Class 10 Exam > Class 10 Questions > What is triple bond or triple covalent bond? ... Start Learning for Free
What is triple bond or triple covalent bond? explain the formation of triple bond giving two examples.?
Verified Answer
What is triple bond or triple covalent bond? explain the formation of ...
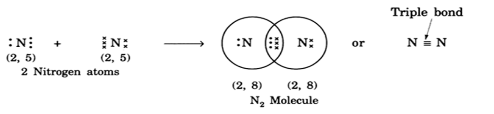
The bond formed between two atoms through the sharing of three electron pairs is called a triple bond. Three horizontal lines between two atoms denote a triple bond, e.g., nitrogen molecule is given asN≡N.
(i) Formation of N2 molecule: Nitrogen atoms have five electrons in their outermost shells. Therefore, in N2 molecules, two nitrogen atoms share 3 electrons with each other, thus acquiring the stable structure and forming a N2 molecule with triple bonds.
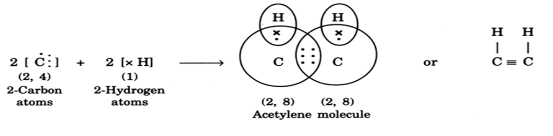
(ii) Formation of acetylene molecule (C2H2): In acetylene molecule, each carbon atom satisfies its valency by sharing one electron with a hydrogen atom and sharing the other three electrons with the other carbon atom as shown:
 This question is part of UPSC exam. View all Class 10 courses
This question is part of UPSC exam. View all Class 10 coursesMost Upvoted Answer
What is triple bond or triple covalent bond? explain the formation of ...
C - C - C this is the single bond
C = this the double bond. kisi bhi carbon ya element ke bich m ek choti si line ho usse single bond khete h.
veese hi kisi bhi carbon ya element ke bich m 3 line ho usse tiple bond khete h
C = this the double bond. kisi bhi carbon ya element ke bich m ek choti si line ho usse single bond khete h.
veese hi kisi bhi carbon ya element ke bich m 3 line ho usse tiple bond khete h
Community Answer
What is triple bond or triple covalent bond? explain the formation of ...
Triples Bond or Triple Covalent Bond:
A triple bond, also known as a triple covalent bond, is a type of chemical bond formed between two atoms when they share three pairs of electrons. It is one of the strongest types of covalent bonds and is characterized by its high bond energy and short bond length.
Formation of Triple Bond:
The formation of a triple bond involves the sharing of three pairs of electrons between two atoms. This occurs when two atoms have a strong attraction for each other and need to achieve a stable electron configuration.
Here are the steps involved in the formation of a triple bond:
1. Overlap of Atomic Orbitals:
The atoms involved in the bond must have unpaired electrons in their valence shells. The atomic orbitals of these atoms overlap to allow the sharing of electrons. In the case of a triple bond, three atomic orbitals overlap.
2. Sharing of Electrons:
Once the atomic orbitals overlap, the electrons are shared between the two atoms. In a triple bond, each atom contributes three electrons, resulting in the formation of three shared electron pairs. This sharing allows both atoms to achieve a more stable electron configuration.
3. Formation of Sigma Bond:
The first electron pair is shared through the end-to-end overlap of atomic orbitals, resulting in the formation of a sigma (σ) bond. This sigma bond is formed by the overlap of two s orbitals or one s orbital and one p orbital.
4. Formation of Pi Bonds:
The remaining two electron pairs are shared through sideways overlap of p orbitals. This results in the formation of two pi (π) bonds. Pi bonds are weaker than sigma bonds and are formed by the side-to-side overlap of p orbitals.
Examples of Triple Bonds:
1. Nitrogen Molecule (N2):
In a nitrogen molecule, two nitrogen atoms are connected by a triple bond. Each nitrogen atom contributes three electrons, resulting in the sharing of six electrons. The first bond is a sigma bond formed by the overlap of two s orbitals, while the remaining two bonds are pi bonds formed by the overlap of two sets of p orbitals.
2. Acetylene (C2H2):
Acetylene is a hydrocarbon with the molecular formula C2H2. It consists of two carbon atoms connected by a triple bond. Each carbon atom contributes three electrons, resulting in the sharing of six electrons. The first bond is a sigma bond formed by the overlap of two sp hybrid orbitals, while the remaining two bonds are pi bonds formed by the overlap of two sets of p orbitals.
In conclusion, a triple bond or triple covalent bond is formed by the sharing of three pairs of electrons between two atoms. It involves the overlap of atomic orbitals and the formation of sigma and pi bonds. Two examples of compounds with triple bonds are nitrogen molecule (N2) and acetylene (C2H2).
A triple bond, also known as a triple covalent bond, is a type of chemical bond formed between two atoms when they share three pairs of electrons. It is one of the strongest types of covalent bonds and is characterized by its high bond energy and short bond length.
Formation of Triple Bond:
The formation of a triple bond involves the sharing of three pairs of electrons between two atoms. This occurs when two atoms have a strong attraction for each other and need to achieve a stable electron configuration.
Here are the steps involved in the formation of a triple bond:
1. Overlap of Atomic Orbitals:
The atoms involved in the bond must have unpaired electrons in their valence shells. The atomic orbitals of these atoms overlap to allow the sharing of electrons. In the case of a triple bond, three atomic orbitals overlap.
2. Sharing of Electrons:
Once the atomic orbitals overlap, the electrons are shared between the two atoms. In a triple bond, each atom contributes three electrons, resulting in the formation of three shared electron pairs. This sharing allows both atoms to achieve a more stable electron configuration.
3. Formation of Sigma Bond:
The first electron pair is shared through the end-to-end overlap of atomic orbitals, resulting in the formation of a sigma (σ) bond. This sigma bond is formed by the overlap of two s orbitals or one s orbital and one p orbital.
4. Formation of Pi Bonds:
The remaining two electron pairs are shared through sideways overlap of p orbitals. This results in the formation of two pi (π) bonds. Pi bonds are weaker than sigma bonds and are formed by the side-to-side overlap of p orbitals.
Examples of Triple Bonds:
1. Nitrogen Molecule (N2):
In a nitrogen molecule, two nitrogen atoms are connected by a triple bond. Each nitrogen atom contributes three electrons, resulting in the sharing of six electrons. The first bond is a sigma bond formed by the overlap of two s orbitals, while the remaining two bonds are pi bonds formed by the overlap of two sets of p orbitals.
2. Acetylene (C2H2):
Acetylene is a hydrocarbon with the molecular formula C2H2. It consists of two carbon atoms connected by a triple bond. Each carbon atom contributes three electrons, resulting in the sharing of six electrons. The first bond is a sigma bond formed by the overlap of two sp hybrid orbitals, while the remaining two bonds are pi bonds formed by the overlap of two sets of p orbitals.
In conclusion, a triple bond or triple covalent bond is formed by the sharing of three pairs of electrons between two atoms. It involves the overlap of atomic orbitals and the formation of sigma and pi bonds. Two examples of compounds with triple bonds are nitrogen molecule (N2) and acetylene (C2H2).
 | Explore Courses for Class 10 exam |  |
Question Description
What is triple bond or triple covalent bond? explain the formation of triple bond giving two examples.? for Class 10 2026 is part of Class 10 preparation. The Question and answers have been prepared according to the Class 10 exam syllabus. Information about What is triple bond or triple covalent bond? explain the formation of triple bond giving two examples.? covers all topics & solutions for Class 10 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for What is triple bond or triple covalent bond? explain the formation of triple bond giving two examples.?.
What is triple bond or triple covalent bond? explain the formation of triple bond giving two examples.? for Class 10 2026 is part of Class 10 preparation. The Question and answers have been prepared according to the Class 10 exam syllabus. Information about What is triple bond or triple covalent bond? explain the formation of triple bond giving two examples.? covers all topics & solutions for Class 10 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for What is triple bond or triple covalent bond? explain the formation of triple bond giving two examples.?.
Solutions for What is triple bond or triple covalent bond? explain the formation of triple bond giving two examples.? in English & in Hindi are available as part of our courses for Class 10. Download more important topics, notes, lectures and mock test series for Class 10 Exam by signing up for free.
Here you can find the meaning of What is triple bond or triple covalent bond? explain the formation of triple bond giving two examples.? defined & explained in the simplest way possible. Besides giving the explanation of What is triple bond or triple covalent bond? explain the formation of triple bond giving two examples.?, a detailed solution for What is triple bond or triple covalent bond? explain the formation of triple bond giving two examples.? has been provided alongside types of What is triple bond or triple covalent bond? explain the formation of triple bond giving two examples.? theory, EduRev gives you an ample number of questions to practice What is triple bond or triple covalent bond? explain the formation of triple bond giving two examples.? tests, examples and also practice Class 10 tests.
 | Explore Courses for Class 10 exam |  |
Top Courses for Class 10
Explore CoursesSignup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.






