NEET Exam > NEET Questions > The alkyl halide is converted into ethyl alco... Start Learning for Free
The alkyl halide is converted into ethyl alcohol by
- a)addition reaction
- b)substitution reaction
- c)elimination reaction
- d)dehydrohalogenation reaction
Correct answer is option 'B'. Can you explain this answer?
Most Upvoted Answer
The alkyl halide is converted into ethyl alcohol bya)addition reaction...
Free Test
FREE
| Start Free Test |
Community Answer
The alkyl halide is converted into ethyl alcohol bya)addition reaction...
Conversion of Alkyl Halide to Ethyl Alcohol
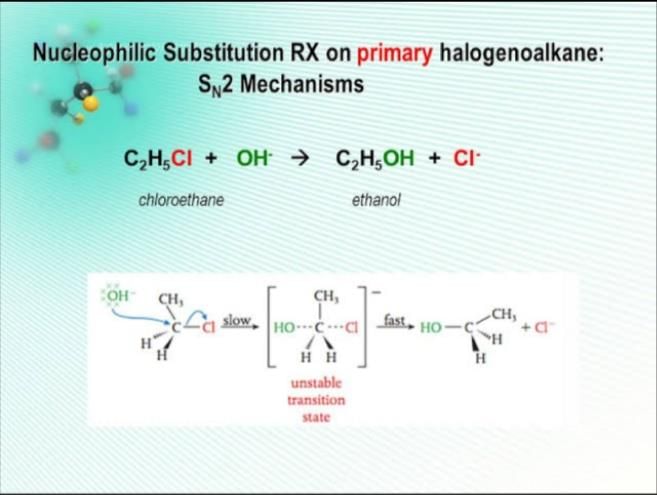
The conversion of an alkyl halide into ethyl alcohol involves a specific type of chemical reaction known as a substitution reaction. Here's a detailed explanation:
What is an Alkyl Halide?
- Alkyl halides are organic compounds containing carbon, hydrogen, and halogen atoms.
- They are often reactive due to the polar bond between carbon and the halogen.
Substitution Reaction Explained
- In a substitution reaction, one functional group in a molecule is replaced by another.
- For alkyl halides, the halogen atom is replaced by a hydroxyl group (-OH) to form an alcohol.
Mechanism of the Reaction
- When an alkyl halide reacts with a nucleophile like hydroxide ion (OH-), the nucleophile attacks the carbon atom bonded to the halogen.
- The halogen leaves, and the hydroxyl group takes its place, resulting in the formation of ethyl alcohol (ethanol).
Example of the Reaction
- For instance, when bromoethane (C2H5Br) reacts with sodium hydroxide (NaOH):
C2H5Br + NaOH → C2H5OH + NaBr
- Here, bromoethane is converted to ethanol.
Why Not Other Reaction Types?
- Addition Reaction: Involves adding atoms to a molecule without removing any atoms (not applicable here).
- Elimination Reaction: Involves the removal of elements from a molecule (not the case).
- Dehydrohalogenation Reaction: Specifically refers to the removal of a hydrogen and halogen to form alkenes, not alcohols.
Conclusion
The correct classification of the reaction that converts an alkyl halide to ethyl alcohol is indeed a substitution reaction, making option 'B' the right choice.
The conversion of an alkyl halide into ethyl alcohol involves a specific type of chemical reaction known as a substitution reaction. Here's a detailed explanation:
What is an Alkyl Halide?
- Alkyl halides are organic compounds containing carbon, hydrogen, and halogen atoms.
- They are often reactive due to the polar bond between carbon and the halogen.
Substitution Reaction Explained
- In a substitution reaction, one functional group in a molecule is replaced by another.
- For alkyl halides, the halogen atom is replaced by a hydroxyl group (-OH) to form an alcohol.
Mechanism of the Reaction
- When an alkyl halide reacts with a nucleophile like hydroxide ion (OH-), the nucleophile attacks the carbon atom bonded to the halogen.
- The halogen leaves, and the hydroxyl group takes its place, resulting in the formation of ethyl alcohol (ethanol).
Example of the Reaction
- For instance, when bromoethane (C2H5Br) reacts with sodium hydroxide (NaOH):
C2H5Br + NaOH → C2H5OH + NaBr
- Here, bromoethane is converted to ethanol.
Why Not Other Reaction Types?
- Addition Reaction: Involves adding atoms to a molecule without removing any atoms (not applicable here).
- Elimination Reaction: Involves the removal of elements from a molecule (not the case).
- Dehydrohalogenation Reaction: Specifically refers to the removal of a hydrogen and halogen to form alkenes, not alcohols.
Conclusion
The correct classification of the reaction that converts an alkyl halide to ethyl alcohol is indeed a substitution reaction, making option 'B' the right choice.
 | Explore Courses for NEET exam |  |
Question Description
The alkyl halide is converted into ethyl alcohol bya)addition reactionb)substitution reactionc)elimination reactiond)dehydrohalogenation reactionCorrect answer is option 'B'. Can you explain this answer? for NEET 2026 is part of NEET preparation. The Question and answers have been prepared according to the NEET exam syllabus. Information about The alkyl halide is converted into ethyl alcohol bya)addition reactionb)substitution reactionc)elimination reactiond)dehydrohalogenation reactionCorrect answer is option 'B'. Can you explain this answer? covers all topics & solutions for NEET 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for The alkyl halide is converted into ethyl alcohol bya)addition reactionb)substitution reactionc)elimination reactiond)dehydrohalogenation reactionCorrect answer is option 'B'. Can you explain this answer?.
The alkyl halide is converted into ethyl alcohol bya)addition reactionb)substitution reactionc)elimination reactiond)dehydrohalogenation reactionCorrect answer is option 'B'. Can you explain this answer? for NEET 2026 is part of NEET preparation. The Question and answers have been prepared according to the NEET exam syllabus. Information about The alkyl halide is converted into ethyl alcohol bya)addition reactionb)substitution reactionc)elimination reactiond)dehydrohalogenation reactionCorrect answer is option 'B'. Can you explain this answer? covers all topics & solutions for NEET 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for The alkyl halide is converted into ethyl alcohol bya)addition reactionb)substitution reactionc)elimination reactiond)dehydrohalogenation reactionCorrect answer is option 'B'. Can you explain this answer?.
Solutions for The alkyl halide is converted into ethyl alcohol bya)addition reactionb)substitution reactionc)elimination reactiond)dehydrohalogenation reactionCorrect answer is option 'B'. Can you explain this answer? in English & in Hindi are available as part of our courses for NEET. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
Here you can find the meaning of The alkyl halide is converted into ethyl alcohol bya)addition reactionb)substitution reactionc)elimination reactiond)dehydrohalogenation reactionCorrect answer is option 'B'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of The alkyl halide is converted into ethyl alcohol bya)addition reactionb)substitution reactionc)elimination reactiond)dehydrohalogenation reactionCorrect answer is option 'B'. Can you explain this answer?, a detailed solution for The alkyl halide is converted into ethyl alcohol bya)addition reactionb)substitution reactionc)elimination reactiond)dehydrohalogenation reactionCorrect answer is option 'B'. Can you explain this answer? has been provided alongside types of The alkyl halide is converted into ethyl alcohol bya)addition reactionb)substitution reactionc)elimination reactiond)dehydrohalogenation reactionCorrect answer is option 'B'. Can you explain this answer? theory, EduRev gives you an ample number of questions to practice The alkyl halide is converted into ethyl alcohol bya)addition reactionb)substitution reactionc)elimination reactiond)dehydrohalogenation reactionCorrect answer is option 'B'. Can you explain this answer? tests, examples and also practice NEET tests.
 | Explore Courses for NEET exam |  |
Top Courses for NEET
Explore CoursesSignup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.






