NEET Exam > NEET Questions > Which one of the followings has maximum numbe... Start Learning for Free
Which one of the followings has maximum number of atoms? (2020)
- a)1g of O2(g) (Atomic mass of O = 16)
- b)1g of Li(s) (Atomic mass of Li = 7)
- c)1g of Ag(s) (Atomic mass of Ag = 108)
- d)1g of Mg(s) (Atomic mass of Mg = 24)
Correct answer is option 'B'. Can you explain this answer?
Verified Answer
Which one of the followings has maximum number of atoms? (2020)a)1g ...
(a) no. of atoms
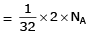
(b) no. of atoms
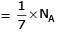
(c) no. of atoms
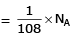
(d) no. of atoms
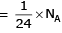
∴ 1 gm of Li is having maximum no. of atoms.
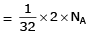
(b) no. of atoms
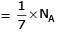
(c) no. of atoms
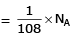
(d) no. of atoms
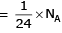
∴ 1 gm of Li is having maximum no. of atoms.
Most Upvoted Answer
Which one of the followings has maximum number of atoms? (2020)a)1g ...
Maximum number of atoms
Explanation:
The number of atoms in a substance depends on its atomic mass and Avogadro's number. Avogadro's number is the number of atoms in one mole of a substance, which is approximately 6.022 x 10^23.
To find the number of atoms in a given amount of substance, we need to convert the mass of the substance to moles using its atomic mass and then multiply by Avogadro's number.
Calculations:
a) 1g of O2(g)
Atomic mass of O = 16
Molar mass of O2 = (2 x 16) = 32 g/mol
Number of moles in 1g of O2 = 1/32 = 0.03125 mol
Number of atoms in 1g of O2 = 0.03125 x 6.022 x 10^23 = 1.88 x 10^22 atoms
b) 1g of Li(s)
Atomic mass of Li = 7
Molar mass of Li = 7 g/mol
Number of moles in 1g of Li = 1/7 = 0.1429 mol
Number of atoms in 1g of Li = 0.1429 x 6.022 x 10^23 = 8.61 x 10^22 atoms
c) 1g of Ag(s)
Atomic mass of Ag = 108
Molar mass of Ag = 108 g/mol
Number of moles in 1g of Ag = 1/108 = 0.009259 mol
Number of atoms in 1g of Ag = 0.009259 x 6.022 x 10^23 = 5.56 x 10^21 atoms
d) 1g of Mg(s)
Atomic mass of Mg = 24
Molar mass of Mg = 24 g/mol
Number of moles in 1g of Mg = 1/24 = 0.04167 mol
Number of atoms in 1g of Mg = 0.04167 x 6.022 x 10^23 = 2.51 x 10^22 atoms
Conclusion:
Therefore, the substance with the maximum number of atoms is 1g of Li(s) with 8.61 x 10^22 atoms.
Explanation:
The number of atoms in a substance depends on its atomic mass and Avogadro's number. Avogadro's number is the number of atoms in one mole of a substance, which is approximately 6.022 x 10^23.
To find the number of atoms in a given amount of substance, we need to convert the mass of the substance to moles using its atomic mass and then multiply by Avogadro's number.
Calculations:
a) 1g of O2(g)
Atomic mass of O = 16
Molar mass of O2 = (2 x 16) = 32 g/mol
Number of moles in 1g of O2 = 1/32 = 0.03125 mol
Number of atoms in 1g of O2 = 0.03125 x 6.022 x 10^23 = 1.88 x 10^22 atoms
b) 1g of Li(s)
Atomic mass of Li = 7
Molar mass of Li = 7 g/mol
Number of moles in 1g of Li = 1/7 = 0.1429 mol
Number of atoms in 1g of Li = 0.1429 x 6.022 x 10^23 = 8.61 x 10^22 atoms
c) 1g of Ag(s)
Atomic mass of Ag = 108
Molar mass of Ag = 108 g/mol
Number of moles in 1g of Ag = 1/108 = 0.009259 mol
Number of atoms in 1g of Ag = 0.009259 x 6.022 x 10^23 = 5.56 x 10^21 atoms
d) 1g of Mg(s)
Atomic mass of Mg = 24
Molar mass of Mg = 24 g/mol
Number of moles in 1g of Mg = 1/24 = 0.04167 mol
Number of atoms in 1g of Mg = 0.04167 x 6.022 x 10^23 = 2.51 x 10^22 atoms
Conclusion:
Therefore, the substance with the maximum number of atoms is 1g of Li(s) with 8.61 x 10^22 atoms.
Free Test
| FREE | Start Free Test |
Community Answer
Which one of the followings has maximum number of atoms? (2020)a)1g ...
Answer is
1g of Li (s) ,
,
1g of Li (s)
 ,
, | Explore Courses for NEET exam |  |
Question Description
Which one of the followings has maximum number of atoms? (2020)a)1g of O2(g) (Atomic mass of O = 16)b)1g of Li(s) (Atomic mass of Li = 7)c)1g of Ag(s) (Atomic mass of Ag = 108)d)1g of Mg(s) (Atomic mass of Mg = 24)Correct answer is option 'B'. Can you explain this answer? for NEET 2026 is part of NEET preparation. The Question and answers have been prepared according to the NEET exam syllabus. Information about Which one of the followings has maximum number of atoms? (2020)a)1g of O2(g) (Atomic mass of O = 16)b)1g of Li(s) (Atomic mass of Li = 7)c)1g of Ag(s) (Atomic mass of Ag = 108)d)1g of Mg(s) (Atomic mass of Mg = 24)Correct answer is option 'B'. Can you explain this answer? covers all topics & solutions for NEET 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Which one of the followings has maximum number of atoms? (2020)a)1g of O2(g) (Atomic mass of O = 16)b)1g of Li(s) (Atomic mass of Li = 7)c)1g of Ag(s) (Atomic mass of Ag = 108)d)1g of Mg(s) (Atomic mass of Mg = 24)Correct answer is option 'B'. Can you explain this answer?.
Which one of the followings has maximum number of atoms? (2020)a)1g of O2(g) (Atomic mass of O = 16)b)1g of Li(s) (Atomic mass of Li = 7)c)1g of Ag(s) (Atomic mass of Ag = 108)d)1g of Mg(s) (Atomic mass of Mg = 24)Correct answer is option 'B'. Can you explain this answer? for NEET 2026 is part of NEET preparation. The Question and answers have been prepared according to the NEET exam syllabus. Information about Which one of the followings has maximum number of atoms? (2020)a)1g of O2(g) (Atomic mass of O = 16)b)1g of Li(s) (Atomic mass of Li = 7)c)1g of Ag(s) (Atomic mass of Ag = 108)d)1g of Mg(s) (Atomic mass of Mg = 24)Correct answer is option 'B'. Can you explain this answer? covers all topics & solutions for NEET 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Which one of the followings has maximum number of atoms? (2020)a)1g of O2(g) (Atomic mass of O = 16)b)1g of Li(s) (Atomic mass of Li = 7)c)1g of Ag(s) (Atomic mass of Ag = 108)d)1g of Mg(s) (Atomic mass of Mg = 24)Correct answer is option 'B'. Can you explain this answer?.
Solutions for Which one of the followings has maximum number of atoms? (2020)a)1g of O2(g) (Atomic mass of O = 16)b)1g of Li(s) (Atomic mass of Li = 7)c)1g of Ag(s) (Atomic mass of Ag = 108)d)1g of Mg(s) (Atomic mass of Mg = 24)Correct answer is option 'B'. Can you explain this answer? in English & in Hindi are available as part of our courses for NEET. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
Here you can find the meaning of Which one of the followings has maximum number of atoms? (2020)a)1g of O2(g) (Atomic mass of O = 16)b)1g of Li(s) (Atomic mass of Li = 7)c)1g of Ag(s) (Atomic mass of Ag = 108)d)1g of Mg(s) (Atomic mass of Mg = 24)Correct answer is option 'B'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of Which one of the followings has maximum number of atoms? (2020)a)1g of O2(g) (Atomic mass of O = 16)b)1g of Li(s) (Atomic mass of Li = 7)c)1g of Ag(s) (Atomic mass of Ag = 108)d)1g of Mg(s) (Atomic mass of Mg = 24)Correct answer is option 'B'. Can you explain this answer?, a detailed solution for Which one of the followings has maximum number of atoms? (2020)a)1g of O2(g) (Atomic mass of O = 16)b)1g of Li(s) (Atomic mass of Li = 7)c)1g of Ag(s) (Atomic mass of Ag = 108)d)1g of Mg(s) (Atomic mass of Mg = 24)Correct answer is option 'B'. Can you explain this answer? has been provided alongside types of Which one of the followings has maximum number of atoms? (2020)a)1g of O2(g) (Atomic mass of O = 16)b)1g of Li(s) (Atomic mass of Li = 7)c)1g of Ag(s) (Atomic mass of Ag = 108)d)1g of Mg(s) (Atomic mass of Mg = 24)Correct answer is option 'B'. Can you explain this answer? theory, EduRev gives you an ample number of questions to practice Which one of the followings has maximum number of atoms? (2020)a)1g of O2(g) (Atomic mass of O = 16)b)1g of Li(s) (Atomic mass of Li = 7)c)1g of Ag(s) (Atomic mass of Ag = 108)d)1g of Mg(s) (Atomic mass of Mg = 24)Correct answer is option 'B'. Can you explain this answer? tests, examples and also practice NEET tests.
 | Explore Courses for NEET exam |  |
Top Courses for NEET
Explore CoursesSignup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.






