability of an atom to attract shared electrons towards itself in a covalent bond.
- As we move down the group from nitrogen to antimony, the electronegativity of the central atom decreases.
- This results in a weaker pull on the bonding electron pairs, leading to a larger bond angle.
Effect on Bond Angles:
- In NH3 (ammonia), nitrogen is more electronegative compared to antimony in SbH3 (antimony hydride).
- The greater electronegativity of nitrogen results in stronger electron pair-bond pair repulsions, leading to a smaller bond angle in NH3.
- As we move down the group, the decreasing electronegativity of the central atom reduces the repulsion between the lone pair and bond pairs, allowing for a larger bond angle in SbH3.
Conclusion:
- Therefore, the decreasing values of bond angles from NH3 to SbH3 down Group-15 of the periodic table are primarily due to the decreasing electronegativity of the central atom.
- This change in electronegativity affects the repulsion between electron pairs, leading to variations in bond angles across the group.
Electronegativity:
- The decreasing values of bond angles from NH3 to SbH3 down Group-15 of the periodic table can be attributed to the decreasing electronegativity of the central atom.
- Electronegativity is the ability of an atom to attract shared electrons towards itself in a covalent bond.
- As we move down the group from nitrogen to antimony, the electronegativity of the central atom decreases.
- This results in a weaker pull on the bonding electron pairs, leading to a larger bond angle.
Effect on Bond Angles:
- In NH3 (ammonia), nitrogen is more electronegative compared to antimony in SbH3 (antimony hydride).
- The greater electronegativity of nitrogen results in stronger electron pair-bond pair repulsions, leading to a smaller bond angle in NH3.
- As we move down the group, the decreasing electronegativity of the central atom reduces the repulsion between the lone pair and bond pairs, allowing for a larger bond angle in SbH3.
Conclusion:
- Therefore, the decreasing values of bond angles from NH3 to SbH3 down Group-15 of the periodic table are primarily due to the decreasing electronegativity of the central atom.
- This change in electronegativity affects the repulsion between electron pairs, leading to variations in bond angles across the group.
Attention JEE Students!
To make sure you are not studying endlessly, EduRev has designed JEE study material, with Structured Courses, Videos, & Test Series. Plus get personalized analysis, doubt solving and improvement plans to achieve a great score in JEE.

|
Explore Courses for JEE exam
|

|
The decreasing values of bond angles from NH3 (106º) to SbH3 (101º) down group-15 of the periodic table is due to [2006]a)decreasing lp-bp repulsionb)decreasing electronegativityc)increasing bp-bp repulsiond)increasing p-orbital character in sp3Correct answer is option 'B'. Can you explain this answer?
Question Description
The decreasing values of bond angles from NH3 (106º) to SbH3 (101º) down group-15 of the periodic table is due to [2006]a)decreasing lp-bp repulsionb)decreasing electronegativityc)increasing bp-bp repulsiond)increasing p-orbital character in sp3Correct answer is option 'B'. Can you explain this answer? for JEE 2024 is part of JEE preparation. The Question and answers have been prepared according to the JEE exam syllabus. Information about The decreasing values of bond angles from NH3 (106º) to SbH3 (101º) down group-15 of the periodic table is due to [2006]a)decreasing lp-bp repulsionb)decreasing electronegativityc)increasing bp-bp repulsiond)increasing p-orbital character in sp3Correct answer is option 'B'. Can you explain this answer? covers all topics & solutions for JEE 2024 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for The decreasing values of bond angles from NH3 (106º) to SbH3 (101º) down group-15 of the periodic table is due to [2006]a)decreasing lp-bp repulsionb)decreasing electronegativityc)increasing bp-bp repulsiond)increasing p-orbital character in sp3Correct answer is option 'B'. Can you explain this answer?.
The decreasing values of bond angles from NH3 (106º) to SbH3 (101º) down group-15 of the periodic table is due to [2006]a)decreasing lp-bp repulsionb)decreasing electronegativityc)increasing bp-bp repulsiond)increasing p-orbital character in sp3Correct answer is option 'B'. Can you explain this answer? for JEE 2024 is part of JEE preparation. The Question and answers have been prepared according to the JEE exam syllabus. Information about The decreasing values of bond angles from NH3 (106º) to SbH3 (101º) down group-15 of the periodic table is due to [2006]a)decreasing lp-bp repulsionb)decreasing electronegativityc)increasing bp-bp repulsiond)increasing p-orbital character in sp3Correct answer is option 'B'. Can you explain this answer? covers all topics & solutions for JEE 2024 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for The decreasing values of bond angles from NH3 (106º) to SbH3 (101º) down group-15 of the periodic table is due to [2006]a)decreasing lp-bp repulsionb)decreasing electronegativityc)increasing bp-bp repulsiond)increasing p-orbital character in sp3Correct answer is option 'B'. Can you explain this answer?.
Solutions for The decreasing values of bond angles from NH3 (106º) to SbH3 (101º) down group-15 of the periodic table is due to [2006]a)decreasing lp-bp repulsionb)decreasing electronegativityc)increasing bp-bp repulsiond)increasing p-orbital character in sp3Correct answer is option 'B'. Can you explain this answer? in English & in Hindi are available as part of our courses for JEE.
Download more important topics, notes, lectures and mock test series for JEE Exam by signing up for free.
Here you can find the meaning of The decreasing values of bond angles from NH3 (106º) to SbH3 (101º) down group-15 of the periodic table is due to [2006]a)decreasing lp-bp repulsionb)decreasing electronegativityc)increasing bp-bp repulsiond)increasing p-orbital character in sp3Correct answer is option 'B'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of
The decreasing values of bond angles from NH3 (106º) to SbH3 (101º) down group-15 of the periodic table is due to [2006]a)decreasing lp-bp repulsionb)decreasing electronegativityc)increasing bp-bp repulsiond)increasing p-orbital character in sp3Correct answer is option 'B'. Can you explain this answer?, a detailed solution for The decreasing values of bond angles from NH3 (106º) to SbH3 (101º) down group-15 of the periodic table is due to [2006]a)decreasing lp-bp repulsionb)decreasing electronegativityc)increasing bp-bp repulsiond)increasing p-orbital character in sp3Correct answer is option 'B'. Can you explain this answer? has been provided alongside types of The decreasing values of bond angles from NH3 (106º) to SbH3 (101º) down group-15 of the periodic table is due to [2006]a)decreasing lp-bp repulsionb)decreasing electronegativityc)increasing bp-bp repulsiond)increasing p-orbital character in sp3Correct answer is option 'B'. Can you explain this answer? theory, EduRev gives you an
ample number of questions to practice The decreasing values of bond angles from NH3 (106º) to SbH3 (101º) down group-15 of the periodic table is due to [2006]a)decreasing lp-bp repulsionb)decreasing electronegativityc)increasing bp-bp repulsiond)increasing p-orbital character in sp3Correct answer is option 'B'. Can you explain this answer? tests, examples and also practice JEE tests.

|
Explore Courses for JEE exam
|

|
Suggested Free Tests
Signup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.






















