Class 10 Exam > Class 10 Questions > The number of electrons constituting one coul... Start Learning for Free
The number of electrons constituting one coulomb of charge is
- a)6.25 × 10–18
- b)6.1 × 10–19
- c)6.25 × 10–19
- d)6.25 × 1018
Correct answer is option 'D'. Can you explain this answer?
Most Upvoted Answer
The number of electrons constituting one coulomb of charge isa)6.25 &t...
Understanding Charge and Electrons
The question revolves around the relationship between electric charge and the number of electrons. To find the number of electrons in one coulomb of charge, we need to know the charge of a single electron.
Charge of an Electron
- Each electron carries a fundamental charge of approximately -1.6 × 10^-19 coulombs.
Calculating the Number of Electrons
To find the total number of electrons in one coulomb, we divide the total charge (1 coulomb) by the charge of one electron.
- Formula:
Number of electrons = Total charge / Charge of one electron
- Using the values:
Number of electrons = 1 C / (1.6 × 10^-19 C/electron)
- Performing the calculation:
Number of electrons = 1 / (1.6 × 10^-19)
≈ 6.25 × 10^18 electrons
Conclusion
Thus, one coulomb of charge is constituted by approximately 6.25 × 10^18 electrons, confirming that the correct answer is option 'D'.
This relationship underscores the concept of charge at the atomic level and illustrates how a seemingly large unit of charge like a coulomb is composed of an exceedingly vast number of elementary particles like electrons. Understanding this helps in grasping the fundamentals of electric charge and its quantization.
The question revolves around the relationship between electric charge and the number of electrons. To find the number of electrons in one coulomb of charge, we need to know the charge of a single electron.
Charge of an Electron
- Each electron carries a fundamental charge of approximately -1.6 × 10^-19 coulombs.
Calculating the Number of Electrons
To find the total number of electrons in one coulomb, we divide the total charge (1 coulomb) by the charge of one electron.
- Formula:
Number of electrons = Total charge / Charge of one electron
- Using the values:
Number of electrons = 1 C / (1.6 × 10^-19 C/electron)
- Performing the calculation:
Number of electrons = 1 / (1.6 × 10^-19)
≈ 6.25 × 10^18 electrons
Conclusion
Thus, one coulomb of charge is constituted by approximately 6.25 × 10^18 electrons, confirming that the correct answer is option 'D'.
This relationship underscores the concept of charge at the atomic level and illustrates how a seemingly large unit of charge like a coulomb is composed of an exceedingly vast number of elementary particles like electrons. Understanding this helps in grasping the fundamentals of electric charge and its quantization.
Free Test
FREE
| Start Free Test |
Community Answer
The number of electrons constituting one coulomb of charge isa)6.25 &t...
The charge on one electron, e = 1.6 × 10–19 C. Number of electrons constituting 1 coulomb is given by n = Q/e
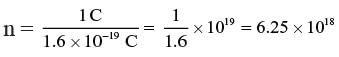
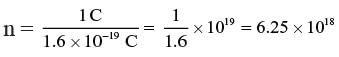
 | Explore Courses for Class 10 exam |  |
Question Description
The number of electrons constituting one coulomb of charge isa)6.25 × 10–18b)6.1 × 10–19c)6.25 × 10–19d)6.25 × 1018Correct answer is option 'D'. Can you explain this answer? for Class 10 2026 is part of Class 10 preparation. The Question and answers have been prepared according to the Class 10 exam syllabus. Information about The number of electrons constituting one coulomb of charge isa)6.25 × 10–18b)6.1 × 10–19c)6.25 × 10–19d)6.25 × 1018Correct answer is option 'D'. Can you explain this answer? covers all topics & solutions for Class 10 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for The number of electrons constituting one coulomb of charge isa)6.25 × 10–18b)6.1 × 10–19c)6.25 × 10–19d)6.25 × 1018Correct answer is option 'D'. Can you explain this answer?.
The number of electrons constituting one coulomb of charge isa)6.25 × 10–18b)6.1 × 10–19c)6.25 × 10–19d)6.25 × 1018Correct answer is option 'D'. Can you explain this answer? for Class 10 2026 is part of Class 10 preparation. The Question and answers have been prepared according to the Class 10 exam syllabus. Information about The number of electrons constituting one coulomb of charge isa)6.25 × 10–18b)6.1 × 10–19c)6.25 × 10–19d)6.25 × 1018Correct answer is option 'D'. Can you explain this answer? covers all topics & solutions for Class 10 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for The number of electrons constituting one coulomb of charge isa)6.25 × 10–18b)6.1 × 10–19c)6.25 × 10–19d)6.25 × 1018Correct answer is option 'D'. Can you explain this answer?.
Solutions for The number of electrons constituting one coulomb of charge isa)6.25 × 10–18b)6.1 × 10–19c)6.25 × 10–19d)6.25 × 1018Correct answer is option 'D'. Can you explain this answer? in English & in Hindi are available as part of our courses for Class 10. Download more important topics, notes, lectures and mock test series for Class 10 Exam by signing up for free.
Here you can find the meaning of The number of electrons constituting one coulomb of charge isa)6.25 × 10–18b)6.1 × 10–19c)6.25 × 10–19d)6.25 × 1018Correct answer is option 'D'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of The number of electrons constituting one coulomb of charge isa)6.25 × 10–18b)6.1 × 10–19c)6.25 × 10–19d)6.25 × 1018Correct answer is option 'D'. Can you explain this answer?, a detailed solution for The number of electrons constituting one coulomb of charge isa)6.25 × 10–18b)6.1 × 10–19c)6.25 × 10–19d)6.25 × 1018Correct answer is option 'D'. Can you explain this answer? has been provided alongside types of The number of electrons constituting one coulomb of charge isa)6.25 × 10–18b)6.1 × 10–19c)6.25 × 10–19d)6.25 × 1018Correct answer is option 'D'. Can you explain this answer? theory, EduRev gives you an ample number of questions to practice The number of electrons constituting one coulomb of charge isa)6.25 × 10–18b)6.1 × 10–19c)6.25 × 10–19d)6.25 × 1018Correct answer is option 'D'. Can you explain this answer? tests, examples and also practice Class 10 tests.
 | Explore Courses for Class 10 exam |  |
Top Courses for Class 10
Explore CoursesSignup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.






